MEETING OF THE LONDON SECTION, HELD AT THE HORSE SHOE HOTEL,
TOTTENHAM COURT ROAD, W.1, ON MONDAY, 11th JANUARY, 1960
Dr. J. I. Webb in the Chair
The following paper was read and discussed:
DEVELOPMENT OF BEER CANNING IN BRITAIN
by J. D. Felmingham, B.Sc, M.R.S.H., M.Inst.Pkg.
(The Metal Box Co., Ltd., Kendal Avenue, Acton, W.3)
Following a description of the construction of a flat-top beer can, it is shown that the pressure inside at a pasteurizing temperature of 140° F. is less than would be calculated from Henry’s Law; a pressure of 30-35 Ib. per sq. in. can be tolerated for short periods such as are involved in pasteurization, and constructional developments have ensured that there is no permanent distortion thereafter. Considerable attention has been given to lacquering problems, resulting in the initial use before forming of an oleoresinous lacquer, followed by a vinyl side-stripe after forming and a second vinyl spray; the ends receive an oleoresinous basecoat and a vinyl top coat. Can inspection is rigorously carried out visually, dimensionally, and by special tests, e.g., for exposed metal. Filling details must be considered in special relation to air elimination, and attention should also be paid to heating and cooling times in pasteurization. Beers for canning must be inherently of acceptable stability.
Introduction.— An enormous amount has been said and written concerning the commercial aspects of packaging beer—the reasons for canning, advantages and disadvantages of canning compared with bottling, the rate of increase in can-sales and the economics of canning.2
It is not the intention here to consider such commercial arguments but rather to start from the point at which it has been decided that the canning of beer is a worthwhile project and then to consider the consequent problems in both can manufacturing and canning.
When the canning of beer commenced in this country in 1935-36, there were two major obstacles to the use of a normal “open-top” food can, assuming that this could have been suitably lacquered. These were first, the special machinery that would be required by brewers for closing and, secondly, how to open it. As events turned out, these problems remained unresolved and the cone-top can came into existence. It is probably fair to say that the reception accorded to this can was never enthusiastic and its replacement by the flat-top beer can, once this container started to become established, was rapid and complete—there now being no further production of cone-top cans for beer in this country. Consequently it is not proposed to spend any time considering this container but to concentrate entirely upon the present package—the Oat-top beer can. Full scale production of the flat-top can commenced in 1955, by which time the major technical barriers had been broken but, naturally, there were refinements to come.
Can construction.—The open-top or sanitary can is fabricated from tinplate and consists of three components. First is the body cylinder, having an interlocking and soldered seam called the sideseam and second, the bottom or maker’s end which is mechanically seamed on to the body prior to leaving the can manufacturing factory. Thirdly, the top end, which is identical in construction to the bottom end, is supplied loose to the canner for subsequent seaming on to the body after the contents have been filled into the can; hence it is called the canner’s end and hence also, the derivation of the name open-top. The construction of such a can is shown in Fig. 1.
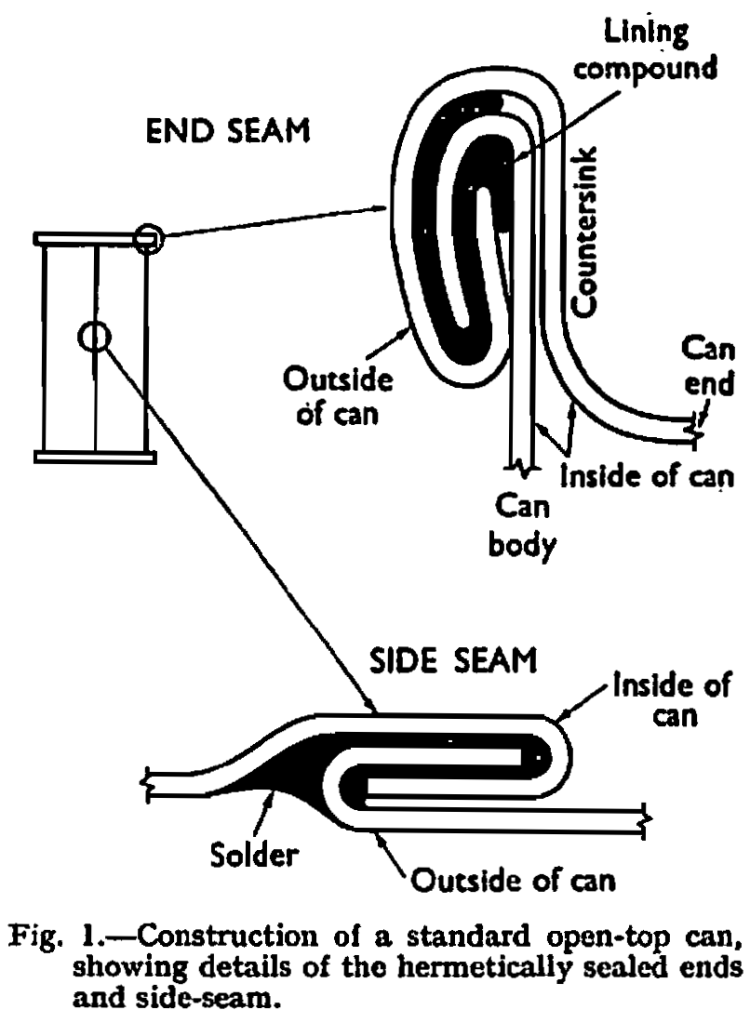
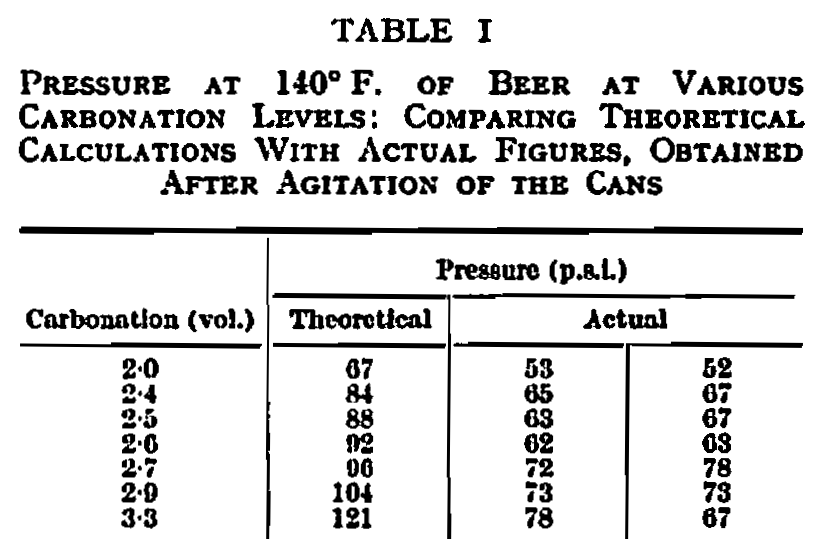
It is an accepted feature of the normal open-top can that, after the canning procedure has been completed, there is an internal vacuum, achieved by either hot filling or some means of mechanical vacuum closure. This internal vacuum naturally tends to pull in the can ends so that they are virtually fiat, or perhaps even show a slight concavity. If this is not so and the ends swell outwards, it is a recognized sign that the contents are unfit for use. When considering this type of container for beer, the first radical change in thinking and in can construction is concerned with this question of relative pressure. Not only has the can to withstand the continual pressure exerted by beer during storage at room temperature but, during the pasteurizing period, it has to withstand pressures which are very considerably higher. The theoretical pressure to be expected during a normal pasteurization at 140° F. can be calculated from Henry’s Law, which states that the partial pressure is proportional to the mass of dissolved gas. Figures for this theoretical pressure at various levels of carbonation are given in Table I, alongside figures which have been determined by practical experiment. These actual figures were recorded after the can contents had been allowed to reach 140° F. and after agitation. It will be seen that, whilst we have found that Henry’s Law is fairly closely obeyed at room temperature, the practical figures recorded at pasteurizing temperature are appreciably below those which have been derived theoretically. The reasons for this discrepancy are not fully understood but they are probably similar to those which govern wildness and gushing of beer, about which much has been written.1,4,14 However, despite this slight advantage in practical results over what would be expected according to theory, it will be apparent that very high pressures may be achieved. Other minor factors must be taken into consideration such as the partial pressure exerted by air residuals, and expansion of the pack which decreases headspace volume and the slight expansion of the container which has a reverse effect.
The conventional open-top can has a side-seam capable of withstanding pressures in the range of 30-35 lb. per sq. in. for short periods during the time that elapses after steam pressure processing has been completed and before cooling is appreciable; it is certainly not built to withstand the pressures of up to 80 lb. per sq. in. during beer pasteurization and up to 35 lb. per sq. in. continually during beer storage. This seam (see Fig. 1) is interlocked, hammered flat and then soldered. The solder normally penetrates into the first and second folds of the seam and, if the can is subjected to sustained pressures of around 35 lb. per sq. in., a gradual slippage or “creep” of the solder may occur, causing the seam to unfold. Similarly, this may happen much more rapidly as a result of extremely high pressure. Experiments showed that a much stronger seam could be obtained if the solder penetrated into the third or innermost fold. Solder flows into the seam principally as a result of capillary action, but this is opposed by the force of escaping air and gases from the flux that is used to facilitate the soldering operation. These gases are trapped by the advancing solder, expanded by the heat and consequently exert a back pressure. Modifications to the soldering flux were not very effective in overcoming this difficulty, and the solution was found eventually in the use of side seam “vents” to allow the escape of gases. The side-seam currently used on beer cans is illustrated in Fig. 2, and this shows the vents or slits which are cut away on the body blanks prior to cylinder manufacture. This type of seam satisfies the unorthodox pressure requirements of beer canning.

The other problem presented by the high internal pressure exerted during pasteurization is that of end distortion. Normal open-top can ends for the general range of processed foods are stamped out with a contour comprising expansion rings which impart sufficient flexibility to withstand the moderate internal pressures developed during the heat sterilization process. In the case of pressure processed foods, super-imposed air pressure can be used during the cooling cycle to further alleviate the strain on the container. The effect of pressures on countered during beer pasteurization would certainly be a permanent distortion, which is termed “peaking,” of these ends. The most obvious way to tackle this difficulty would be to use heavy substance tinplate with improved temper characteristics. The latter has, in fact, been achieved to some extent by the use of re-phosphorized steel which confers added stiffness or temper but, unfortunately, the increase of tinplate substance is naturally accompanied by a progressive increase in the difficulty of opening the filled cans. Thus, although it is generally inevitable to have some increase in substance, this is kept to a minimum by adjusting other factors such as the contour or profile of the end. Also of importance are the double seaming characteristics of the end. The mechanical double seam of a beer can end to body is similar to that on a conventional open-top can as shown in Fig. 1 except that the countersink measurement which is indicated should, with beer cans, be as deep as possible, consistent with good double seam characteristics. The sharper the angle at the bottom of the countersink, known as the countersink radius, the less tendency will there be for inversion which would entail peaking.
One other consideration in the design of beer can end contour is that of occluded air. It is essential that the end should be so designed that the minimum of air is entrapped when the canner’s end is sealed on to the can.
Internal lacquering.—Metal exposure has been a problem with certain food products since the introduction of canning, and some highly efficient lacquer systems have been introduced to fulfil specific functions, such as sulphur resistance with meat and vegetable products and acid resistance with coloured fruit products. Until the advent of beer canning, perhaps the most complicated system was an acid-resistant fruit lacquer, consisting of two separate coats of lacquer applied to the flat sheets prior to can manufacture, plus a sprayed stripe of lacquer covering the side-seam area where the base lacquers are most liable to damage during manufacture. Lacquering greatly reduces the metal—mainly tin—content of all products, most fruits and vegetables showing only 10-30 p.p.m. of tin after storage at normal temperature for 12-18 months. Table II gives some figures for tin pick-up by canned prunes in both plain and lacquered cans and the remarkable reduction in tin content in the lacquered cans will be noted. Prunes have been chosen as representing an abnormally corrosive product.

However, when it was decided that beer should be packed, the canning technologist was faced with an entirely new situation. Until now, it had been accepted that some small degree of metal exposure was bound to occur owing to minute imperfections or pinholes in the lacquer coverage and unavoidable damage during fabrication. It was discovered rapidly that this situation could not be tolerated in beer cans, since minute traces of metal pick-up could result in off flavours and, more particularly in this country (because of the nature of the beers which enjoy popularity), in rapid loss of clarity with haze formation.9,13,5
Since a double lacquer coat with the additional stripe at the side-seam was found to be inadequate for beer, it was apparent that a drastic change in lacquering technique was going to be necessary so that a virtually impervious coating could be formed on the tinplate. In the old cone-top can this was achieved by flushing with a micro-crystalline wax. Among its disadvantages perhaps the most serious was the tendency of the wax to absorb flavour during storage. In the case of the flat-topped can, a much more successful answer to the problem has been found by spraying the inside of the can cylinders with a vinyl resin coating after manufacture. The normal objection to this procedure is of course one of economics, since, with the extremely high speeds reached in can manufacture, the necessity to spray and bake every can after fabrication represents a considerable economic disadvantage compared with roller coating much larger flat sheets before these have been made into cans. However, for beer cans it has been found to be essential.
Thus the complete lacquer system which has been developed consists of a base coat of oleoresinous beer-can lacquer applied to the flat sheet by roller coating, followed by an inside side stripe of a colourless vinyl after the fabrication of the cylinder and, finally, the further application of another colourless vinyl sprayed in a fan-shaped pattern, whilst the can rotates in such a manner as to direct the spray into the bend formed by the side-seam and to ensure that the whole of the body is covered. The ends receive two coats of lacquer, an oleoresinous base coat, followed by a colourless vinyl top coat. Thus, in all components the top coat is a vinyl and this is because these materials are the only ones which have, so far, been found which will meet all of the essential requirements in a beer can, namely to be colourless, non-toxic, tasteless (this is an outstanding virtue of vinyl resins) and chemically resistant, and to have good forming properties. (Even though the cylinder has been fabricated when the top vinyl coat is sprayed on, there are still the two double seaming operations to be carried out.) One may well ask why vinyl resins are not employed as base coats also, and there are two main reasons for this. Firstly, their adhesion to tinplate does not compare with that displayed by the oleoresinous lacquer used, and secondly, they are heat sensitive and would char badly during the side-seam soldering operation. There is another factor concerning this soldering operation: when the base coat is applied to the flat sheets, stencilled margins have to be left free of lacquer at those parts of the plate which are going to form the side-seam, otherwise of course, the soldering would not be possible. Since both of the subsequent spray lacquers are colourless materials, it is inevitable that the completed can will show a bright streak down the side-seam which is sometimes mistaken for an area which has not been lacquered at all whereas, in fact, like the rest of the can, two coats cover it. There are two reasons for the use of these transparent lacquers: in the case of the side stripe, the heat which still remains from the soldering operation when this lacquer is applied would disperse any colour pigment; and, with both spray lacquers, there is a possibility of some contamination of the outside of the can which would disfigure the decoration if coloured lacquers were used.
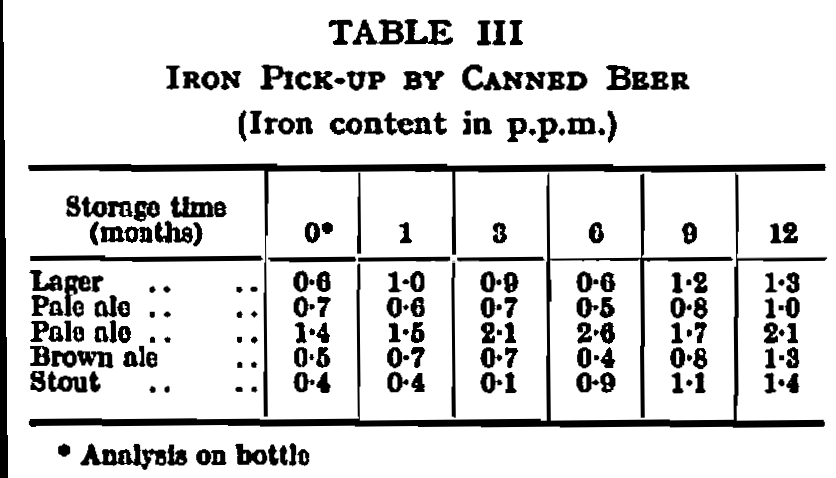
The degree of protection afforded by the lacquering system described above is illustrated by comparing metal pick-up figures with those quoted previously for a highly corrosive fruit pack in a fruit lacquered can. Analysis of beer from the container in current use has never shown a tin content higher than 0·04 p.p.m., and the average figure may be even lower. Some figures for iron pick-up during storage are shown in Table III from which it will be seen that, even after prolonged storage, contamination by more than about 1 p.p.m. is unusual.
Can inspection.—The inspection of cans before they leave the can-making factory may be roughly divided under three headings: (a) visual inspection; (b) inspection of dimensional characteristics; and (c) special tests. Possibly the most important factor in ensuring adequate visual inspection is the training of all factory personnel to understand the reasons for the care required with beer cans, to recognize faults however minute and to ensure that immediate action is taken. It is not sufficient to leave the control of quality to the specialized personnel, although these people must be available for reference. Visual inspection must take place at every stage during the can manufacturing procedure and, when the fabrication of can cylinders is completed up to the point of attaching the maker’s end, they first pass over an illuminated plate so that virtually every one may be visually examined for damage to internal lacquering or external decoration, accretions of lacquer and solder, and anything else which is not quite normal.
Most of the dimensional tests which are carried out are standard open-top can practice. Thus, the double seam quality must be checked at frequent intervals as must such measurements as body diameter, height, thickness of side-seam, etc. Furthermore, the correct lacquer application is checked by determining film weights.
There are two main special tests which are applied to beer cans. Both of these tests are destructive but they must be carried out on sample cans periodically during production. The first is a pressure test meant to simulate what will happen during beer pasteurization. This consists of filling cans with a dilate hydrochloric add solution, adding a pellet which contains a known amount of sodium bicarbonate, seaming immediately and then heating the cans to normal pasteurization temperature. The main purpose of this test is to detect can leakage but, at the same time, it gives an indication of whether peaking is likely to occur. If any fault is noticed, immediate action is required.
The other major procedure is designed to examine for metal exposure and is called electro-testing. The can is filled to within about £in. of the top with a specially formulated electrolyte solution, and then a circuit is completed from a contact with the bottom of the can and an electrode which is lowered into the solution in the can. A steady voltage is then applied and the current in micro-amp, is recorded. The interpretation of results obtained is still a matter of some controversy, but at least it is fair to say that the instrument gives a good indication of the standard of production which is taking place, and rapid action must be taken if the readings become abnormally high. A modification, known as the Canometer, operates on a similar principle but not only indicates the degree of metal exposure but also reveals the exact position of the exposed areas. Where breaks in the lacquer film occur, the tinplate is subjected to electrolytic attack and tiny bubbles of hydrogen are evolved which pin-point the faulty areas. However, this instrument is less reliable than the electrotester for routine measurements and is used as an adjunct to it.
Filling, fobbing and seaming.—All that has been said so far concerns the problems of beer can manufacture. Let us now consider the use of the container.
Before filling, the only brewery operations are uncasing of the cans, generally accompanied by mechanical unscrambling, followed by rinsing—usually with cold water. Then comes the filling operation. Most bottle-filling machines in use in this country can be adapted for cans, and the technical problems in the early days were primarily of a mechanical nature. New filling heads with modified dip tubes have to be fitted, and increased base pressure is necessary to achieve the required tight seal. This seal has probably constituted the major difficulty but, with the use of correct pressures and carefully contoured rubber pads, the trouble has been overcome.
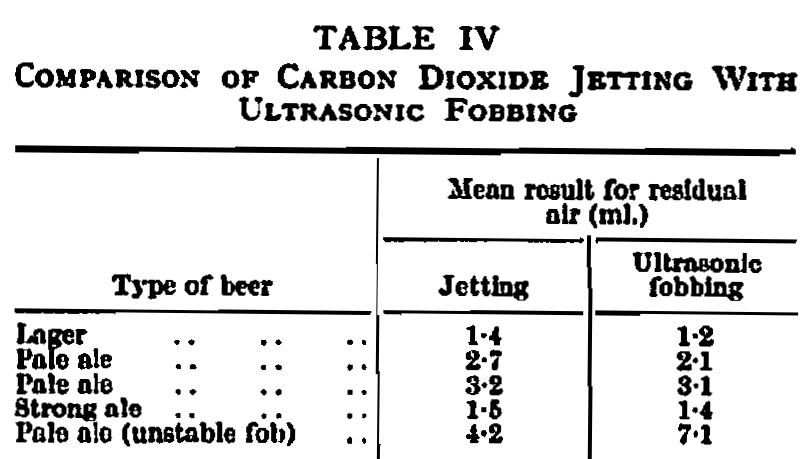
The next operation—fobbing—is undoubtedly that which, for a large majority of beers at any rate, determines whether the shelf life will be adequate. The effect of air on beer is well known and it is not necessary to reiterate, at this stage, the theories concerning oxidation hazes and flavor changes.6,8,10,12 However, in cans the exclusion of air is even more important since its presence will accelerate iron pick-up which, in turn, will hasten deterioration. In addition, high air will increase the pressure developed during pasteurizing which, as previously explained, is a critical factor already, without further aggravation. Carbonated beer when filled at a normal temperature of 36-40° F. is in a metastable state which normally prevents carbon dioxide gas from being released.14 This equilibrium can be upset by a plain shock or by providing nuclei for the release of carbon dioxide. “With bottles, shock is applied by the simple procedure of tapping, but this method would not be sufficient for open-top cans with their wide orifices, and the most usual method in this country, up to the present, has been some form of jetting to introduce carbon dioxide gas under the surface of the beer and provide the necessary impetus for the release of more gas, thus producing a foam on the surface. Recently the application of ultrasonic energy in industrial processes has provided a possible method of producing a shock-induced fob with beer cans. The ultrasonic device consists essentially of a generator connected to a magneto-striction transducer through a remote control box. A shoe is mounted on the end of the transducer which contacts the cans and, in that manner, transmits the energy. From observation, the main advantages claimed over carbon dioxide jetting are that the fob produced is more stable and consists of finer bubbles which, it is felt, may trap less residual air. Table IV gives air residuals determined on jetted cans compared with cans that have been ultrasonically fobbed and from these results, which were obtained under controlled laboratory conditions, it will be seen that there is a fair comparison at this stage. No marked advantage is yet shown by ultrasonic fobbing, but it should be remembered that this is still in an experimental stage and some improvement may be expected. An undoubted advantage, of course, is that no carbon dioxide gas is consumed.
The third and last method of expelling air from the headspace is known as under cover gassing. Although widely used in America, this technique, which is to inject carbon dioxide gas into the headspace of the can immediately prior to closing, has found little favour in this country so far, mainly because of the mechanical difficulties involved in fitting it to existing machinery. However, active investigation is proceeding.
It is not proposed to describe double seaming in detail, since this is standard canning practice, but it should be emphasized that it is a very precise procedure and one on which the whole success of any canning venture depends. The standards which are laid down must be adhered to strictly and regular examination, both visually and by destructive seam checking, must be carried out.”
Apart from contour modifications which have been described earlier, beer can ends differ slightly from standard open-top can ends in the location of the rubber composition, known as lining compound, which is placed in the end curl to assist in forming the hermetic seal. This is illustrated in Fig. 3 from which it will be seen that, in the beer can end, the compound reaches farther up the shoulder. This feature helps to reduce the danger of metal exposure during the double seaming operation.

Between double seaming and pasteurizing the cans are inverted, normally through a twist track, so that the canner’s end is at the bottom and leakage during pasteurizing will, therefore, result in loss of weight and rejection of the cans at the check weigher. This ensures that leaking cans do not pass unnoticed.
Pasteurizing and cooling.—Canned beer must be pasteurized because sterilization of the cans before filling, as is done with bottles, is neither practicable at present nor desirable in view of the equipment required. Also the likelihood of re-infection may be greater with the wider orifice. Furthermore, cans may well be kept longer than would be expected with bottles, because the public tends to expect an indefinite microbiological storage life with all canned products.
For these reasons cans have always been pasteurized, but it is fairly obvious—from consideration of the heat conductivity and thickness of the materials used—that there will be a difference in the rate of heat penetration into cans compared with bottles when subjected to similar heating conditions. This comparison is illustrated graphically in Fig. 4, which traces the temperature at the slowest heating point of cans and bottles when immersed in a water bath at 140° F., a very common pasteurizing temperature.
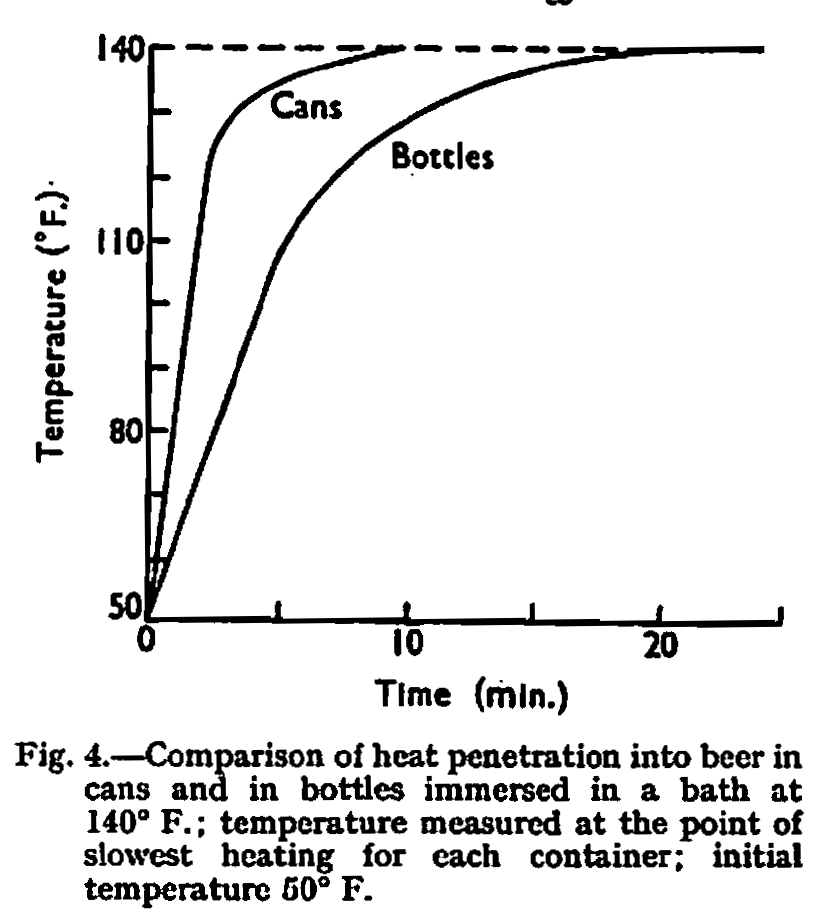
It will be seen from this graph that, whereas with bottles it takes some 24min. for the slowest heating point to reach bath temperature, with cans this state is achieved after only 10 min.
The unit of reference when considering the sterilization of beer is the pasteurization unit or P.U., defined as the lethal effect obtained by holding beer for 1 min. at 140° F., and there are references in the rather limited literature on this subject to the effect that a value of approximately 6 P.U. is the absolute minimum desirable for safety with beer.3,5,7 However, it is believed that brewers work to a considerably higher value in this country—somewhere between 20 and 30 P.U.—and no doubt the exact pasteurizing cycles chosen are well founded on experience. Let us assume that a pasteurization cycle giving 25 P.U. is required. Now from Fig. 4 and a knowledge of the thermal death characteristics of the organisms involved, it is possible to calculate that after 20 min. the bottles have received 5·6 P.U. and the cans 14·5 P.U. If we assume that the internal temperature of both is at 140° F. after this time (this is not quite true in the case of the particular bottles we have described, but near enough for this illustration) it can be seen that, to achieve our desired 26 P.U., the bottles will require approximately another 20 min. in the bath, whereas cans need only about 11 min. more. Thus, the total time for bottles has been approximately 40 min. as compared with about 30 min. for cans.
Cooling should be carried out with cans efficiently to about 85° F. Under-cooling may cause the beer to acquire a “pasteurized” flavour and will cause the end seams to be strained for a prolonged period; this may promote leakage. Over-cooling may result in external rusting of the double seams of the can, and for the same reason, it is advisable that surplus water should be removed from the countersink of the top end, by passing the cans under a compressed air jet as they leave the pasteurizer.
Testing brews for canning.—Every beer which eventually goes out on to the market in cans supplied by the author’s Company, has been canned in a trial run in the laboratory first of all. This is known as acceptance testing. Although this is done on laboratory scale, great efforts are made to ensure that the procedure is as near as possible to that which would exist commercially, and small-scale commercial equipment is used. After canning, examinations are carried out for residual air contents, carbonation, metal pick-up during storage, and flavour and clarity changes during storage. Normally a shelf life of three months at ambient temperature is required before the brew is considered acceptable for commercial canning, and almost invariably it has been found that shelf life terminates by haze development rather than by any other feature, e.g., flavour. This is believed to be a characteristic of British beers. Up to the present, some 300 brews have been acceptance tested. Some 10-15% have failed the test at first attempt but, frequently, co-operation with the brewer in improving beer stability and canning technique has eventually achieved an acceptable result.
For several years a system of routine testing of samples from canners’ production was carried out in a somewhat similar manner to the acceptance testing in order to ensure that the necessary high standard had been maintained. Now that the canning of beer has become more fully established, this routine testing has been abandoned, but it is replaced by a limited examination of samples from brewers who are new to canning; this is now called establishment testing.
Further research.—Despite the undeniable fact that the birth of soft-drink canning has tended to shift the limelight from beer, the impetus which has been given to research work must benefit beer canners. Apart from the acceptance and establishment tests described above, some of the projects which are still under investigation include: the improvement of existing methods of air removal and development of new ones; better methods of can inspection and the interpretation of results therefrom; Pasteurization—the possibility of using temperature differentials during pasteurizing or bulk pasteurization and some examination of the microbiological characteristics of British beers; and, finally, the ever-constant search to improve the can which is being supplied for beer.
Acknowledgement.—The author wishes to thank the Directors of The Metal Box Company, Ltd., for permission to publish this paper.
References
1. Anderson, J. H., Bottler & Packer. Aug., 1959, 47.
2. Anon., Brewers’ Guardian, May, 1959, 59.
3. Baselt, F. C, Daybarsh, C. A., & Del Vecchio. H. W., Proc. A.M. Amer. Soc. Brew. Chem.,
1954. 141.
4. Brenner, M. W., Proc. Eur. Brew. Conv., Copenhagen, 1967, 349.
5. Dayharsh, C. A., & Del Vecchio, H. W., Proc. A.M. Amer. Soc. Brew. Chem., 1952. 48.
6. de Clerck, J., A Textbook of Brewing. London: Chapman & Hall, 1957, Vol. I, p. 545.
7. Del Vecchio, H. W., Dayharsh, C. A., & Baselt, F. C, Proc. A.M. Amer. Soc. Brew. Chem.,
1951, 45.
8. Gray, P. P.. & Stone, I., Wallerstein Lab. Commun., 1939, (v) 5.
9. Lebreton, P. A.. Gagnaire, B. J., & Michel, G. F., Wallerstein Lab. Commun., 1957, 20, 233.
10. Lloyd Hind, H., Brewing Science &- Practice.London: Chapman & Halt, 1948, p. 992.
11. Metal Box Co. Ltd., Tech. Commun., No. 15, July. 1955, 17 pp.
12. Precce, I. A., The Biochemistry of Brewing. Edinburgh: Oliver & Boyd, 1954, p. 336.
13. Silbereisen, K., & Wittmann. G., Proc. Eur.Brew, Conv., Copenhagen, 1957, 263.
14. Stewart, E. D., Amer. Brewer, Aug., 1958, 27.