BREWING INDUSTRY RESEARCH FOUNDATION
FACTORS AFFECTING BEER FOAM
I. CARBON DIOXIDE EVOLUTION
by S. Jackson, M.A., B.Sc, D.Phil. (Brewing Industry Research Foundation, Nutfield, Surrey)
Received 8th April, 1953
The rate of evolution of carbon dioxide from beer has been investigated by withdrawing samples at suitable intervals, running them into excess 0.05 M borax buffer solution, and analysing the resulting carbonate solutions manometrically by regenerating the carbon dioxide in the Warburg apparatus. Results show that complex formation is not concerned in the evolution of carbon dioxide, and that the solubility of carbon dioxide in beer is close to its solubility in water. As with a supersaturated solution of carbon dioxide in water, the rate of release of the gas from beer is dependent on the presence of nuclei for bubble formation. More nuclei are available when beer is served into a dry than into a wet glass, and in this way a more effective renewal of head may sometimes be achieved.
Introduction
One of the important factors by which a beer is judged is the quality of its head. Few beers consistently give a compact, stable head and although numerous investigations of head formation and stability have been made,2,3,10,14,15,16,19 no generally reliable method of ensuring good head retention has emerged.
When beer is poured, a head is formed by evolution of carbon dioxide and entrainment of air during the considerable agitation which takes place. The durability of the head depends partly on the stability of the original foam and partly on the production of new foam by the continued liberation of carbon dioxide after pouring. Foam stability and carbon dioxide evolution will be considered separately, and the present paper is concerned only with the latter factor.
If carbon dioxide release after pouring could be controlled so as to give adequate renewal of foam without excessive “flattening” of the taste, it would be an important step towards solving the problem of head retention. The concentration of carbon dioxide in beer is normally about 0.4% by weight.4,11,21,23,24 A solution of carbon dioxide in water at this concentration is highly super-saturated with regard to air, and when exposed to the air it loses carbon dioxide until the concentration falls to 0.02%, at which the solution is in equilibrium with atmospheric carbon dioxide. The gas escapes by diffusion into the air or into bubbles. Escape is slow unless effervescence is vigorous, when the presence of numerous bubbles greatly increases the area of surface across which carbon dioxide can diffuse. The rate of evolution is therefore mainly dependent on the rate of bubbling, and the fact that the solution is supersaturated is not itself a sufficient condition for the formation of bubbles. For effervescence to occur, it is necessary that gas bubbles should be already present to provide a gas/liquid interface and so act as nuclei. Ordinarily, minute bubbles will be present, trapped in crevices of the containing vessel, when the cycle of events may be visualized as follows. Carbon dioxide diffuses into a minute bubble, causing it to grow until its buoyancy exceeds the force (surface tension) holding it on the surface of the vessel; the major portion of the bubble then breaks away and floats to the surface. A small part of the bubble, similar in size to the original nuclear bubble, may be left behind in the crevice, so that another bubble can grow, and this may be repeated indefinitely. The stream of bubbles is therefore only liberated because of the presence of the original nucleus, and the rate of escape of carbon dioxide from supersaturated solution in water depends on the number of nuclei available.7
Hopkins & Krause,13 Lloyd Hind,12 Helm & Richardt,11 Mohr,17 and others consider that the rate of evolution of carbon dioxide from beer is limited only by this factor; but, on the other hand, Emslander & Freundlich,8 Schilfarth,20 Petit18 and Siegfried22 have expressed the opinion that the rate-determining step is not a purely physical one, but that the release of the gas is retarded because carbon dioxide in beer is chemically combined. If this were correct and if combination could be controlled, a means of regulating carbon dioxide evolution might be available. The aim of this investigation was accordingly to establish whether the rate of carbon dioxide evolution from beer is, or is not, limited by chemical combination.
Experimental
Method of analysis for carbon dioxide.—The investigation necessitated the analysis of samples for their carbon dioxide content, and this was carried out with the Warburg micromanometric apparatus.6 The principle of the method was to stabilize the .carbon dioxide as carbonate and then to regenerate it inside the Warburg apparatus by acidification, determining the carbon dioxide concentration by the change in manometric pressure. In order to obtain a stable carbonate solution, the pH must be brought to a high level, the limit depending on the carbonate concentration. The routine procedure was to pipette out a 4 ml. sample of the test liquid into 90 ml. of 0.05 M borax buffer and then make up the total volume to 100 ml. with buffer solution. The resulting solution of carbonate (not more than 0.004 M at pH 9.2) was completely stable. Bubbling during sampling was minimized, though not completely eliminated, by dipping the pipette into the liquid without applying suction. Portions (3 ml.) of the solution were analysed in triplicate in 20 ml. Warburg units, at 25° C, the carbon dioxide being regenerated by mixing with 0.2 ml. of 2 N sulphuric acid from the side arm. The manometric liquid was Brodie’s fluid; the flasks were calibrated with mercury, and Dixon’s nomogram6 was used to calculate the flask constants. It was necessary to subtract from the results a blank determined on the buffer solution alone. The water used in making the buffer solution was twice distilled, the second time in pyrex glass, and was completely freed from carbonate by passage through columns of Amberlite IR-4B(0H) and IR-100(H) ion-exchange resins. Borax buffer solution (0.05 m) gave a blank of 40-50 μl. Borax was chosen as the reagent in preference to sodium hydroxide because of the much higher blanks given by the latter. The accuracy of the method was tested by experiments in which known amounts of sodium carbonate were dissolved in 0.05 M borax buffer solution. The results are collected in Table I; the limit of accuracy is 0.01% of carbon dioxide (in the original sample). With the apparatus available, five samples, each in triplicate, could be analysed simultaneously, the group of determinations taking 2 hr. Tests showed that degassed beer, when added to known sodium carbonate solutions in borax buffer, did not affect the evolution of carbon dioxide in the course of analyses. The applicability of the method to beer was confirmed by checking it against the chemical method of the American Society of Brewing Chemists.1
Rate of spontaneous carbon dioxide evolution from beer.—It was mentioned above that the rate of bubbling in a supersaturated solution of carbon dioxide in water is dependent on the number of bubble nuclei available. It is impossible to prepare two vessels so that they will each provide the same number of nuclei, and it is therefore necessary to eliminate these completely in comparing the behavior of different solutions. If the vessels are perfectly wetted by the solutions, no nuclei are present, and for vessels of similar shape the area of surface across which carbon dioxide can diffuse, i.e., the area of the top surface of the liquid, is the same in each case, so that the behaviour of different solutions may legitimately be compared under these conditions. In comparing beer with carbonated water, glasses were prepared by the following successive treatments, in order to promote complete wetting:
(a) soaking overnight in chromic acid mixture;
(b) soaking overnight in a solution of Santomerse wetting agent;(c) soaking overnight in water; and
(d) maintaining full of water until immediately before use for experiments.
The glasses were thoroughly rinsed with water after each of these treatments.

A British pale ale (top fermentation beer, chilled and carbonated) and a Danish Pilsner (bottom fermentation beer, naturally conditioned) were compared with carbonated water made from solid carbon dioxide and distilled water. Two experiments were carried out, one at room temperature (17° C), the other at 1° C. The rates of carbon dioxide evolution were determined by taking samples at suitable time intervals and analysing them as described above. The results are plotted in Figs. 1 and 2. It appears that the rate of diffusion of carbon dioxide across the liquid-air interface is similar for beer and carbonated water at the higher concentrations, whilst at lower concentrations the water loses carbon dioxide more slowly than do the beers. The closely similar behavior of the pale ale and Pilsner is very striking, but the result is nevertheless not finally decisive because it is impossible to prove that absolutely no bubbling occurred in the tests, in spite of the elaborate precautions.
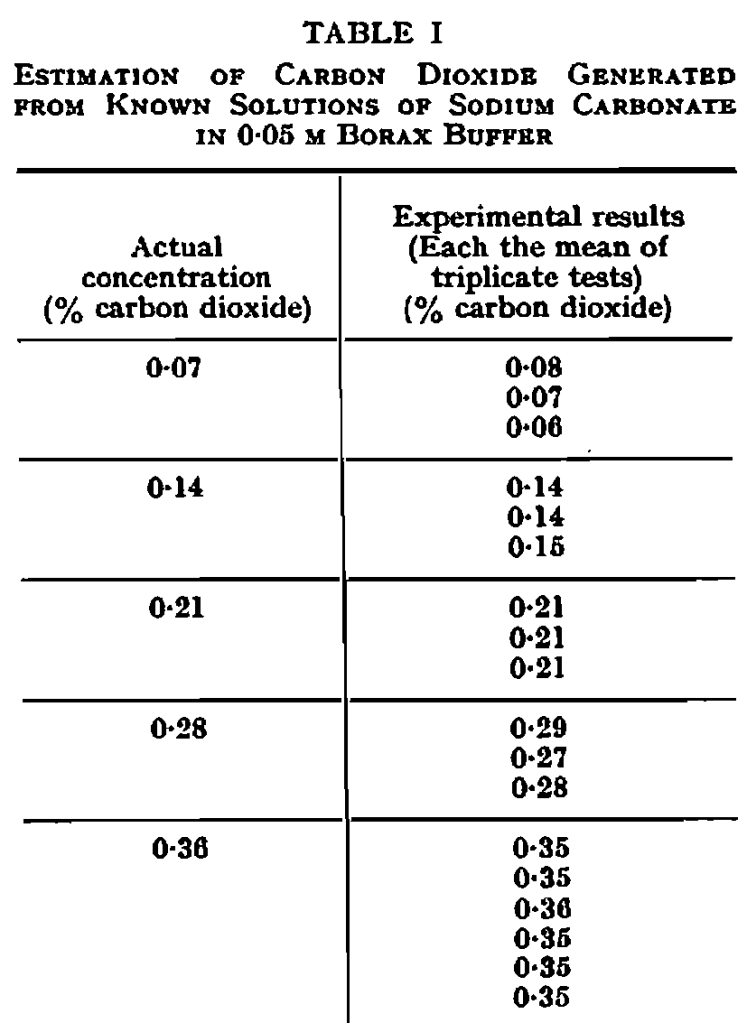
Effect of bubbling carbon dioxide through beer.—By bubbling carbon dioxide gas through beer, it is possible to make a critical test for the nature of the rate-determining factor in the evolution of carbon dioxide. Escape of the gas should obviously be accelerated if bubble formation is the limiting step, but not if the rate of escape is determined by the rate of decomposition of a carbon dioxide complex.
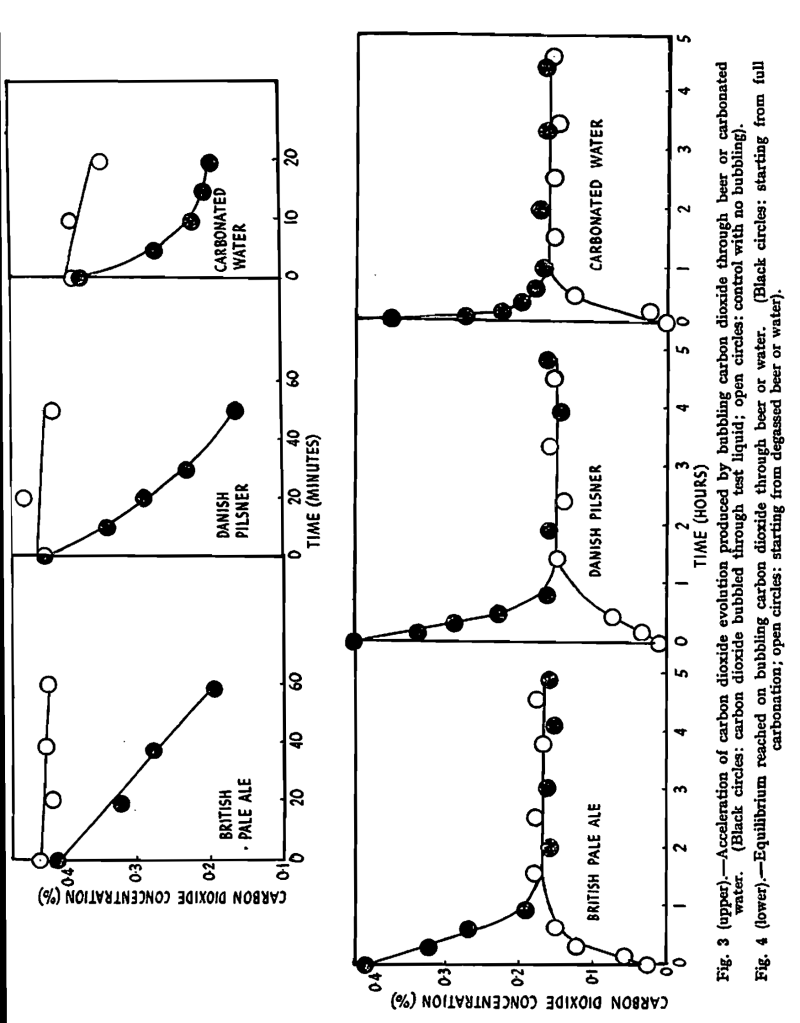
A pint bottle of pale ale was equilibrated to a temperature of 20° C. in a constant temperature water-bath before opening. Half was poured into a Burton ale glass, prepared as before, for use as a control. The other half was poured into a measuring cylinder, diameter 2 in., also specially prepared. Both vessels were kept at 20° C. in the water-bath. Carbon dioxide from a cylinder was bubbled through a capillary tube into the beer in the measuring cylinder, the top being loosely stoppered, with a clearance of about 5 in. above the beer, to leave room for the foam; the carbon dioxide was previously passed through a spiral of copper tube and a water bubbler, immersed in the water-bath, to bring the temperature to 20° C. and to saturate the gas with water vapour. The rates of evolution of carbon dioxide were measured as before, the results being plotted in Fig. 3, along with the corresponding results for Pilsner and carbonated water. In each case, the rate of evolution was markedly accelerated by bubbling carbon dioxide through the liquid. Clearly, the factor which sets the limit on the rate of escape of the gas is the extent to which bubble formation can take place.
These experiments yielded further information, as seen in Fig 4, in which a smaller time scale is used, this Fig. incorporating the results of Fig. 3 together with further measurements. It can be seen that evolution of carbon dioxide ceases when the concentration has fallen to 0.16%; at this point the system has reached equilibrium, as was proved in the following way. When each control glass had lost all its carbon dioxide, after standing for 24 hr., the liquid was transferred to the measuring cylinder, and carbon dioxide was bubbled through. The concentration of carbon dioxide in solution rose rapidly until it reached 0.16%, after which there was no further increase. The results of these experiments are also plotted in Fig. 4. The gas in the bubbles is in equilibrium with a carbon dioxide concentration of 0.16% in solution. The pressure of carbon dioxide in the bubbles is equal to atmospheric pressure plus the pressure exerted by the liquid. The latter is comparatively small, so that the equilibrium concentration is effectively equal to the solubility of carbon dioxide at 20° C, at a pressure of one atmosphere. Although the method of experiment does not permit a precise determination, it is clear that the solubility of carbon dioxide in pale ale or Pilsner is close to the solubility in water. Findlay & Shen,9 using a more precise method, found that the solubility of carbon dioxide in beer (which had been boiled) was in fact slightly less than its solubility in water; such a slight fall in solubility is to be expected because of the presence of other non-reacting solutes. These experiments provide conclusive proof that carbon dioxide in beer, whether introduced by chilling and carbonating or by natural conditioning, is in a simple state of solution. The solution is supersaturated when exposed to the air, and the rate of evolution of carbon dioxide in practice is dependent only on the availability of nuclei for bubble formation.
Discussion
Practical considerations in the serving of beer—effect of drying the glass.—The presence of bubble nuclei in a glass of beer depends, as has been described, on the glass being imperfectly wetted. The wet-ability can be reduced by coating the glass surface with hydrophobic substances, such as silica or paraffin wax, but this is obviously not a practical possibility in serving beer. An important practical consideration is, however, that the glass should be dried, because it is then less completely wetted than a glass still damp from rinsing, and the evolution of carbon dioxide after pouring is correspondingly more brisk. In an experiment carried out to demonstrate this, two half-pint glasses, one wet, the other dry, were filled from the same bottle of pale ale. The concentration of carbon dioxide in both glasses immediately after pouring was 0.30%. After 30 min. there was no detectable loss of carbon dioxide from the wet glass, but the concentration in the dried glass had fallen to 0.24%. This was sufficient to maintain a slight head, whereas there was no head on the beer in the wet glass.
Summary
1. Carbon dioxide is not chemically combined in beer.
2. Beer freshly exposed to the air is supersaturated with carbon dioxide.
3. The rate at which carbon dioxide is lost depends on the extent of bubble formation; this depends on imperfect wetting of the glass surface and it is therefore important that glasses in which beer is to be served should be dried.
Acknowledgement.—The author expresses his thanks to Sir Ian Heilbron, D.S.O., F.R.S., Director of the Brewing Industry Research Foundation, for permission to publish this paper, and to Monsanto Chemicals, Ltd., for a gift of Sanlomerse concentrate.
References
1. American Society of Brewing Chemists, Methods of Analysis, 1949, 27.
2. Baker, J. L., & Hulton, H. F. E., this Journal, 1923, 747.
3. Blom. J., this Journal, 1937, 251.
4. Brenner, M. W., McCully. R. E., & Laufer, S., Amer. Brewer, Feb., 1951, 39.
5. Dixon, M., Biochem. J., 1945, 39, 427.
6. Dixon, AI., Manometric Methods, 3rd Ed. Cambridge: University Press, 1951.
7. Edwards, L. J., private communication.
8. Emslander, F., & Freundlich, H., Z. phys. Client.. 1904, 49, 317.
9. Findlay, A., & Shen, B., /. chem. Soc, 1911, 99, 1317.
10. Helm, E., this Journal. 1951, 33.
11. Helm, E., & Richardt, O. C, this Journal, 1936, 191.
12. Hind, H. L., J. Inc. Brewers’ Guild, 1947, 33, 252.
13. Hopkins, R. H., & Krause, B., Biochemistry Applied to Malting and Brewing (2nd Imp.).
London: George Allen & Unwin, Ltd., 1947, pp. 302-305.
14. Krauss, G., & Harreis, F., Woch. Brau., 1940, 57, 1.
15. Laufer, S., & Ziliotto, H., Amer. Brewer, Aug., 1939, 25; Sept., 1939, 33.
16. Mischke, W. Kolloid-Z., 1940, 90, 77.
17. Mohr, O., Woch. Brau., 1903, 20. 153.
18. Petit, P., Brass, el Malt., 1921-22,11, 289, 305.
19. Sandegren, E., Proc. Eur. Brew. Conv., Scheveningen, 1947. 35.
20. Schilfarth, H., Brauerei, Wissensch. Beit., 1951, 4, 96.
21. Siegfried, H., Schweiz. Brau.-Runds., 1945, 56, 1.
22. Seigfried, H., Schweiz. Brau.-Runds., 1950, 61, 19.
23. Wallerstein. J. S., Wallerstein Lab. Commun., 1937, No. 1, 31.
24. Warth, A. H., Amer. Brewer, Dec, 1944, 26.