North of England Institute of Brewing
MEETING HELD ON THURSDAY, 5th MAY, 1898, AT THE GROSVENOR HOTEL, MANCHESTER.
Full Beers of Low Alcoholic Strength
By A. E. Berry
During the sittings of the Departmental Committee appointed by Parliament to inquire into the materials now used in brewing, several of tho witnesses, among them being Mr. Frank Wilson and Dr. G. Harris Morris, gave evidence as to the marked change that had occurred of late years with regard to the public demand for light alcoholic ales as compared with the strong heavy ales of 12 years ago.
I think we all agree that such is the case, and that more of the consumer’s attention is given to the quality of the beer than ever before, so much so, that beer brewed as in “the olden times” would find a very limited sale in the present day.
So far, however, the difficulty has been in producing a beer of low alcoholic strength and of desired roundness and fulness. These have been partly surmounted by the use of malt adjuncts; but there is still a wide field for improvement, as no English beer made by the high fermentation system can compare in fulness or mellowness with those made on the Continent by the thick mash and low fermentation process. Those brewed on the Continent, however, as you know, must be fermented at a low temperature, and, even at
the time of consumption, this must be maintained or else they lose their character and are liable to turn sour in a few hours.
In many parts of the world, and in fact in some parts of our own country, the ordinary English ale is still found to be too heavy, and what seems to be wanted is a light ale which will leave a fulness on the palate after drinking.
Tho United States Consul in Ceylon writes to say that attempts to supply light ales, both from English and Australian breweries, failed, the liquor being undrinkable. He adds, however, that German beer would be easily displaced if an English ale of low alcoholic strength—and not higher in price—could be supplied.
In the same way the Consul at the Gold Coast says:—”German is preferred to the English as lighter;” and from Sierra Leone the Consul reports that “German lager beer being lighter, but not superior, is preferred to the English, and is gradually rising over the British manufacture;” so that it is quite evident that the “old English ale” will soon find a halting place around the equator.
The Hon. G. H. Reid, Colonial Premier of New South Wales, said in a speech: “Well, I have sampled some of the new light beer, and, having a palate which is entirely unadulterated, am, I think, in a position to give a good opinion. I do not hesitate to predict for the beer a very successful career. I do not think it is quite strong enough for the old hands who used to live on colonial beer as it used to be, but by the largely increasing number of Australians who do not want so much a stimulant as an agreeable harmless beverage, I think this product will be highly esteemed.”
This new light beer refers, I think, to Iager beer, and although this certainly has great fulness when made by the thick mash process, yet there is the peculiarity from which all lager beer is inseparable caused by the type of yeast used in low fermentation.
To obtain ales of low alcoholic strength and good gravity combined with fulness and mellowness with the old system of brewing from malt alone, is almost impossible. In speaking of ales of good gravity, I mean to infer that the body, which is the cause of a
palate fulness, naturally follows when high gravity results after brewing from articles that are composed of carbohydrates.
Besides these properties, a good ale should possess pure taste, brilliancy, good and pure aroma, and a foaming head. Certain substances participate, partly directly and partly indirectly, in bringing about these desired results, namely, the albuminoids, the albuminoses (which have an extraordinary influence in formation of foam), and lastly, the malto-dextrins. It is desirable that the beer when ready to be consumed should be well charged with carbonic acid, and this is attained when the wort contains the right amount of maltodextrins, since these are the cause of a steady and slow fermentation in the cask. The production of fulness and foam-keeping quality arises from the same source.
Worts that are strongly concentrated in brewing, of proper composition, and normal degree of attenuation are at present higher in gravity than those which are brewed light and attenuate to a low degree.
These facts regulate the percentage of alcohol, and in almost all cases the amount of alcohol is in proportion to the extract. I am of the opinion that this should not necessarily be so, and that the fulness of a beer should not be in proportion to the alcohol. You might say that a certain amount of alcohol is absolutely necessary in order to impart a character to the beer, and to a certain degree this is correct, but not to the extent it is generally carried.
I think a beer with a full taste and moderate degree of attenuation would always find more favour with the public at large than one with a full taste and high degree of attenuation.
In order to attain this end it in necessary to increase the unfermentable carbohydrates (classed as “dextrin”) in the wort at will, and to maintain a proper balance between them and the fermentable carbohydrates (classed as “maltose”). These results in practice are rarely attained by brewing entirely from malt alone owing to a variety of circumstances, such as change in composition of malt, form of plant, &c.
The temperature in the mash-tun is scarcely ever a uniform one, and the thermometer does not always give the accurate heat on the malt. For instance, it is found that the temperature gradually decreases towards the centre, and if the thermometer is placed at the side and the heat regulated by it so that a good yield of unfermentable sugars (malto-dextrins) would occur, then we find around the centre of the mash-tun (which contains most of the wort), being a few degrees cooler, is favourable to the formation of fermentable sugars, and does not convert the starch to the right extent.
Now, even the character of a beer is already determined by the malt from which it is to be brewed, there is not the least doubt that certain very important properties of the beer are greatly influenced by tho methods used in the brewhouse.
A patent has been granted in Germany for a process for improving the palate fulness of beer. In order to avoid the repeated operations of boiling and stirring the mash, as usually practised with the object of increasing the dextrin contents of the wort, the object of this invention is to destroy the activity of the diastase of a large portion of the malt, before crushing, by heating it in a rotating drum up to 100° C. and further heating until a smell of freshly baked bread is generated.
This treatment will destroy the diastase and render the elastin of the skin of the malt soluble without browning or caramelising the malt itself. The water driven off in this process is replaced either by the introduction of three per cent, of water into the apparatus or by blowing in steam, the drum being heated and rotated as before until the malt appears dry enough to crush.
This prepared malt is mixed with a proportion of untreated malt varying according to the class of beer required, and after being mashed in cold or warm water and heated gradually up to 75° C. the wort, as soon as “breaking” occurs, is drawn off from the grains without any further boiling of the mash, and then treated in the usual way.
This process no doubt presents many interesting features, but as I have not yet been able to confirm the accuracy of tho statement I cannot speak with confidence as to its merits. One thing, however, is certain, and that is, that by heating at 100° C. the greater part of the diastase will be destroyed, although at the same time I see objections that might be put forward to treating malt in this way.
The ratio of unfermentable carbohydrates to fermentable carbohydrates in worts can, to a great extent, be governed by the temperatures used in mashing. If the character, the properties, and the chemical and physiological behaviour of the malt enzymes were known it would be, comparatively speaking, an easy matter to so regulate the working that good, round, and full ales of low alcoholic strength could be easily obtained, or in other words it would be possible to obtain a wort and, finally, ale of desired composition.
As a wort of a certain composition corresponds to an ale of a certain composition and character, then, with an accurate knowledge of the chemical behaviour of the enzymes, the foundation ot ales of known character could be laid down.
It is found, however, that tho knowledge of the chemical properties of malt enzymes leaves something to be desired—a fact which prevents us finding the proper way to reach the desired object. There are various ways of mashing by which brewers endeavour to reach this goal, each no doubt thinking that he has overcome the difficulty.
This, the most important branch of brewing, is involved in mystery, and during the mashing process a brewer is in complete ignorance as to the precise action of the malt enzymes, because their properties as yet have hardly received the attention they should have.
The process of forming an extract of malt is, in a sense, a purely mechanical operation, or, in other words, a dissolving of those substances which are soluble in water, but this is followed by chemical changes which are brought about by the enzymes in the malt, and herein lie the difficulties already mentioned.
The enzyme known as diastase seems to have received the most, attention from chemists, its function being to saccharify the starch. There are organic laboratories in which preparations of diastase have been obtained, but although this enzyme plays such an important part in brewing, how few really know what it is. The enzyme which dissolves and decomposes the albuminoids is peptase, the knowledge of which is only in its infancy, as it is not even recognised as a substance of itself, and its existence is only inferred from its action. Very important work has already been done on these enzymes in England, and very great credit is due to the researches of our noted brewers’ chemists for the way they have brought this most important point before our notice. When information on the subject is more complete, we shall most likely be able to produce ales of desired roundness and fulness, and at the same time of low alcoholic strength; until then, I do not think we can. We are therefore constrained to resort to other methods, of which I will speak later.
The most important points in the present system are: the temperature of the mash, the time during which the enzymes are allowed to act, and the quality of the malt.
Although it is difficult to state with any degree of accuracy the composition of the wort formed, it may be broadly stated to consist of—
(1) Readily fermentable sugars
(2) Unfermentable sugars, which do not, or only partly, ferment, but which give roundness and fulness to the finished beer.
The formation of the second class in as large a percentage as possible is, I think, every brewer’s desire, but concerning which almost every one has his own ideas.
In a brewery where the raw materials change from year to year, and brewers from place to place, this is a very difficult task. When the chemistry of the enzymes is mastered, science will help us to determine the way the mash should be carried out. I do not mean to offer any advice on the right way to mash, this can only be elucidated by repeated trials, and by the brewer using all his intelligence and practical experience.
I do not agree with the methods just described for attaining high final attenuations, and think it is the best plan to secure this desired result by other means, such as the addition of a body to the wort in the copper, with the properties we desire to increase, and, in conjunction with colleagues in London, I have been working with the object of finding a sugar that will answer this calling.
When a brewer tries to attain this end by regulating the temperature and time of mashing, he very often causes cloudiness and fretfulness, owing to change in the weather, &c, leaving large amounts of nitrogenous substances in the beer that should have been removed by fermentation.
By the addition of a carbohydrate in the form of a malto-dextrin to the wort, low attenuation, and a good cleansing of the wort by the use of a strong yeast, is secured, whereas by limiting the attenuation haze is generally caused, and an excessive amount of fermentable carbohydrates is retained. Every now and then brewers have to contend with what we call “haze” in the beers, and no amount of fining will clear it. This “haze” is very difficult to get rid of, and usually plunges the brewer into the depths of despair. I attribute this “haze” to small particles of gluten, which become suspended in the beer, and have not sufficient gravity to allow them to settle.
I have examined several of the so-called unfermentable sugars, and have found that they generally ferment almost to the extent of ordinary glucose and invert-sugar. Now what is an unfermentable sugar? You continually hear of malto-dextrins being spoken of as
unfermentable, yet we find that they ferment slowly after the primary fermentation is over, and so, in the true sense of the word, they are not unfermentable. Then, again, we have malto-dextrins of still higher molecular weight, which are termed dextrinous malto- dextrins, and ferment after a few months only to a slight extent, and not until all primary fermentation is over.
If, as I before mentioned, we were fully acquainted with, and thoroughly understood, the enzymes of malts, their malto-dextrins no doubt might be so regulated to yield beers required for both rapid consumption and export, or stock purposes, without the addition of unfermentable sugars.
Such, however, cannot be done with our present state of knowledge, and as most brewers use sugar now, why not use part of this in an unfermentable form, say in the form of dextrinous maltodextrin. You will, no doubt, ask how are you going to obtain part of the sugar in this way, and be sure that it will retain its chief properties. This is certainly a very difficult thing to do, and in all my experiments I have always aimed at bringing about this end by a redistribution of the elements, and not by changing the sugar into a new body altogether.
Starting from glucose, I have worked in such a way as to form (C6H12O6-H2O)2, and find that you can obtain almost endless substances of complicated composition, and varying in their chemical properties. They are undoubtedly a very interesting group, and should, I think, be classed with the dextrins, but I have not yet had time to ascertain their probable composition, having contented myself with their behaviour with yeast when mixed with wort in various proportions, and a rough outline of their general properties. The higher you get the less unfermentable they are, and, as you would naturally expect, their reducing power decreases, and they become less soluble in alcoholic solutions.
In making an examination of them, tho results so obtained are rather misleading, and do not give any indication as to their probable source of origin. In some respects they answer the tests of glucose, reduce Fehling’s solution, and give an optical rotatory power, varying from [a]; 3·86 = + 100 to 120. The angle, however, varies, as does also the extent to which they reduce Fehling’s solution, and I may say that these two tests do not afford the slightest indication of the extent to which the substances are unfermentable.
It is a curious coincidence that although the curing and roasting of barley, &c, has received great attention, and various schemes have been invented for the purpose of effecting changes in the nature of the albuminoids and carbohydrates, that a similar treatment has never been extended to carbohydrates in the form of maltose and invert-sugar.
Now that sugar plays such an important part in brewing, I think strict attention ought to be given to the same to try and find a means of so treating the sugar, that it will also contain valuable proportions of fermentable and unfermentable carbohydrates, and make it answer some of the deficiencies that we now find with changes of malt, &c. I have made a large number of experiments on sugar in this way, and have obtained very encouraging and satisfactory results.
If heating is carried far enough, a product is obtained which is wholly unfermentable, but unsuitable for producing the fulness required in beers, because, in the first place, the flavour of the product so obtained is unfavourable, owing to traces of assamar, and, furthermore, is insolubile in alcoholic solutions. This insolubility, however, can be overcome by judicious working as in the manufacture of caramel, a product with which I think all are more or less acquainted, and is generally found unfermentablc. The use of
ordinary caramel for producing low alcoholic beers is altogether out of the question, because the colour produced by the quantity necessary for the purpose would be so great, that the tint of the beer would be completely spoiled. If an attempt be made to reduce the colour so as to make it applicable, the substance is at once rendered fermentable, and I find that the extent to which a caramel is ferment able is in proportion to the heat used in its manufacture.
On heating cane-sugar to 100° C. very little change takes place; if water vapour is present, however, it melts and is slowly converted into glucose and levulose as the temperature rises. Then if maintained at 170° C. for a few hours, it gives off water and is rendered partly unfermentable, probably forming traces of glucosan. If the heat is increased to 220° C, it is converted into caramel, which has a formula something like C126H168O80.
The results obtained from glucose are the same, except that you find it stated in the text-books, that an unfermentable sugar called glucosan is formed from glucose by heating it to 170° C, but 1 have not been able to render glucose unfermentable by heating it to 170° C. On heating to 220° C, I obtained a product which, when made into solutions of various strengths and treated with yeast in the ordinary way failed to lose one degree in gravity in 14 days. This product, however, is not applicable forgiving sufficient fulness and roundness to low alcoholic beers because, when added at the rate of 1 ½ lbs. per barrel, the depth of colour in the beer is about doubled.
There are, however, ways of heating sugars so as to render them practically speaking unfermentable without caramelizing them, so that when they are made up into solutions of equal gravity to the beer to which they are to be added, the colour of the solution will not be darker than the average pale ale.
There is a class of dextrins formed from glucose by means of acids, -which probably cause the condensation of several molecules under separation of water. These products have been examined by Wohl, and are called by him “reversion products”. They are contained in the commercial glucose, and in most of the dextrins, and are neither attacked by diastase, nor are they fermentable.
I have also tried the action of various acids on glucosc, and obtained products which were partly unfermentable, but which I think you will agree are unsuitable for brewing.
For instance, after prolonged boiling with hydrochloric acid, glucose becomes unfermentable, forming probably an isomer of cane sugar having a disagreeable flavour, soluble in alcohol and reducing Fehling’s solution.
Most of the mineral acids ace in the same way, and in a few instances a small quantity of a saccharide is formed. Working with cane-sugar or I might say invert, for this is what is first formed on treatment with acids, very similar results were obtained, generally charring the sugar with formation of CO3, and an organic acid, and giving a product with a very unpleasant flavour.
By the action of alkalis slightly better results are obtained, but the heating has to be carried so far before the sugar is rendered unfermcntable, that the use of the finished product in brewing is then entirely out of the question.
None of these means would produce an article that anyone would like to see used in brewing, and the rendering of a sugar unfermentable to be of advantage must be accomplished almost rather by a physical change than by a chemical one.
This is the point I have always tried to work on, and have tried to imitate as far as possible the composition of the dextrinous maltodextrins found in wort.
One of the first experiments I made, was boiling glucose with about twelve times its weight of alcohol and recovering the alcohol as boiled off. This gave favourable results, which I hope to turn to good advantage later.
By heating a sugar to certain stages in some cases it is rendered unfermentable, the formation of glucosan as recorded in the textbooks being an instance of this kind.
If I were to go into all the methods that I have tried for rendering starch after saccharification, glucose, maltose, and other sugars unfermentable I should probably weary you before I had gone very far into the matter.
There is, however, one way of treating the sugars, that I think will interest you, and that is by heating strongly in a stream of oxygen, or in other words aerating a hot solution of the sugar. The product so obtained can hardly be classed as a dextrin, although it is to a
certain extent unfermentable, but not sufficient to make it answer the purposes for which it would be required. For instance, if you start with a solution of the sugar so prepared and ferment with yeast you find fermentation commences at once, losing 1 degree gravity after 24 hours, and when added to wort the whole of the attenuation of the product added seems to take place during the primary fermentation, so that this product is out of the question for ensuring fulness and foaming properly.
The samples that I have with me tonight are practically unfermentable, and I think if you were asked what form of a sugar would you like to add to your beers in order to improve them, that the description you would give would be found to almost exactly correspond with those possessed by these samples.
I have often heard brewers say that they are quite pleased and satisfied with their beer; and the beer comes up to the required standard in almost every respect, but it might perhaps be improved as regards fulness and capacity for producing “head” (foam).
A very important point is, that the increased fulness imparted to the beer seems to supply a medium for retaining the carbonic acid naturally generated in the beer, thus ensuring a more permanent “head.” It is an undoubted fact that beers brewed with these unfermentable sugars retain their “head” much longer than is usually the case.
From a large number of experiments made with these unfermentable sugars, I am led to the conclusion that during primary fermentation, they resist the action of the yeast. After primary fermentation is over, and the beer is run into the cask a very slow fermentation sets in by means of the primary yeast cells that find their way into the trade cask. The loss in gravity caused by the unfermentable sugar I calculate to be about 1/100 part of a degree per day when added at the rate of 3 lb. to the barrel of 36 gallons.
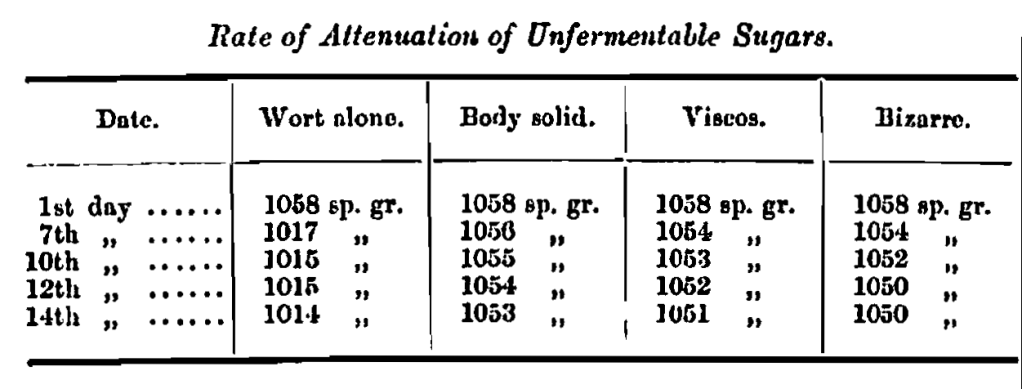
Temperature on first day 60° F., gradually increased up to 70° towards end of reaction.
These solutions were made by dissolving the sugars in hard water such as might be used in brewing to the above gravities, after which the colour of the solution very much resembled that of a pale ale.
I also find after the sugar has been so treated, that the extract obtained from it is quite 20 per cent, more than what was obtained from sugar before treatment, and as the whole of this goes through tho primary fermentation without loss of gravity, the consequent effect upon the finished beer must be very important. The extract of these sugars per excise quarter of 256 lbs. is about 100 lbs., and when added to the copper at the rate of 5 lbs. per barrel gives a final gravity of 55 per cent., which is equal to 2 lbs. This gravity remains for a very long time, the attenuation of dextrinous maltodextrins in the sugar being very slow and generally lasting a few months. For stock, or export beers, this most important feature will at once commend itself, and it may be taken to furnish a means of producing a beer that will continue in good and steady condition for several months.
In brewing ales of low alcoholic strength for quick consumption it is necessary to add a solution of glucose or invert-sugar at racking time, so that you really get a continuation of the vat fermentation.
These unfermentable sugars have a pronounced flavour – which is perhaps, not very agreeable now, but when used in a dilute form for brewing, the character and flavour are distinctly favourable. ln order to show this on a practical scale, I have a few samples of a beer which has been brewed from one of these sugars with the addition of hops and invert-sugar to give head and a slow fermentation. The amount of absolute alcohol is very low, only being 0•79 per cent., which makes it a non-alcoholic drink according to excise regulations.
I have made a large number of brewings, on a small scale, with various proportions of these sugars added to the wort, and obtained very good results. In each case the sugar was first dissolved in water and made up to the same gravity as the wort to which it was to be added, although on a large scale it is usual and far the better plan to add the sugar in the solid form to the copper before hopping.
The quantity to be added must be regulated by the class of beer required, and as will be seen from the results below, almost any desired strength can be obtained with gradually decreasing percentage of alcohol.

This, as will be seen, presents a ready means of increasing the final gravity of the beer 11 degrees, which is equal to about 4 lbs. per barrel, and at the same time of reducing the alcohol over 1 per cent.
There is another way of obtaining these low alcoholic beers of good gravity, namely, by treatment of the wort, so as to render part of the sugar in such a state that it resists the action of yeast.
Various means for accomplishing this end might be employed, but I have always thought that such were undesirable methods of attaining this desideratum.
I find there is a patent in Germany for obtaining non-alcoholic beers from malt, the principle of the patent being as follows :—
The boiled and clarified wort is treated with diastase at 57•5° C, so as to produce the maximum amount of maltose and very little dextrin. The wort is then boiled with hops, and exposed to the atmosphere while hot in a very fine state, such as the form of a spray, so as to bring about a separation of the albumin and such substances which might afterwards produce unpleasant flavour and cloudiness.
A further separation of undesirable constituents and the production of a pleasant flavour is brought about by again subjecting the beer whilst hot to the same treatment as it went through in the early part of the process, but by substituting carbonic acid gas for air.
The beer is then cooled rapidly to 0° C. by means of freezing coils, which cause the deposition of other detrimental substances. The beer is fined and saturated at a temperature of 0° C. with carbonic acid gas, after which, according to the patentee, the non-alcoholic beer so prepared is free from germs, and has great stability and digestibility.
This is certainly a very interesting process, and curious to say part of the above is almost on the same lines that 1 worked some months ago. However, as the patent is now out and the principles of it have appeared in the Journal of the Society of Chemical Industry, I will refrain from going into any of the details of my process, and furthermore, because I arrived at the conclusion that non-alcoholic beers could be more easily and more cheaply prepared by other means.
I will, however, mention a few of the methods I employed, such as boiling the wort with organic acids and aerating by means of oxygen gas under pressure whilst boiling in the copper.
First with, regard to the latter, I find that aeration either by means of oxygen alone or atmospheric air has no appreciable effect on the after fermentation unless organic acids are present in the wort to an excessive amount. The quantity and the nature of the acids formed during mashing has a marked effect upon after fermentation, as you will readily see from the following results:—
Small batches of wort were boiled with .05 per cent, of various organic acids, and, after cooling, fermented with yeast in the ordinary way, when it was found that varying results were obtained. In the case of acetic, lactic, and tartaric acids, the difference was not very great and might be ignored, but when using oxalic the fermentation was greatly retarded and a deposit was produced.
No one would even dream of using oxalic acid to check fermentation, nor do I think that the German patent of heating the wort to 57•5° C., and then subjecting it to air and carbonic acid gas will ever be appreciated in this country. The only sate way of obtaining a low alcoholic beer, of required fulness and mellowness (until our knowledge of the enzymes of malt is more complete), seems to be by brewing in the usual way, and replacing a portion of the invert-sugar or glucose now used by an unfermentable sugar.
In the case of brewers who do not use ordinary brewing sugars, the addition of 3 to 5 lbs. of unfermentable sugar to the quarter of malt will give equally good results. In endeavouring to obtain equally good results from malt alone many dangers are encountered and very often defective solution.
Some very interesting work has been done by Dr. Charles Graham showing how the time, the temperature, and the mass affect the formation of maltose and dextrin during the mashing process.
I think his results prove that it is impossible to obtain from malt alone the necessary amount of dextrins for brewing low alcoholic beers and at the same time effect complete solution of the malt.
A certain way of brewing might answer well enough, with one class of malt, but with another the same method might give exactly opposite results. For instance, the quantity of water used during mashing has a marked effect, and in evidence of same I quote one of Dr. Graham’s tables as given in a lecture before the Society of Chemical Industry.

By limiting the time of infusion and also the quantity of water employed is the common way of obtaining a dextrinous wort from malt, but a glance at tho above table will at once show how untrustworthy such methods may be with changes of malt.
Lastly, I should like to say a word against defective solution of malt which often results on changing malts and working to get high dextrinous worts from same.
As you all know this can readily be detected by using the iodine test. If its turns blue, some of the starch has escaped conversion and remains unconverted in the wort, its presence causing proneness to decomposition, and a cloudiness which no fining will remove.
It is also a cause of sarcina infection, and it has been found (L. Aubrey) that the quantity of sarcina organisms found in a beer was encouraged by starch and found to be in proportion to the starch.
The unreliability of the iodine test for badly dissolved worts has caused it to be well looked into, and on the Continent they first precipitate the dextrin insoluble in alcohol with four to five times its value of alcohol and shaking the mixture well, when any starch that may be present is lightly deposited on the sides of the vessel. It is then dissolved in water after separating from the alcoholic solution and treated with iodine in the usual way.
Discussion
The Chairman said that after the interesting paper they had listened to, some of those present might like to ask some questions, if so, Mr. Berry would be glad to answer them. It was a long and complicated paper, and when they had it in their hands in printed form, they would be better able to deal with it. The next thing they would have to secure in connection with the papers read before the members would be that they should be printed, so as to be better able to follow the author. Up to the present time, however, they had not been able to see their way to do it, but he hoped the thing would be accomplished during the next session, or that some arrangement would be made whereby they could better follow the readers of papers, and be more qualified to hold discussions upon them.
In answer to a question as to whether unfermentable sugars had any marked effect upon primary fermentation or upon the yeast, Mr. Berry said so far as he had been able to gather, he could not find they had. He knew where unfermentable sugars were used in large quantities, in conjunction with invert and cane-sugars, and fermentation went on just the same. In other cases, when brewing with 3 lbs. and 5 lbs. per barrel, he had noticed no effect. As an instance, he had some samples of beer brewed with one of these sugars; they contained .079 per cent, of alcohol, and when fermented in the usual way. No malt was used, it being replaced by invert sugars and an unfermentable sugar (malto-dextrin), so that they would readily see how nearly it corresponded to malt ales. He had also a small cask of beer in the room, part of 18 barrels brewed with the use of 1 ½ cwts. of glucose, and ½ cwt. unfermentable sugar; also a similar brew of 18 barrels in which 2 cwts. of glucose was used, but no unfermentable sugar. By substituting ½ cwt. of glucose by ½ cwt. of unfermentable sugar the effect was very great, for even with this slight change the beer brewed with unfermentable sugar gave a finished beer of higher gravity and containing about ½ per cent, of alcohol less than the one brewed without unfermentable sugars.
In answer to the Chairman, Mr. Berry said that the gravity of the beer in the cask before fermentation was 1.056, and after fermentation 1.019 when brewed with unfermentable sugar, and in tho other case 1.056 before and 1.016 after fermentation. The beers were only finished a few hours before he left London, and he had not been able to obtain all the information from them he would have liked, but the one brewed with unfermentable sugar contained 3·60 per cent, of alcohol, and the one brewed without contained 4 per cent. In order to show the difference between the beer of our forefathers and that which found more favour at the present time, he instanced a sample of beer brewed by Messrs. Worthington and Robinson, of Burton-on-Trent, in 1862. Its final gravity, in 1862, was 1.120, and it now contained over 20 per cent, of proof spirit.
Mr. Hyde, in moving a vote of thanks to the author for his paper, said if by replacing malt with sugar they could arrive at something better, they would be ready to try the article. He had had no experience with unfermentable sugars. He got more fulness and body by putting more malt into the mash-tun. In his early days among brewers the gravity used to ran down below zero. He was a beer drinker in those day, 17 years ago, and those beers did not taste thin. He had seen beers brewed with 20 per cent, of dextrins, and they did taste thin. A good deal depended on the condition of beers when drunk. There was a good deal yet to be discovered with regard to them.
Mr. Bennett seconded the motion, and said he had tried to make dextrins with malt only; whether they could produce ales of a satisfactory character here by the methods proposed remained to be seen.
The vote of thanks was adopted.