MEETING HELD AT THE MIDLAND HOTEL, MANCHESTER, On November 7th, 1907.
Mr. A. L. Lees in the Chair
The following paper was read and discussed:—
The Influence of Mashing Temperature upon the Composition of Wort
by C. G. Matthews and R. A. Auty.
At the outset of this communication we wish to remark that we are very doubtful as to whether the title adequately describes the nature and scope of the experiments to be detailed, for it was found very difficult to put the matter into brief terms. The main object in view has been to gain information about the state in which starch exists in malt, and to find out something further about the manner in which malt starch breaks down under the influence of the active enzymes of the malt.
In 1885 one of us, in conjunction with Mr. Walk’s Evershed, published an article entitled “A Method of determining the Extract of Malt, and some Facts connected with the Effect of Time and Temperature on Experimental Mashings” (the Brewers’ Guardian, January 13th, 1885). The publication in a Brewing Journal (before the days of the Institute of Brewing) limited, one would suppose, the publicity the matter might possibly have attained, and taking this into consideration, together with the fact that in the light of further experiment a value attaches to the results which was hardly perceived at the time, we have ventured to reproduce the more important features of the work.
The method of determining the extract may be dealt with and dismissed in a few words; it is the method, of which not a few modifications are extant, of mashing a definite weight of malt with an approximate settled volume of water at the temperature desired, sustaining the required temperature in a water bath for a given period, cooling to 60o F., and making up to a definite weight on a balance (the vessel, of course, being tared). A portion is filtered off from the well-mixed mash, the specific gravity is determined, and from this the volume of wort present, and so the extract. It is a good method, but, perhaps, gives somewhat high results as compared with sparging-out methods. One object at the time the experiments were conducted was to determine the degree of conversion of the starch of finely-ground malt at different temperatures. In each case the mash was kept constant at the stated temperature for two hours, and the extract then determined in the manner just described. Initial heats were obtained as near the required temperature as was practicable. The results are shown in Table I.
In Series I (Table I) it will be seen that nearly 9/10 of the total extract was obtained at 130° F., a temperature far below the gelatinization point of barley starch.
Series II (Table I) shows a more extended range of experiments with another malt. It may be noted that there is a period of marked activity between 100° and 110° F., and another at 120° F.; 8/10 of the extract has been gained at 130° F., and more than 9/10 at a temperature 5° higher, whilst at 140° F. the greater part of the available extract has been obtained.
We are able here to introduce a new series (Series III, Table II) of results obtained within the last few months.
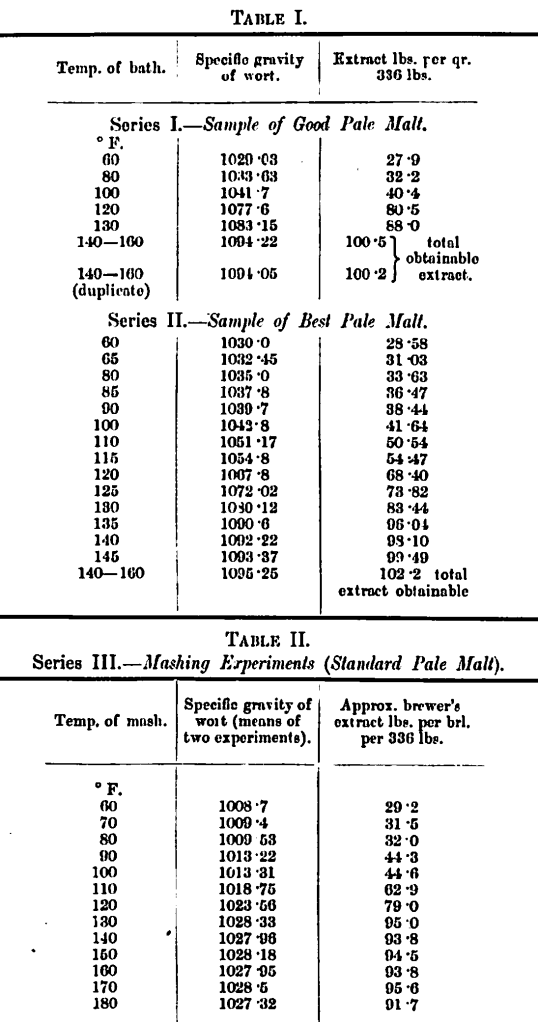
The figures show some activity of transformation at 110° F., and from this point a very rapid accession of activity up to 130° F., at which point practically the maximum of extract has been obtained.
The results of all three series can, we think, be more graphically displayed as curves, and we have put thorn together in one chart (see Plate 1). The higher figures for specific gravity (right hand) refer to Series I and II, whilst the lower ones (left hand) refer to Series III. The curves are, however, comparable, the wort in Series I and II being practically 3–4 times as strong as Series III, the gravities proportioning in this way throughout.
The chief points to be noted are:—
(a) Indications of a reaction between 80° F. and 100° F. (more particularly in Series III); (b) the extremely rapid transformation taking place between 100° F. and 130″ F. (all three malts show this plainly).
Inferences.—(1) That up to a point of temperature the starch granules mainly resist the action of the enzymes; (2) but a slow action is going on which is the preface to an extremely energetic one; (3) that the temperature of the energetic period is far below the gelatinisation point of barley starch.
The following possibilities suggest themselves :—
That the matter of the starch granule is not homogeneous (this may be taken as pretty certainly the case); that a definite preliminary attack on the starch granule has to be made by some enzyme; or that some carbohydrate substance, other than starch, is the first to undergo change.
A few experiments were tried with pure potato starch (loc. cit.) by introducing small quantities into fresh cold water malt extract of known strength, and keeping for two hours in the water bath at a constant temperature, then determining the gain of extract, if any, from the conversion of the starch.
The conclusion drawn from the above experiments is that potato starch begins only to be energetically attacked by diastase as the temperature nears the point of gelatinisation. It is obvious that there must be some remarkable difference between starch in this form and that in which it is present in malt.
The method of determining extracts was next applied in a series of experiments, the object of which was to determine the rate at which the extractable matters are brought into solution from malt at temperatures known to be favourable, viz., from 142° F. upwards. The action was stopped when necessary by adding a small quantity of a solution of salicylic acid in borax, then the mash was thoroughly mixed, weighed and adjusted, and filtered. The malt was, as before, finely ground.
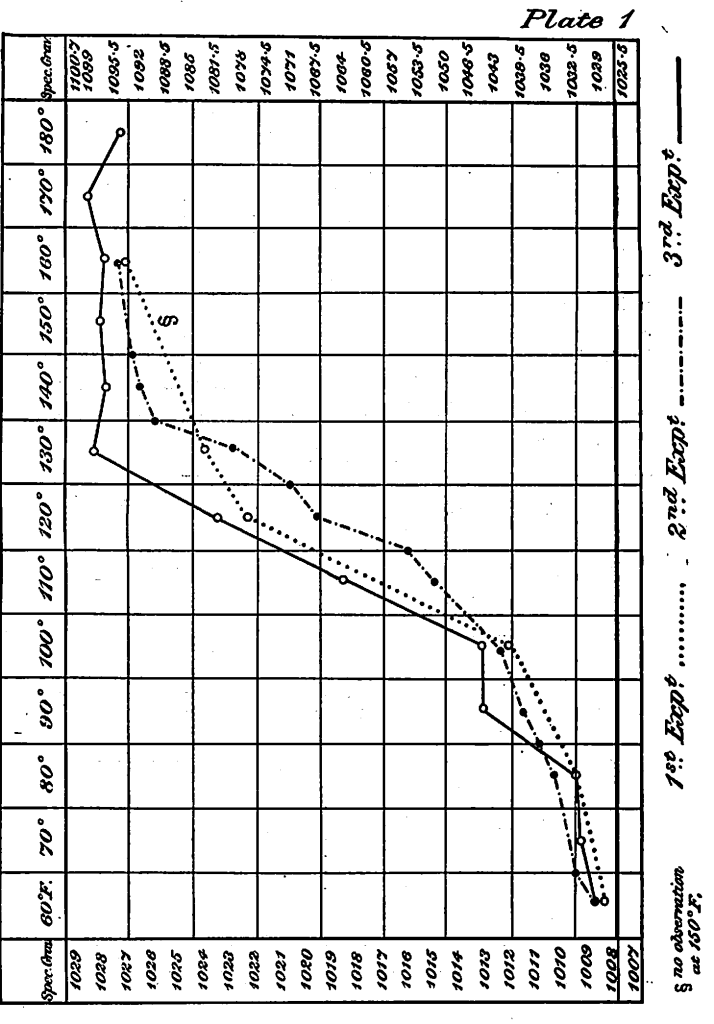

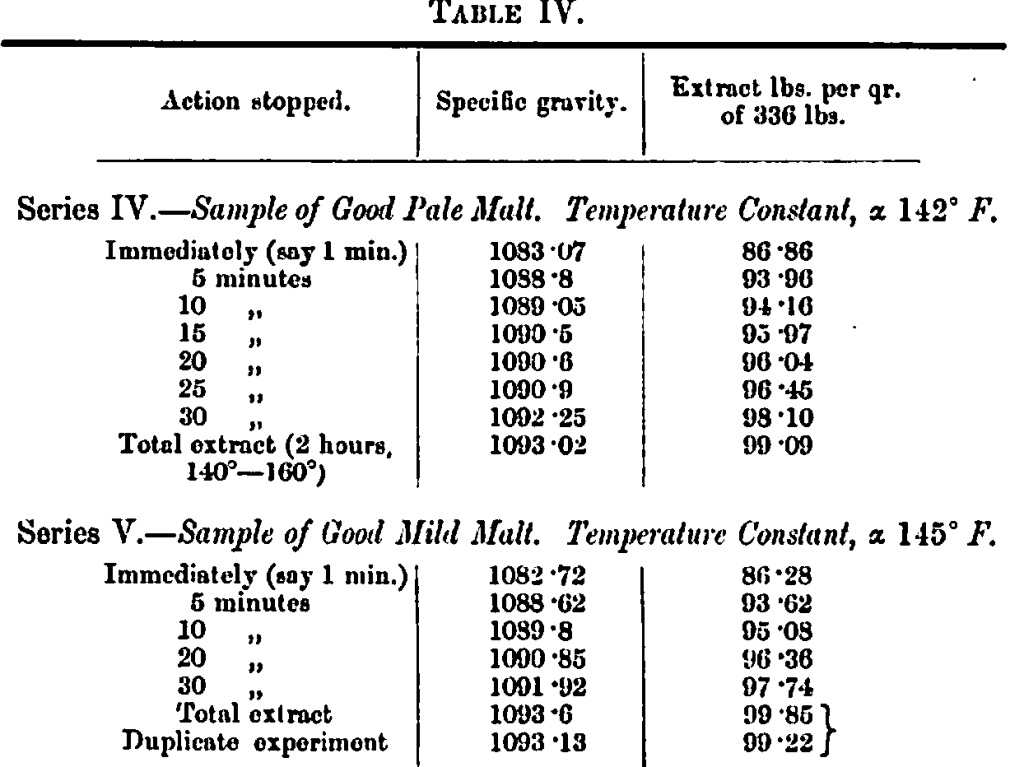
The most noticeable feature in these experiments is the large amount of extract obtained in a very limited time of action, thus showing the rapid conversion of the malt starch at temperatures which have been shown to be favourable to transformation. The greater part of the extract is obtained at once and 19/20ths in 10 minutes at the temperature employed. Of course, fine grinding greatly favours this rapid action, but again the experiments indicate how easily malt starch is converted at temperatures 30° F. below the ascertained gelatinisation point of barley starch. The question that next presented itself and which we have endeavoured to elucidate is, taking the whole range of temperature between 60° and 180° F. into account, what is happening in a mash conducted at different points in the range 1 We have tried to answer this question by undertaking a series of experiments the results of which we now proceed to detail.
A stock malt—a very good quality of pale—from English barley well and evenly grown, was bottled in 1900, in perfectly dry bottles, the corks being covered with melted paraffin wax. We have made three complete analyses of this sample—all of which are in fair agreement—conducted on the following lines:—50 grams of the malt, ground to a fineness fairly comparable with 25 Seek, mashed with 180 c.c. of distilled water for an initial temperature of 150° F. This temperature was sustained for two hours; the mash was then filtered and sparged at 167° F. up to a volume of 500 c.c.
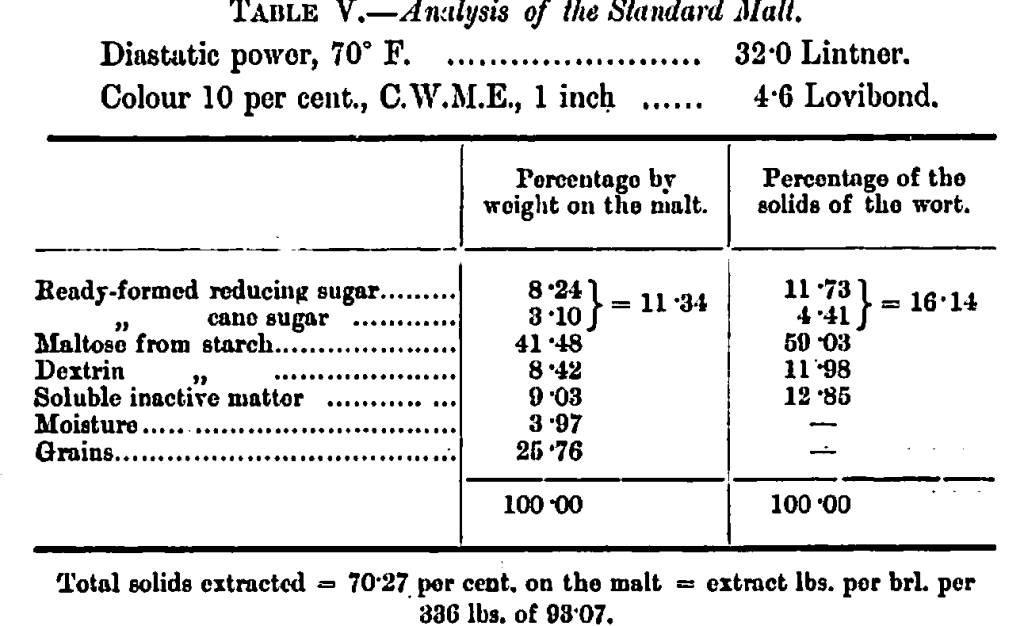
These figures represent a malt of normal character and in very good condition considering the time of storage. It was used throughout the experiments now to be described.
It appeared convenient to take malt and water on a 10-per-cent. basis, and to get the needful quantity of wort, 90 grams of malt finely ground were mashed with 900 c.c. of distilled water at a temperature calculated to give the required initial or mixing heat as nearly as possible. The temperature was maintained at the required point in a water bath with fairly frequent stirring of the mash. At the end of two hours, the whole mash was poured into large funnels with plaited filter papers and as much clear filtrate was obtained as possible, which was usually from 650 to 750 c.c. The clear, sweet wort was well shaken and divided into two equal parts, A and B. A was taken untreated and the specific gravity, the polarimetric reading in a 200-mm. tube in Ventzke divisions, and the cupric reducing power as CuO per 100 c.c. determined. B was raised to boiling and actually boiled for a few seconds, then cooled down to 60° F., and the same constants determined as in the unboiled wort. To each portion was now added 1 gram per 100 c.c. (about 3½ lb. per brl. as pressed yeast). of pressed clean active brewery yeast, and fermentation was allowed to proceed in a warm place (about 65° F.) till the fermentation was judged to be complete. In the case of the worts of higher specific gravity the time occupied was about 60 to 65 hours. The original gravity was then determined and the polarimetric reading in a 200-mm. tube, and the cupric reducing power (usually taken in the extract gravity portion).
The following constants were employed for calculation. Inland Revenue tables for the original gravity determination, divisor 3·95 for solids per 100 c.c. derived from specific gravity. The cupric reducing power of maltose was taken as 1·37 gram CuO = 1 gram maltose. The Soleil-Ventzke-Scheibler polarimeter was used.
The rotation constants of the under-mentioned carbohydrates in a concentration of 1 gram per 100 c.c. taken in the Soleil-Ventzke-Scheibler polarimeter in a 200-mm. tube and recorded in Ventzke divisions were as follows:—
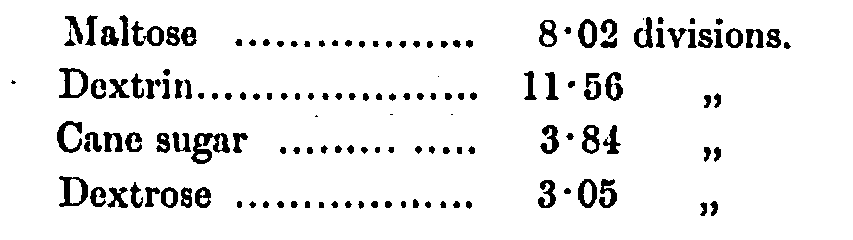
The results obtained are given in Table VI.
The values for the cold water malt-extract at 60° F. are from the average of the three analyses of the standard malt already given, with two additional experiments, so as to obtain the most exact correction obtainable to be used throughout the range of the experiments whenever necessary. The figures stand opposite A, the un-boiled cold water malt extract. The composition of the boiled portion is not very materially different, but rests only on one experiment.
It may be noted that there is a very satisfactory agreement between what we would call the O’Sullivan-Heron sugar values and the sugars arrived at by fermentation, and we believe, as we have always done, that the O’Sullivan-Heron values applied to cold water extract are substantially correct. It might be deemed a more satisfactory method of estimating the sugars removed by fermentation from the amount of alcohol produced, but it has been found in the course of these experiments, as in many other cases, that the fluctuations in the quantity of alcohol produced—due to known and unknown causes—are so considerable, that no reliance could be placed on figures so obtained. The loss during fermentation, or matter unaccounted for, varied from 0·29° of specific gravity to as much as 3·76° and averaged 1·98° on the 26 experiments. The sugars, if calculated from the alcohol, would vary proportionately. It might be supposed that the losses shown are matter removed by fermentation, but we are convinced that this is not the case; that on the basis of the Inland Revenue tables a deduction of say 1° specific gravity suffices as a correction, and wo have used this as a correction throughout, though it is possibly a somewhat insufficient value where much sugar was fermented, as doubtless the matter removed is greater the greater the amount of sugar fermented. The amount of yeast employed was, however, practically such as to be non-multiplying.
Let us now direct our attention to the results set forth in Table VI.
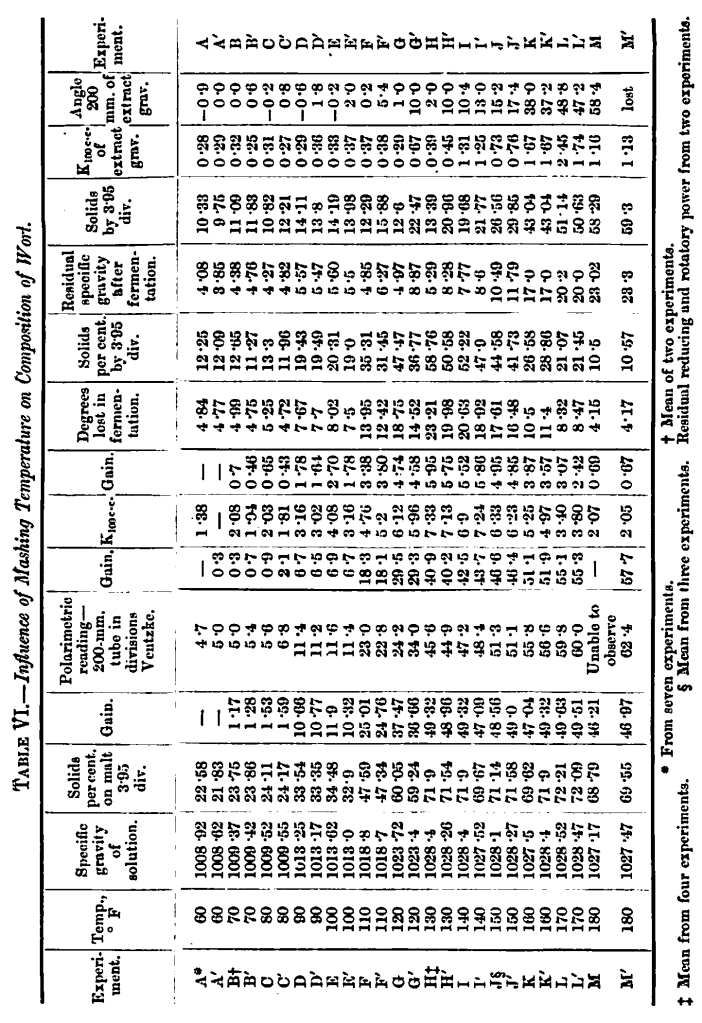

The mean specific gravities obtained from each pair of experiments have been already charted in Plate 1. There is no very marked gain in extracted solids up to 80° F. At 90°, 110°, 120°, and 130′ F., very marked increases are shown; very nearly the maximum extract, as before observed, being obtained at 130°.F. Up to 80° F. the polarimetric reading corresponds to the O’Sullivan-Heron values (4·3 divisor for mixed sugars). At 90° F. there is .a considerable gain in optical activity, whilst at 100° F. the composition appears to have remained stationary.
At the next temperature points, 110°, 120°, and 130° F., there is a marked step forward. There is no particular change (except in the boiled portion = C) in the rotation till 90° F., where there is a great advance and a similarity of composition between the unboiled and boiled portions.
As regards fermentable sugars, there is a great advance at 110° F. and at 120° and 130° F. The highest, K100 c.c.. is touched at 130° F. (Experiment H), and here we find the maximum amount of sugar fermented, nearly 6/7ths of the solids of the wort being fermentable matter. The first marked residual rotation is found at 110° F. (F’), whilst the first marked residual reducing power is at 120° F. (G’). The dextrin produced at 90° F. (D), shown by the largely increased rotation, is apparently fermentable in the presence of the residual diastase at this point and up to 130° F. (unboiled worts), although it shows plainly in the boiled worts F’, G’, and H’ above 130° F. It seems fair to conclude that at a temperature above 130° F. the diastase was so modified in its powers that it either gave rise to undegradable dextrin or was not able to degrade the combined dextrin it had produced.
In Experiment M (180° F.) a condition is reached in which there is not only no increase on the original ready formed sugars, but a slight decrease, and the residual rotation of M is on a par with the observed rotation of M’; there is no fermentable sugar formed from starch, the action of the diastase being almost wholly arrested. The dextrin produced here must be nearly akin to soluble starch,* very little of it is present as malto-dextrin, and it is probably readily degradable by cold water malt extract.
* Experiments were made to settle this point, and showed as follows:—
K 160° F., unboiled filtered wort with iodine showed neither soluble starch nor erythrodextrin.
L 170′ F., unboiled filtered wort with iodine, some violet erythrodexlrin and strong red- brown erythrodextrin, some starch in grains.
M 180° F., unboiled filtered wort with iodine, soluble starch and violet erythrodextrin, much starch in grains.
It seemed to us that further light could be thrown upon these results by calculating a value or index, which we will call p (CuO value per 1 gram of sugars fermented), and an optical value or index, a (rotation in Ventzke divisions for a solution of 1 gram of solids per 100 c.c. taken in a 200-mm. tube); the figures obtained are shown in Table VII, and some interesting points have thereby been brought to light, and not susceptible of easy explanation.
The p and a values have been worked out for the solids in grams per 100 c.c. gained at each step of temperature, and also for the calculated sugars removed by fermentation. The solids gained are, of course, influenced by any fluctuation in the soluble inactive matter; the p and a figures from sugars fermented should give a clue to the nature of these last.
Comparing the two sets of figures, solids gained and sugars fermented, we find that, with a general tendency to a less amount of fermented sugar in the boiled portions, they march fairly together till 110° F., when the two sets fairly agree in the unboiled portion, but in the boiled portion, 4·5 per cent, of solids gained has not been fermented, whilst at the next stage (120′ F.), G’, the fermented sugar is less by 12 per cent, than the gained solids, whilst we encounter a high residual rotation in the fermented liquid. It may be affirmed that at 110° F. there is a first indication of unfermented dextrin not associated with uufermented maltose, and at 120° F. there is a very definite production of combined maltose, both combined maltose and combined dextrin having been degraded and fermented in the unboiled portions during the progress of fermentation; and the power of the diastase to degrade during fermentation is plainly to be seen even in H at 130° F. (this is n very interesting and important point for the distiller). From a comparison of the values for a and p of F, G, and H, the carbohydrates would seem to be not very dissimilar in character.
Comparing the a and p values and discussing them, we find that in all the experiments up to 100° F. there is a tendency to a high p; at 110° F. we get practically the maltose value (p = 1-37), and up to 150° F. this value is fairly well maintained. It is quite evident that some substance of much higher cupric reducing value than maltose is being produced in the earlier experiments, whilst the a of the gained solids tends to a figure much below the rotation of maltose, or it shows a value so disproportioned to the p that we can only suppose bi-rotation to have occurred (we refer especially to C). There can be no doubt that a sugar of much higher reducing power than maltose is formed in the early experiments, B, C, D, and E. We suggest, that dextrose is the most likely substance and, excepting the anomalous case (C), the a is not altogether inconsistent with this supposition (z for dextrose, 3·05). There is probably a very complex state of affairs at the low temperatures of conversion, degradable dextrin, maltose, and dextrose being possibly present at the same time. Up to 100° F. the dextrin susceptible of degradation would seem to be degraded on raising the wort to boiling, so that very little end or residual dextrin is shown. The complication is possibly due to there being two phases of enzymic action, during the earlier of which dextrose is produced, the outer portion of the starch granule being the first which is attacked; from this the dextrose may come, or it may be due to the transformation of some carbohydrate substance other than starch, the amylans, for instance.
If the a and p values from 110°—170° F. are averaged, we get the following figures, which, by this mode of treatment, come closer together. Boiling leads to an increase in the reducing power and a decrease in rotation, due to degradation of amyloin substances :—
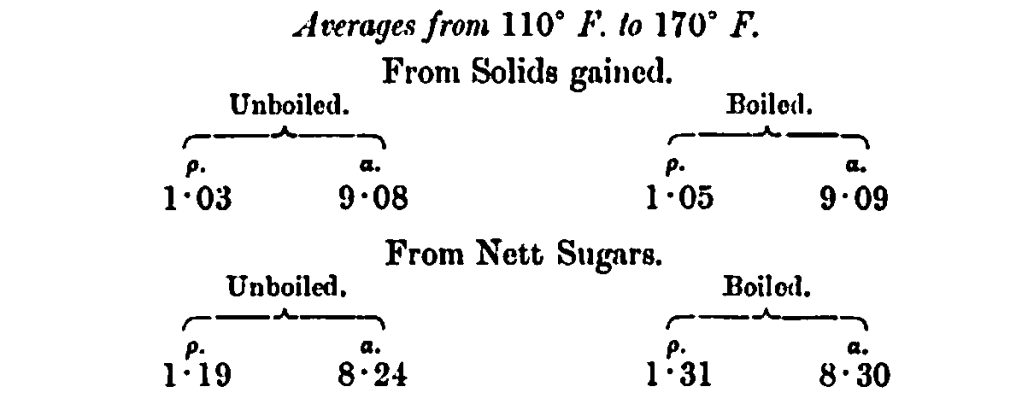
The figures from the solids gained indicate more dextrin and less maltose. The values a and p of the sugars fermented, as between the limits of 110° and 170°, approximate to maltose only.
We feel that there is very little chance of absolutely solving these questions till barley and malt starch be made the subject of enquiry, and the issues somewhat narrowed.
From what has been already said, it will be gathered that an expression of the analytical data, in terms of composition of wort, is no easy matter.
If the fermentable sugars of A be taken at 11·25 per cent, on the malt (the constant figure adopted), we get for the value of a 4·7/1·125 = 4·17 (O’Sullivan-Heron divisor = 4·3), and for the value of p 1·38÷1·125 = 1·22 (O’Sullivan-Heron divisor = 1·65), but since some of this is cane sugar, the p would be just so much lowered, and by Heron’s, method we can, of course, calculate what the amount of cane sugar is. It is shown in the analysis of the standard malt. The a and p values for the first three pairs of experiments are:—
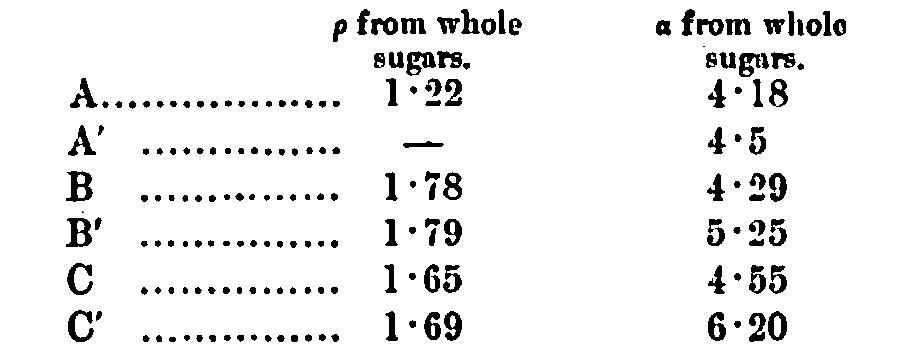
The O’Sullivan-Heron values apply fairly well, excepting that it is evident in C’ that the gain in angle is not cane sugar; the abnormally low inactive matter shows this plainly; the gained angle is probably due to starch products.
The figures of D and D’, E and E’ appear to us to be only calculable on the supposition that dextrose is formed at these temperatures and exhibits bi-rotatiou. Tho p is far too high to permit the idea of amyloin substances. There appears to be only a trace of residual maltose, but D’ and E’ show some residual dextrin. It will be noticed that the optical activity has fallen materially in the boiled portions, taking the a of the sugars fermented. If the whole gain in reducing power correcting by the cold water malt extract reducing power of 1·38 be taken as dextrose in a bi-rotating state, and that the a be raised somewhat by the presence of maltose, we can calculate out the following values:—

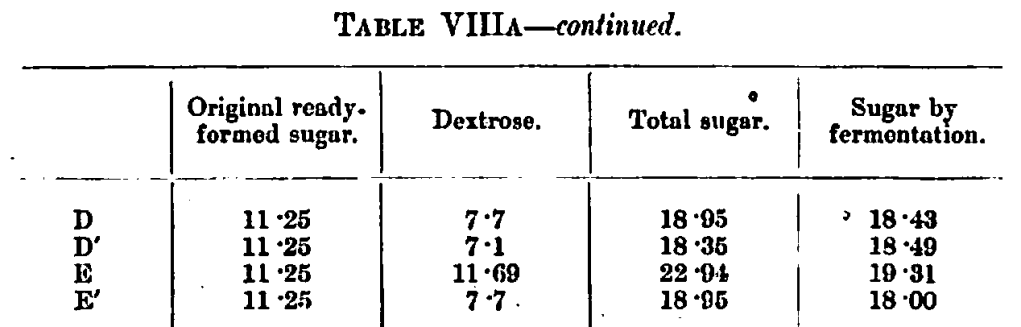
We now come to F and G experiments at 110° F., and find that the boiled worts F’ and G’ are not calculable in terms of maltose and dextrin. The assumption that suits the case best is that dextrose was formed in F’ in the proportion of one of dextrose to one of maltose, and in G’ one of dextrose to three of maltose. This was foreshadowed by the a and p values for sugars fermented shown in. Table VII.
The agreement between the fermented maltose and dextrin, calculated from the reducing power and rotation as compared with the net sugars removed by fermentation, is good.
It should be noted, too, that where some diastase has been left (unboiled worts), the residual dextrin is far less than in the boiled worts F’, G’, and H’. By the time a temperature of 140° F. is reached, the diastatic power is presumably much modified in its action, but is still able to degrade dextrin or amyloins, and so leave less residual dextrin than the boiled portion I’. This seems to hold somewhat even at 150° F., but at this temperature the composition of the wort is very similar, whether boiled or unboiled, but there is a fresh complexion about the composition of the wort shown by the low figures for residual maltose. These results are the moans of several experiments, which on these points agreed well with each other. It seems as if the diastatic power had come to an optimum as regards relationship of fermentable to unfermentable and undegradable products, (degradable by fresh cold water extract, but not by residual diastase of the experiments) and it seems to mark 150° F. as an important point of temperature, which, indeed, we think, it has generally been considered to be. The degradation and fermentation of dextrin during earlier experiments has, doubtless, led to difficulties in interpreting the analytical data, whilst earlier still the apparent production of dextrose is a still more unsettling factor; but most of the experiments from 110° F. to the final temperature of 180° F. can be expressed in terms of maltose and dextrin, and they are thus calculated in the Table VIIIB.
Referring once again to the interesting results obtained at 150° F., we should say that the fact of the malt being finely ground has led to a difference in the composition of the wort (see standard analysis, Table V), the maltose being much lower and the dextrin proportionately higher, the total starch transformation products being about the same.
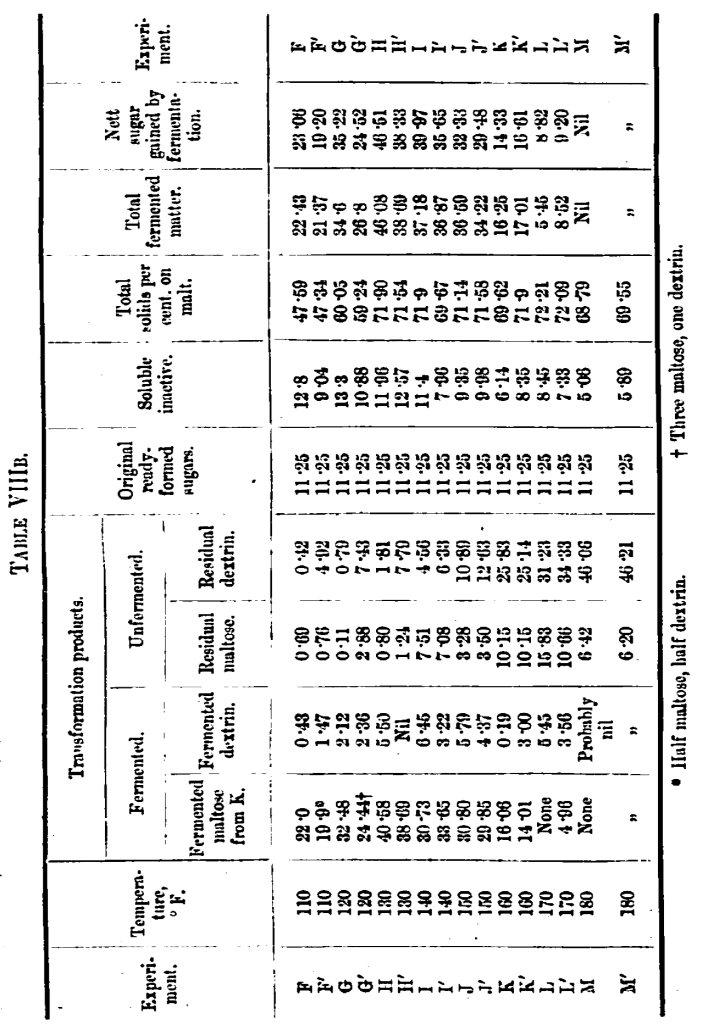

Malto-dextrin or amyloin appears to be formed first markedly at 120° F., though there is evidence of some little combined maltose being present at temperatures lower than this.
Other features shown by the figures are the considerable quantity of dextrin disappearing during fermentation even in boiled solutions. We suppose this to be combined dextrin in a somewhat readily fermentable form. The maximum fermentability of the wort appears to have been reached at 130° F. (H), where the residual maltose and dextrin only amount to 2 per cent, on the malt, 57·7 per cent, being removed by fomentation (this is again a very important point for the distiller, though probably there would be some variation according to the kind of malt which was being dealt with).
At 160° F. the restriction of the diastase is very obvious; the fermentable matters come down with a run, the residual maltose and dextrin together being about double the fermentable matter. At 180° F. there is still a slight conversion into amyloins, but no production of fermentable matter beyond the original ready-formed sugars, and there is even a reduction on these, owing, no doubt, to the fact that 1 or 2 per cent, of the original amount arises from the action of diastase on readily degradable substances.
CONCLUSIONS
The main conclusions we draw from the results may be summarised as follows :—
1. That the starch in a well-made malt exists in a state very different to that of the original barley starch.
2. That this modified starch is readily acted on by diastase at temperatures to which potato starch (and most probably barley starch) would not respond.
3. That malt starch will degrade almost completely to maltose at comparatively low temperatures in unboiled fermented wort. Temperature, 110°, 120° and close on 130° F.
4. That there is strong evidence of dextrose being formed in the low temperature transformations.
5. That the bulk of the extract of a good malt ground to about 25 Seck is obtainable at 130° F.
6. The O’Sullivan-Heron values for the ready-formed sugars of malt agree very well with the estimate of these sugars made from the specific gravity lost during fermentation.
It is purposed to carry out a further series of experiments rather on the lines of those detailed, but using barley and malt starch. Some factors of disturbance should in this way be eliminated and we feel sure that more valuable information is to be obtained than by experimenting with malt or with what might be termed unmalted starch. We have prepared fairly considerable quantities of barley and malt starch (specimens exhibited) which, together with the residuals, we are able to place before you, believing they will be of interest. Without anticipating future work, we may say that the behaviour of the two starches on heating up with water is quite dissimilar. The malt starch readily yields and becomes mobile, something like soluble starch, whilst the barley starch shows a definite gelatinisation at a temperature of 170°—175° F., thus agreeing with Lintner’s figure.
DISCUSSION
The Chairman said he was sure they would all agree that they had had the pleasure and benefit of listening to a most interesting paper, which contained a vast amount of useful information.
Dr. A. K. Miller endorsed the Chairman’s view that the paper was one of much interest, and the amount of work it had involved would be apparent to all. The large number of figures in the numerous tables shown were, however, difficult to assimilate during the reading of the paper, and would require further study before it would be possible to discuss thoroughly the various points raised. He would like to ask a question, however, with regard to the readiness with which the starch granules of a well-modified malt were converted at low temperatures. Had it occurred to the authors to make use of this property as an additional test in malt analysis with a view to compare and judge the degree of modification of the starch in malts? It would, of course, be necessary to make a special mash for this test, but it occurred to him that it might be of value.
Mr. Matthews said, with regard to Dr. Miller’s point as to the conversion of malt starch granules, it had not struck him that this fact could be made of use as a practical test, but he was inclined to think this might be done. As Dr. Miller had indicated, they might turn to practical account the fact of malt starch yielding so readily at such low temperatures. As to the water used: if they adjusted the water temperatures so as to get a fixed initial temperature, and kept it at that for two hours, they would get more dextrin the more water they used. If they altered the volume of water, they must also alter the striking heat to arrive at the same initial.
Mr. Glendinning concurred in what Dr. Miller had said as to the amount of work this paper had involved, and said it was impossible to deal with all the facts and figures produced, off-hand. They required careful study. He was much interested in what Mr. Matthews said as to dextrose being suspected of being formed at the low temperature of 90° F. At that temperature there was very little diastatic action taking place. It occurred to him that the dextrose might be the result of the inversion of the cane sugar present. As regards the possibility of bi-rotation mentioned by Mr. Matthews, had the authors sought to remove this by addition of alkali?
Mr. Matthews did not think the possibility of the inversion of cane sugar weighed much. He could not say, however, that there was no inverting action, but he did not think that in ordinary cases there was any active agent in cold water extract of malt that acted on cane sugar in a degree worth considering. They had not used alkali as they might have done; the bi-rotation was an unexpected occurrence.
Mr. C. F. Hyde asked for information as to the process adopted in separating the starches exhibited from the malt and barley; he also inquired if the authors observed that mashes at about 150° F. filtered with greater facility than at the higher temperature of 160° F.
In reply, Mr. Matthews gave a detailed account of the methods employed for starch separation, and said it had been a work of very great difficulty, and at one time they almost despaired of success. As to filtration, he remarked that if they wanted to make a cold water malt extract filter as badly as possible, all they had to do was to add some alkali to the infusion. (referring to ammonia more particularly) He preferred to deal with these without additions. They experienced the greatest difficulty in filtering at 170°—180° F. Mr. Auty and he found it most difficult to filter these mashes, owing to the presence of the soluble starch, but they had got the filtrates clear by using a little alcoholic potash.
The Chairman remarked that the paper was most instructive and contained a mass of information which would require careful study.
Mr. J. G. Wells proposed a hearty vote of thanks to the authors of the paper, and in doing so, associated himself with the view that the numerous tables of analytical results that had been laid before them could not be discussed without study. They were greatly indebted to Messrs. Matthews and Auty for the paper, which they had all listened to with much interest.
Mr. T. Hyde, in seconding the vote, said the paper had been exceedingly interesting, and more especially must it have been so to the brewers present, who had had the advantage of a scientific training. He thought the authors would appreciate the suggestion that the nature of the paper precluded a full discussion upon it until they had had an opportunity of studying the facts and figures presented to them. He was glad to sec so large an attendance of those engaged in the brewing trade in that district, and they would all admit they had listened to a most valuable paper.
The resolution was carried unanimously and was suitably acknowledged by Messrs. Matthews and Auty.