MEETING HELD THURSDAY, 16th APRIL, 1896, at the Grosvenor Hotel, MANCHESTER.
Dr. W. L. Hiepe (President) in the chair.
The following paper was read and discussed :—
INVERT – SUGAR (Part I.)
by John Heron, B.E., F.I.C., F.O.S.
At one time the term sugar was employed to convey the idea of any substances which were sweet to the taste, many of which had no relation to the wants or usages of ordinary life. For instance, acetate of lead, of a decidedly poisonous character, is commonly called Sugar of Lead from the sweetness of its taste. Nowadays, however, the term is exclusively confined to those sweet substances which exist in, or are abstracted from, plants, and that directly conduce to, or are connected with, our modern comforts. Thus we have cane-sugar, beet-sugar, palm-sugar, maple-sugar, malt-sugar, milk-sugar, grape-sugar, &c.
Scientifically they are all classed under that generic group of organic bodies termed “carbohydrates.”
The term carbohydrate is applied to that group of bodies, the best known members of which contain, in the molecule of which they are composed, 6 or a multiple of 6, atoms of carbon united with hydrogen and oxygen, the two latter elements being present in the proportion in which they unite to form water; thus the formula of grape-sugar is CsHu08, that of cane-sugar C6H12O6. They consist of several distinct groups, most of whose members exhibit optically active properties rotating the ray of polarised light either to the right or left.
That group which most nearly concerns us at present is that one known as the sugar group.
This may be subdivided into the following classes:—
(a.) The glucoses or monoses, represented by the formula C6H12O6, of which the most commonly occurring members are—Dextrose or grape-sugar. Levulose or fruit-sugar. Galactose.
(b.) The saccharoses or bioses, represented by the formula C12H22O11, of which the principal members are—
Sucrose or cane-sugar.
Maltose or malt-sugar.
Lactose or milk-sugar.
(c.) The trioses, represented by the formula C18H32016; only one member of this sub-group is at present well known, namely, Raffinose.
The group, of which cane-sugar is the most prominent member, may be considered as formed by the union of two monose molecules with elimination of water, and its members, therefore, possess a constitution analogous to the ethers. All the members of this group, when boiled with dilute acid or subjected to the action of certain enzymes, take up water and are converted into glucoses; cane-sugar, for instance, when thus treated becomes converted into a mixture of equal proportions of dextrose and levulose. Such a substance is widely distributed throughout the vegetable kingdom, and is called invert-sugar because it rotates the polarised ray to the left; for although dextrose and levulose are present in equal proportions, the rotation produced by levulose being nearly twice as much to the left as that of dextrose is to the right, the result is a left-handed rotation for the mixture of the two.
This inverted sugar was recognised so far back as the last century by Howitz and others as a substance totally distinct from cane-sugar, but nothing was known of its optical properties until the year 1836, when Biot, who made a very profound study of the action of various substances on polarised light, showed that the different kinds of sugar may be distinguished from one another by their action when in solution on a ray of polarised light.
Although invert-sugar was, as I have just mentioned, recognized as being different to cane-sugar more than a hundred years ago, it was not until about the year 1836 that much was known as to the method of its formation and of the nature of inversion of cane-sugar. Since that time an enormous amount of work has been done on this subject; indeed, if I were to go minutely into the history of all the investigations that have been carried out on the inversion of cane sugar, I should produce a large volume of several hundred pages; at the same time, I think it will be more or less opportune if I just refer at this stage in the briefest manner possible to what is known at the present time about the inversion of cane-sugar.
As has been already stated, cane-sugar when acted upon by dilute acids or enzymes becomes transformed into a mixture of equal proportions of dextrose and levulose.
Such a reaction may be represented by the following chemical equation. The formula for cane-sugar, C12H22O11, may he written thus,

which, when undergoing inversion, splits into two molecules, one of which at the same time takes up a molecule of water, thus:
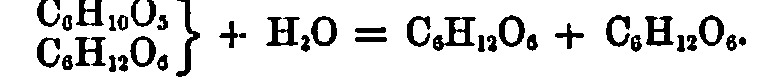
Following up this nomenclature, we see that cane-sugar has already the molecule dextrose contained within itself as it were, whilst the C6H10O6 molecule becomes hydrated to levulose.
1 do not suppose there is any substance which, under favourable circumstances, undergoes hydration so easily or so readily as cane-sugar. Prolonged boiling with water is sufficient to produce inversion. Dilute acids, even weak acids, such as carbonic, produce inversion slowly in the cold, but when heated at higher temperatures the action is in some cases extremely rapid.
Certain enzymes, whether derived from animal or vegetable substances, also have the power of inverting cane-sugar—in some cases with comparative rapidity, as for instance the invertase of yeast.
That member of the carbohydrate group which immediately concerns us is the one known as cane-sugar or sucrose.
The principal varieties of sucrose known in commerce are cane-sugar, properly so called, beet-sugar, palm-sugar, date-sugar, maple-sugar, maize-sugar; of these the two most important are cane-sugar and beet-sugar.
Cane-sugar is that variety of sucrose which is extracted from the sugar-cane, a plant which grows only in tropical and subtropical climates. The sugar-cane (see Fig. 1) is in reality a gigantic grass having a knotty stalk, and at each knot a leaf and joint; the number of joints on each full-grown stalk or cane varies from 40 to 60, and on every joint is a bud enclosing the germ of a new cane;

under cultivation, very few of the canes flower at all, so that propagation is dependent entirely on cuttings. As soon as the canes are ripe they are cut down with hatchets as close to the roots us possible; the juice from the lower joints being the richest in sugar, the top of the cane is discarded, and all leaves are also stripped off. As soon as the canes are cut they are carted away in bundles to the factory where the juice is extracted; this is usually done by passing the canes through powerful rollers, either of wood or iron, whereby the cells of the plant are broken and the juice squeezed out. The cane mills are usually on the ground floor of the factory, whence the juice, as it is expressed from the cane, is pumped up to the top floor; here it passes through a series of strainers from which it is received in a shallow tray, and passes, by means of a shoot, on to the clarifier. The juice at this stage is very apt to undergo fermentation, and, in order to prevent this, is, as soon as over it leaves the mill, subjected to the action of sulphurous acid gas. As soon as it roaches the clarifiers heat is applied, either directly by fire or by steam, until a temperature of 170° to 180° F. is attained, when the liquor is neutralised with milk of lime; defecation then takes place, and, on the source of heat being removed, the impurities separate as scum and sediment; the clear sugar liquor is next run from here into a series of evaporating pans set over a flue, at one end of which is a tire; here the juice undergoes concentration, passing from one pan to the other, until it reaches the one nearest to the fire, and in this one it attains the crystallizing point. The mass is then .removed to coolers, in which it is allowed to grain, and is next transferred to casks or hogsheads having a number of holes in their lower part, which are temporarily closed by means of plugs. When the mass has solidified the plugs are removed and the syrup is allowed to drain off, the drained mass in the casks constitutes what is known as raw sugar.
The sugars of commerce may be divided roughly into two classes, Raw and Refined, each class containing many varieties and qualities, running imperceptibly one into the other; however, there is a broad line of distinction to be drawn between the two, for the term “raw sugar” is generally applied to sugar manufactured in a more or less crude manner on the plantation where it is produced, whereas the term “refined sugar” appertains more directly to those finer qualities of sugar which are manufactured from the raw, by the process called refining, at central factories, and generally far distant from the country in which the raw sugar is grown.
A little reflection will go to show that this is the most practicable and economical plan, for in those countries where the sugar-cane grows, although unskilled labour is cheap enough, on the other hand, fuel and water are usually scarce and dear, so that all the coal required for heating purposes has to be exported from this or some other European country. Machinery can only be obtained under similar conditions, and this has to be attended to and kept in repair by skilled workmen, in most cases Europeans; besides which the juice can only be extracted from the cane at a certain season of the year, that is generally from the middle of November to the end of March or April, when the cane is ripe for cutting, so that, for the remaining months of the year the factory on the plantation is more or less at a standstill.
Hence, on every ground, it is advisable to carry on the refining of the raw product in those countries where the combination of capital, machinery, skilled labour, production on a large scale, and continuous working will enable sugar to be produced at a cheap rate and of a uniform quality.
There are many different varieties of raw sugar, the quality of which depends altogether upon the methods employed in their extraction and curing.
Analyses of some of the principal varieties of raw sugars used in the manufacture of invert-sugar, are given in the following table, as well as analyses of what are known as molasses.
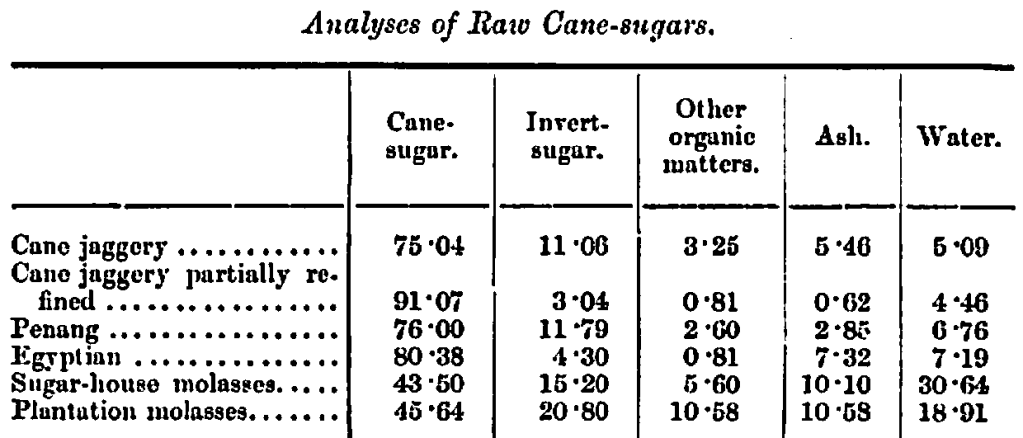
As will be noticed on studying these analyses, all raw sugars contain what, for want of a better name, are termed organic matters other than sugar, or non-sugar; these may be divided into three distinct classes, namely, organic acids, nitrogenous bases, non-nitrogenous substances; from such substances upwards of 60 definite chemical compounds have already been separated, and their properties determined; most of these are very undesirable bodies to have in malt-wort or beer, hence most of the raw sugars should be rejected as unsuitable for the production of even the commoner kinds of ales and stouts.
In the manufacture of refilled sugar, it is a matter of some considerable importance to the refiner not only to know how much cane-sugar the sample of raw product which he is going to treat contains but also the actual amount of cane-sugar which lie can obtain there from in the process of refining, and which is known as the available amount of crystallisable sugar, for it is now a well established fact that the presence of suits of mineral find organic acids, as well as of invert-sugar and organic bodies other than sugar, exercise a marked influence upon the crystallisation of sugar.
A study of these bodies and the effects produced by them upon crystallisation has given rise to a method of valuing raw sugars, which has been adopted in France and Germany, as well as in this country, and is known as the “rendement,” or the net amount of crystallisable sugar obtainable from a given sample of raw sugar.
This assumes that for every 1 per cent, of ash contained in the raw sugar 5 per cent, of cane-sugar is prevented from crystallising, and that for every 1 per cent, of invert-sugar present an equal proportion of cane-sugar is retained in the molasses. Hence the rule, from the amount of cane-sugar found by polarisation, deduct five times the weight of ash plus the weight of the invert-sugar which may be present. The remainder is taken as the refining value or “rendement” of the sample.
Thus, a sugar showing on analysis would, according to the above method, give 95 – (I X 5 + 2) = 88 per cent, available crystallisable sugar.

Now, to the invert-sugar manufacturer it does not matter whether there be much invert-sugar present in the raw material or not; he bases, therefore, the value of the sample on the total amount of sugar (cane and invert) which can be obtained.
So that a sample which might be altogether rejected by the refiner on account of the high percentage of invert present would be acceptable to the saccharam manufacturer, provided that the other bodies present, organic and mineral, were not exceptionally high. Preference is, however, given to one particular class of raw sugar, known commercially as jaggery. It is manufactured exclusively in India by the natives, in a crude manner, which has been pursued from time immemorial.
Each owner of a plot of ground grows a few canes, from which, when ripe, he extracts the juice, the cane being crashed in a rude wooden mill, somewhat resembling a huge pestle and mortar, the juice strained through a cloth and boiled down in earthenware pots to the crystallising point, or until it forms a solid concrete mass.
The impurities occurring in raw sugars are of two kinds:—
1. Mechanical, consisting of clay, sand, fragments of the cane from which the sugar has been extracted, and other insoluble matters. These can be mostly removed by filtration.
2. Chemical impurities, such as colouring matters, albuminous substances, vegetable acids, gums, and mineral salts, some of which are of a most objectionable character, but which are removed to a very large extent, or altogether, during the process of manufacture, reference to which will be made later on.
In the preparation of invert-sugar from raw sugar for brewing purposes, the following operations are carried into effect:—
a. Solution of the raw sugar.
b. Filtration,
c. Inversion with acid.
d. Neutralisation.
e. Filtration.
f. Decolorisation.
g. Concentration, or boiling down.
a. Solution.—The raw sugar is conveyed to the upper floor of the factory, the bags in which it is packed slit open, and the contents emptied into the melting pan (blow-up). This is generally an open cylindrical vessel, 4 or 5 feet deep, and of varying diameter, capable of holding from 3 to 10 tons of sugar in solution (Fig. 2). It is provided with mixing gear and one or two steam coils for heating its contents with close steam, or a perforated pipe for the admission of free steam; there are also cocks at the top for the admission of hot water, bag washings, &c.

The melting process is carried out as follows:—A sufficient quantity of water to cover the coils having been run into the blow-up, steam is turned on, the sugar run in, and water in such proportions that by the time the charge is complete the density of the solution is about 1150 and the temperature about 150° F. As soon as this is attained, the liquor is conveyed through metal strainers to remove the coarse mechanical impurities, and on to the Taylor filters on the floor below, or is first run into large settling tanks, where, after standing some time, a large portion of the sand, clay, and other impurities subside to a greater or less extent. During this stage defecating agents of one kind or another are often added to the liquor to assist in the separation of these impurities, the cleaner supernatant liquor being then run on to the filters.
b. Filtration.—As at present employed, the Taylor filter (Fig. 3) consists of a chamber (a) made of cast-iron plates open at the top, and having a horizontal divisional plate (6) about 1 foot from the top, forming a receiving tank or filter head. This plate is perforated with holes, in rows a short distance apart, to which are fitted gun-metal sockets furnished with screw threads for the reception of gun-metal bells (c), to which the bags are tied. The filter chamber is provided with a tightly fitting door (d), and cocks for supplying steam, and the bottom, which is doubly inclined, has a cock for the exit of the liquor. The rows of holes in the divisional plate are sometimes divided by partitions, so that every row of bags may receive its due charge of liquor. The bottom of the filter head is usually inclined, its lower end being under the liquor cock, so that each row
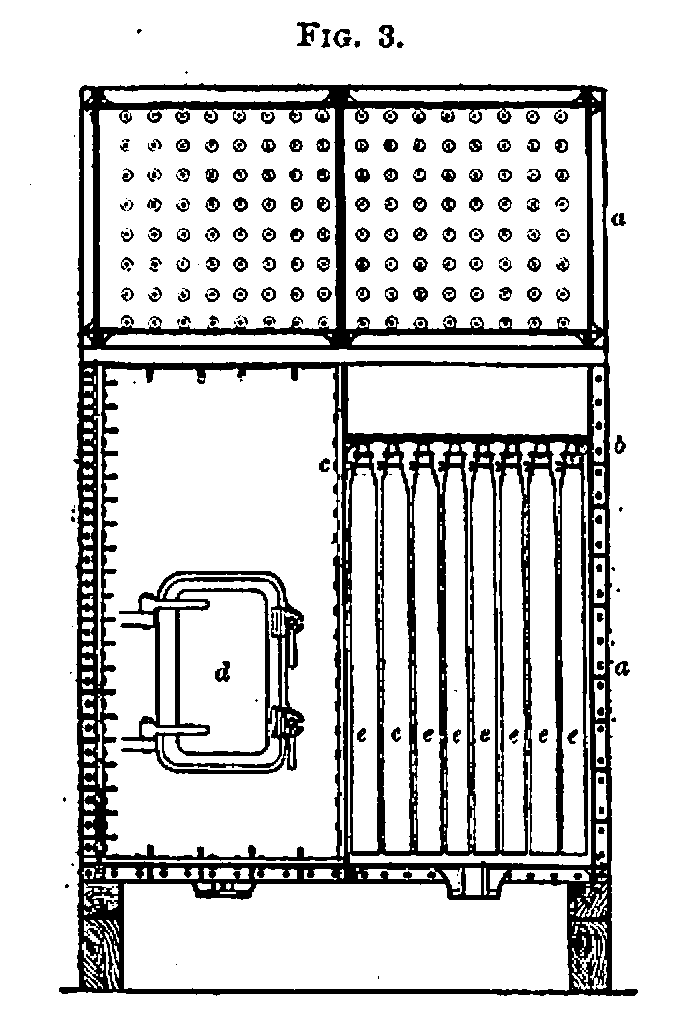
of filters may be filled in rotation and the work of the filter be rendered more uniform, thus attaining the same object as is effected by the use of partitions. The bags (e) are made of twilled cotton, and when laid flat are about 6 feet long by 2 to 3 feet wide, and they are either woven without a longitudinal seam or sewn up. These bags are crumpled up and slipped inside of coarsely woven hempen sheaths, which are open at both ends, and are about 6 inches longer than the bags, and from 6 to 9 inches wide when laid flat. These serve to restrict the expansion of the bags, thus giving a large filtering surface in a small space. The bell is inserted up to the screw part into the opening of the bag, and the latter, together with the upper end of the sheath, which is brought level with it, is firmly tied on with cotton cord. The bell is then screwed in its place, and on account of its shape causes a tight joint to be made when the bag is weighted with liquor. The number of bags contained in each chamber varies from 100 to 500, or even more, in different refineries. The amount of liquor that can be filtered through each bag mainly depends upon the kind of sugar, and also, to some extent, upon the temperature and degree of dilution and use of defecating agents. It is therefore impossible to give precise figures as to this point, but it may be said generally that in a refinery working with a medium quality of sugar from 10 to 13 bags per ton are required.
The mode of conducting the filtration is as follows:—The chambers having been filled with bags, the doors are screwed up, and the steam is turned on for a few minutes so as to thoroughly warm the filters. Any condensed water is then allowed to flow away, and the liquor is run on to the filter heads. As the liquor flows from the filter it is frequently sampled, and should it prove cloudy, owing to the rupture of any of the bags, it is run away into the sweet water tank. By carefully inspecting the filter head, the faulty bags can generally be detected by the unusually rapid currents of liquor passing down them, and their nozzles should be immediately closed up.
The clear liquor from the filters runs into fine liquor tanks on the floor below. When the bags become dirty and practically cease to allow the liquor to flow through them, the inlet cock is closed, and the bags are allowed to drain for some hours. Steam is then turned on, and the bags are filled with hot water and left to drain the second time, and this operation is repeated until a sample of the washings or bag-water no longer shows any indications of the presence of sugar when tested by a hydrometer. The bag-water is run into a tank by itself, and is used in place of water for dissolving raw sugar. When the filter is sufficiently cold, the bells are unscrewed, and the bags and sheaths are removed from the chamber, and the cords being untied, the sheaths are slipped off the bags, and the latter are then washed. The washing is conducted in a series of three or four tanks containing warm water; the first of these tanks contains the dirtiest water, and the last the cleanest. Each, bag is turned inside out in the first tank, and by a dexterous movement on the part of the workman is filled with air, which, is imprisoned in it by closing the opening with one hand. The bag is then forced under water, and the air is driven through its pores, thus removing the adhering impurities, and this operation is repeated in each of the other tanks, and finally the bag is turned with the right side out.
c. Inversion.—After filtration, the liquor is next run into the converter; this is usually a large wooden round, somewhat similar in size and appearance to a fermenting round in a brewery, made of oak or deal staves, and firmly bound with several strong iron hoops, and fitted, like the blow-up pan, with a steam coil or free steam pipe. It is generally covered in on top, and provided with a man-hole. It is also furnished with a stirring gear, consisting of wooden blades radiating from a central wooden axis and moving in opposite or similar directions; pipes pass down to the bottom of the converter for the purpose of conveying the acid for inverting and the lime and chalk for neutralising.
The liquor having reached the converter, the stirring gear is set in motion. Dilute sulphuric acid of about 1200 sp. gr. is next run in, in the proper proportions, and steam turned on until a temperature of about 175—180° F. is reached. The liquor is then kept continually roused, and samples taken out from time to time neutralised, decolorised, and polarised, the inversion being carried on until examination in the polarimeter shows that the proper degree of inversion is reached. When this is attained, a certain proportion of very finely ground animal charcoal is added, and the liquor kept constantly agitated for about 30 minutes.
d. Neutralisation.—The liquor is then neutralised with chalk or whiting. For this purpose whiting free from lumps is mixed up with water into a thin cream and cautiously added to the inverted solution. Owing, however, to the fact that many organic acids originally present in the raw sugar are set free on the addition of the sulphuric acid, and which are incapable of decomposing the chalk, in order to ensure that these acids are completely neutralised, milk of lime is employed to complete the neutralisation of such acids. For the inversion of cane-sugar on the large scale sulphuric acid is the one always employed. Owing to the comparative solubility of its lime salt in sugar solution, only the smallest quantity of acid necessary to bring about complete inversion in a given time is employed. Not only does the acid invert the sugar but it exercises another very useful function in breaking up the albuminous bodies to a certain extent, as well as the vegetable acid compounds, so that when neutralisation with lime takes place the greater part of these bodies become precipitated and are carried down with the sulphate of lime. The addition of phosphoric acid at this stage plays also a very useful part in effecting the ready separation of such impurities.
Perhaps I might here draw attention to an idea which seems to prevail in text books and amongst people ignorant of the manufacture of invert-sugar on the commercial scale, and that is that the sugar is actually boiled up with the acid, whence it must necessarily follow that a great deal of colour is produced in the inverted solution. Now, as I have already stated, the sugar solution is not boiled with acid, everyone who has had any experience in this manufacture knowing well that by such a proceeding an enormous loss of extract, due to caramelisation and destruction of the levulose, would ensue.
e. Filtration.—As soon as the neutralisation is complete the steam is turned off and the liquor let stand for a short time to allow the sulphate of lime to subside, the partially clear liquor is then run on to the filters as before. The deposit or scum which has settled out in the tanks, and which contains a large quantity of sugar liquor is ran into a filter press, whereby the residual liquor is separated from the scum which comes out of the presses in the form of large slabs, and which when broken up and dried, forms a useful basis for artificial manures, and as an absorbent medium for various fluids.
It contains about 40 per cent, organic matter; 52 per cent, mineral matter (consisting chiefly of sulphate of lime); and 8 per cent, of water.
f. Decolorisation.—After filtration the liquor is next decolorized and further purified by means of animal charcoal, or “char,” as it is technically termed. The arrangement employed for this purpose is known as the char cistern (Fig. 4). These cisterns consist of large cast or wrought iron cylinders, and are usually about 10 ft. in diameter and 20 ft. high, but the dimensions preferred by different refiners vary considerably.
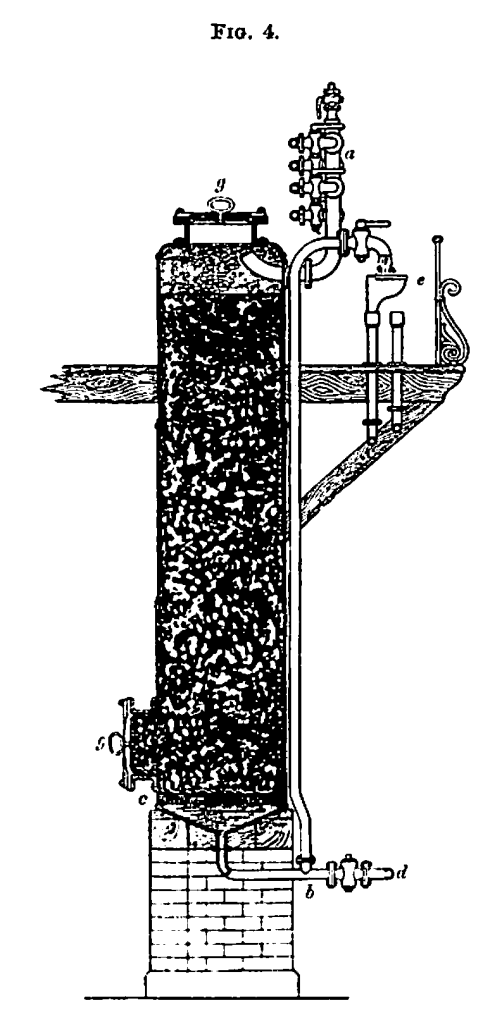
They are provided with manholes, g, g, at the top and side for the introduction and withdrawal of the char.
The raw liquor and water for washing find their way into the cistern through the pipe, a, which communicates with an upright main connected with various tanks containing a supply of water and of the different liquors.
The bottom of the char cistern inclines towards its centre so as to cause the liquor to run to the outlet pipe b. A short distance above the bottom is a perforated wood or iron false bottom, c, which when in use is covered with a blanket, or with a flaxen cloth to prevent the passage of any fine char. As shown in the engraving, the outlet pipe, b, is represented as terminating at a level above the top of the cistern head, and this is usually the case, the liquor being forced into the charcoal cistern tinder pressure. The water, however, is discharged by the cock, d, at the bottom of the charcoal cistern. The outlet pipe, b, terminates in a pipe having a swivel joint, and thus enables the liquor to be discharged into any one of the gutters e, leading to the storage tanks.
The cistern is filled with reburned charcoal through a jointed pipe, surmounted by a hopper into which the char fills from a band conveyer. As the char descends it is leveled by a man inside the cistern who also removes the pipe, in lengths, as the filling progresses. In some refineries this is done by mechanical means. It is questionable whether this leveling is of much consequence, but the prevailing opinion is that less sweet-water is made than when the char is not so leveled.
On the cistern being filled with char to within a short distance of the top, the cover of the upper man-hole is closed and, the cock at the bottom of the cistern having been opened to allow the air to escape, cold water is passed through the char to reduce its temperature, and this is followed by liquor. The whole of the liquor put upon a char cistern, may be of one quality, but it is usual to begin with a good quality, and to follow on with one or more lower qualities, the last liquor being made from syrups, or char washings resulting from previous operations.
When the liquor commences to come through, the outlet-cock is turned off, and the liquor is allowed to rest or “settle” for some time before it is run off.
If good raw sugar is being used, it is not necessary for it to remain in contact with the char for a longer period than the time requisite for its passage through the cistern which is from three to six hours, the time varying with the depth and degree of fineness of the char and other circumstances. The liquor is run off in a steady even stream, the speed of the flow being so adjusted as to leave the liquor sufficiently long in contact with the char to utilise its full decolorising effect. As the liquor runs away it is replaced by more raw liquor, until the increasing colour of the refined liquor indicates that it is no longer advisable to run on more. When this point is reached the raw liquor is replaced by water at nearly the boiling point, which forces out the liquor in front of it. The liquor, as it leaves the cistern, is usually divided into different qualities according to its colour, and the analysis of the raw sugar from which it is made.
The last portion of the liquor running from the char is reduced in gravity, owing to its admixture with water, and becomes cloudy and grey, and on this account has to be kept separate from the liquor, and it is therefore run into a tank by itself. This sweet water, which contains organic and saline impurities derived from the raw sugar, is known as char-water, and is either used for dissolving sugar, or is boiled into n low kind of sugar, which is again refined. When, by means of a hydrometer, it is ascertained that the amount of sugar present will no longer pay for recovery, the washings are run away to waste. The washing, however, is continued for many hours longer, so as to remove as far as is economically possible all organic and other impurities which have been absorbed by the charcoal. It is then allowed to drain, and is removed from the cistern to be dried and reburnt, or, as it is termed, revivified in the char kilns.
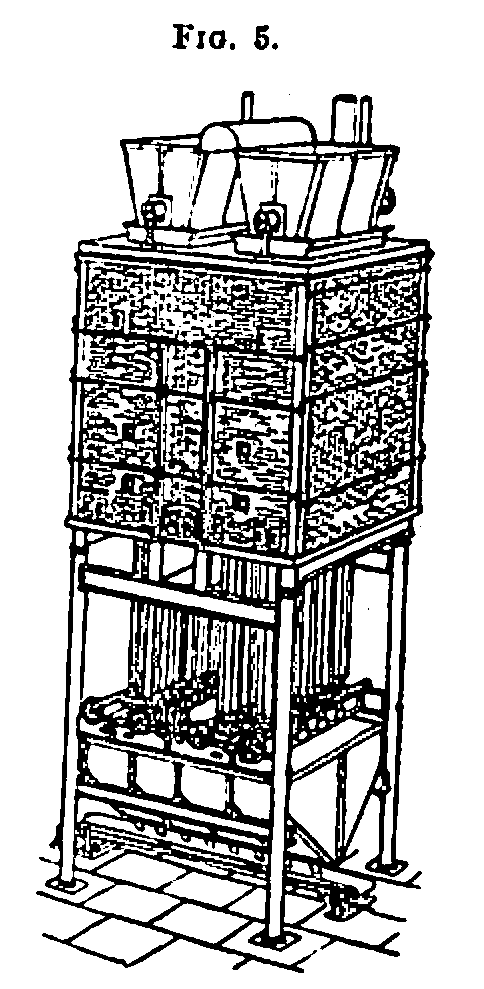
The kilns employed for this purpose may be divided into two classes; 1. revolving kilns; 2. pipe kilns. The first consist of cast iron cylinders revolving in an inclined position, heated externally, and through which the char is passed continuously, being turned over in its passage by lifting shelves, and discharging at the lower end into an air tight chamber. These kilns have now almost
entirely been replaced by the pipe kilns, the latest and best form of which is that designed by Messrs. James Buchanan and Co., of Liverpool, and to whom I am indebted for the loan of the diagrams and photographs relating to this portion of the paper as well as the following description of their particular form of pipe kiln.
This kiln (Fig. 5) contains 22 burning pipes of large diameter, placed vertically in two rows on each side of the furnace, the back rows being placed in such a position as to allow them to receive the direct rays of heat to half their circumference. Each pipe is made to revolve very slowly on its own axis, thereby exposing all parts to the direct action of the fire.
Within each burning pipe, an internal pipe is fixed, thereby leaving only an annular space to be occupied by the charcoal being burned. This pipe is perforated, and all steam and humid gases are freely taken away immediately and in whatever part of the pipe they are generated. Hollow louvres are cast at certain distances over the perforations, to keep them free, form collectors of the vapour, and turn the charcoal as it descends.
To each of the burning pipes coolers are attached, secured without bolts or fixings. The section and lengths give so large a cooling surface, and combined with the rotating motion, that the charcoal is cool when discharged.
The discharging apparatus is quite self-acting, and discharges the charcoal in measured quantities at given intervals of time, without any attention from the men in charge.
This kiln, combined with a drying apparatus, is capable of burning about 24 tons per 24 hours, but being provided with speed-cone gearing, it can be regulated as required to different quantities, according to the quality of the charcoal.
The kiln is made compact and contracted in the interior, so that a small area requires to be heated; the walls are made of such thickness as to retain the heat, which takes speedy effect on the charcoal, owing to the free escape of the steam, giving the result of a large quantity burned with very considerable saving in fuel.
The mechanical parts are well and practically arranged. All speeds being so extremely slow, there is comparatively little wear on the working parts, which are fixed below the coolers, quite away from all heat. The friction is reduced to the lowest point; on 1 H.P. will drive a kiln.
The kilns are made in different sizes, with or without drying apparatus, to suit requirements as regards available space, height, and quantity.
The action of the char on the sugar liquor is primarily to decolorize it, but in addition to this it exercises a decided purifying action as well, the charcoal being composed of carbon in a very finely divided state, deposited on a groundwork of phosphate of lime, it possesses the power of retaining most of the soluble impurities as well as the colouring matters. This absorbent power of char increases up to a certain temperature, somewhere about 180° F.
Certain inorganic salts, notably carbonate of lime as well as albuminous matters, are more or less permanently retained by the char, whilst others, such as salts of the alkalis, sulphate of lime, salts of vegetable acids, &c, are only temporarily retained, and are washed out again on the addition of thinner liquors, or by hot water.
Perhaps it may be of some interest at this stage to give a few analyses of various chars; these are shown in the following table.

The quantity of water employed in the operation of washing the char may be roughly stated as being from once to twice the weight of the charcoal. The cost of the water, the quality of raw sugar melted, and the degree of fineness of the charcoal, influence the refiner to a considerable extent in deciding upon what is the best quality to use.
g. Concentration.—After the liquor leaves the charcoal it has to be concentrated down to a thick syrup, and this is accomplished by means of the vacuum pan.
The principle governing the vacuum pan is based upon the fact that the boiling point of water or any other liquid varies according to the pressure of the atmosphere, the greater the pressure the higher is the temperature at which water boils, and, on the other hand, the less the pressure the lower the temperature of the boiling point; now the vacuum pan is subjected to such conditions in practice that nearly a complete vacuum is obtained, so that the sugar liquor boils at a temperature of 120° F. to 130° F., so that there is very little danger of burning or caramelising the sugar.

Fig. 6 shows the usual form of vacuum pan employed; it is made of stout copper, the lower end is provided with a discharge valve, a, and the upper end tapers off into a neck terminating in a safe or save-all, b, and this is again connected with a vacuum pump. When it is desirable to economise the water required for condensation, a surface condenser is employed.
From the save-all the vapour passes downwards into a condenser of extra large capacity, and fitted with a large injection pipe with valve and perforated copper 6pray pipe. The condenser is connected by means of a conical outlet pipe to the pumping engine.
The heating surface consists of solid drawn seamless copper steam coils, usually 3½ to 4 in. in diameter, with brass inlet and outlet pipes, and stayed to the sides of the pan with brass clips and rods; the pan is also supplied with the following fittings: cocks for the admission of steam, liquor, and air, vacuum gauge and thermometer for indicating the temperature and degree of vacuum, sight glasses through which the contents of the pan can be observed, and an arrangement of pipes and valves whereby samples may be drawn from time to time without the admission of air or reduction of the vacuum.
Each pan is worked by itself in the following manner: the air pump is started, and so soon as the vacuum reaches 26 or 27 in. of mercury the feed-cock is opened, and sufficient liquor is drawn in to completely cover the first coil; steam is next turned on, and the liquor rapidly concentrates, fresh supplies are admitted at short intervals until the liquid boils up to the “bull’s-eye.” The contents quickly boil down, the cock is opened again and shut off as before, when the liquor boils to the same height. This is kept on until the whole of the liquor intended to be boiled down is taken in. As soon as the requisite degree of concentration is effected, the syrup is run down from the vacuum pan into large tanks or receivers, from whence it is drawn by means of a sluice valve and shoot into the carriage casks.
Left to itself, the saccharum, as we must now term it, will remain in a state of syrup for several days or even weeks, depending, to a certain extent, on the gravity to which it was boiled. But if, on the other hand, whilst it is still hot in the tanks into which it bus been run after leaving the vacuum pan, it be agitated for some time, or, better still, some small lumps of a previous lot which has gone hard, be stirred in, on cooling, the whole mass will become crystalline, and solidify to the consistency of crystallised honey. This is known commercially as solid saccharum, whilst the non-crystalline is distinguished as fluid saccharum. Fluid saccharum is generally boiled to such a gravity as that 2 cwt. of it will give an extract in brewer’s pounds equivalent to 69·0, whilst solid saccharum is boiled to give an extract equivalent to 72·5.
Some few brewers are still in the habit of inverting their own sugar in the brewery, previous to adding it to the wort, the methods employed being inversion either (1) by acid, or (2) by yeast.
With regard to the adoption of either of these processes by the brewer, I consider that he runs a very great risk.
In the inversion in the brewery by means of acid, as no two samples of raw sugar are alike in their composition, in order that a proper and complete inversion should be made, it becomes necessary to test each and every lot undergoing inversion by means of the polarimeter. In most cases the sugar solution is actually boiled up with the acid for an hour or longer, resulting always in caramelisation as well ns loss of extract; then, after neutralising, a large amount of the Sulphate of lime formed remains in solution, and is carried into the wort, a proceeding which is exceedingly objectionable.
Where the inversion is carried on by yeast, the variation in composition, as well as other properties of raw sugar, render this action very uncertain; for instance, some raw sugars are distinctly acid to test paper, whilst others are ns distinctly alkaline. Now, it has been found by O’Sullivan and Tompson (Journal Chemical Society, 67, p. 847), when acting upon cane-sugar with invertase, that it is absolutely necessary, in order to get comparable results, that the acidity of the solutions should be such that it is in the most favourable condition for the action of invertase; if the required degree of acidity be overstepped, the action of invertase, especially at 55° to 60 C° (the temperatures most favourable to the invertive action), is almost stopped. On the other hand, the presence of the caustic alkalis, even in very small proportions, is instantly and irretrievably fatal to the reaction, and the hydrates of the alkaline earths are extremely detrimental to it.
Whatever may be stated to the contrary, there is no doubt but that during the process of inversion by yeast a certain amount of weakening of the yeast cells does take place. The action of invertase in the living yeast cell on cane-sugar is not such a simple and direct one as in generally imagined; at one time it was thought that the process of fermentation was simply a splitting up of dextrose into carbonic acid and alcohol, and could be expressed by a simple chemical equation; recent researches, however, tend to prove that it is a complicated process, and that certain other bodies are formed besides alcohol and carbonic acid, and so it is with inversion in the splitting up of and hydration of cane-sugar into dextrose and levulose, contain secondary products, principal among which is lactic acid, are formed, the presence of which certainly do not tend to improve the finished product.
It also appears to me to be a most dangerous as well as a very objectionable proceeding to boil up the yeast used for the inversion with the worts in the copper. We all know what a very turbid solution is obtained when yeast is boiled with water, and how difficult it is to filter it in any way bright; added to this is the fact that raw sugars of themselves never give a bright solution, besides which they contain all the several impurities, which in the ordinary course of refining, would have been removed, and which no brewer, if he were fully alive to the fact, would ever dream of permitting to enter into the composition of his wort.
I have particularly mentioned raw sugars because it is my experience that in some cases where brewers invert their own sugar, they generally select a low and cheap quality. I should have hardly deemed this a matter worth referring to at all, were it not that in a paper by G. R. Burdass (Transactions of the Midland Counties Institute of Brewing, 1, 53) I find it stated “we can use Egyptian sugars, and what is still cheaper, the syrups which refiners always have to dispose of at a low figure,” and which he states can be inverted in the brewery—then are given analyses of a sample of syrup as compared with those of saccharum. Time will not allow me to criticise fully these analyses given by Mr. Burdass, but all I can say is this, that I have never yet seen a sample of sugar-house molasses or syrup yield such comparatively good results on analysis as those obtained by Mr. Burdass, and that the samples of invert against which they are compared are very bad ones indeed; in fact, they could hardly be worse. I can hardly consider this a fair comparison, and indeed in the discussion on his paper Mr. Burdass admits as much.
Of course, it is to be understood that such syrups are only used for mild ales and stout, but from what has been already said, I think it will be readily seen that under any circumstances a cheap raw sugar is not a very desirable product to use even in stout, whilst what in termed syrup, known commercially as sugar-house molasses, is simply the refuse of the factory, and as such contains all the mineral and organic impurities of the original sugar in a concentrated form, and is therefore a totally different article to invert-sugar, and with which it cannot be compared for one second.
Taking the figure obtained for the syrup as shown in Mr. Burdass’ analysis, and on the assumption that 25 per cent, of this syrup would be used in place of malt, a brewer would thus be adding to his beer 40 grains per gallon of mineral and organic bodies, not sugar, the composition of which we are more or less ignorant. What brewer, in his senses, would care to make so dangerous an experiment?
As yon are all well aware, for several years past efforts have been made by a small band of fanatics to try and hamper the brewer in every conceivable way, by raising the cry against the use of so-called adulterants in the manufacture of beer, more especially the use of sugar and other malt adjuncts, it being contended that legislation in this direction would effectually prevent such adulteration, and would, at the same time, afford a measure of relief to the British farmer.
After the very able and exhaustive paper on this subject which has been read before you by Mr. Reginald Mortimer in January last, it is not my intention now to enter largely into this question, suffice to say that, at the present time, with barley of such a character as is now grown on English soil, it would be impossible for any brewer to produce an all-malt beer which would be acceptable to the public or would prove either wholesome, sound, or palatable.
Consequently the brewer is compelled, whether he likes it or not, to substitute for a certain proportion of English malt either foreign malt or some form of malt adjunct. If he is to brew a beer which will be found acceptable to the public taste, that form of malt adjunct which finds the greatest favour amongst brewers, and which has been in use for the longest period is invert-sugar, known commercially as saccharum and levo-saccharum.
Before the year 1847 no sugar was allowed to be used by distillers except that which was duty paid; furthermore it was not permitted to be used as a substitute for malt in the brewing of beer.
Malt, although liable to an excise duty, was allowed to be used duty free in distilleries, the duty being charged on the spirit produced from it. This very naturally caused the West Indian planter, who was at that time the principal sugar producer, to complain of the injustice done to colonial sugar in prohibiting it from being used like malt, duty free in distilleries, so that in 1846, due to this, public attention was directed to the matter. It was not then known what was the value of sugar, as compared with malt, to the brewer and distiller, so that the matter was investigated by the Inland Revenue department, in order to settle these points to the satisfaction of all parties. When this had been done, the ground taken by the sugar producer appeared to be so sound and just, that in the year 1847 sugar was permitted to be used in distilleries duty free, and in breweries on the payment of a customs and excise duty equivalent to that charged on the malt, for which it was used as a substitute. For a number of years, in fact, not until 1875, did the use of sugar make much headway in brewing, very severe restrictions being placed upon it, so that it was only in those years when good barley was scarce that it paid the brewer to employ sugar at all; the system of brewing in vogue in those days also conduced to restrict its use.
The beer brewed then was very much stronger in character than it is now; it was generally brewed in winter, was stored in vats, and kept for long periods, and to ensure its keeping sound was heavily hopped. Slowly a taste for a lighter and less alcoholic class of beer began to spring up. This meant storage for much shorter periods, so that quickness of consumption was thus fostered. This necessarily involved earlier conditioning and brightening in cask, and to meet thin new state of things the brewer has been led to use less malt, fewer hops, and more sugar.
Up to the year 1873 comparatively little sugar was employed in brewing, but from that time to the present the use of sugar has grown in favour with brewers, so that whilst in 1873 only 417,091 cwts. of sugar were used, last year 2,274,148 cwts. of sugar were employed.
For the last 50 years the use of sugar in brewing has been recognised by Government as a legitimate material in the manufacture of beer, and whilst the public taste generally demands a light beer of such a character as can only be produced with the aid of sugar, no agitation of so called protectionists will ever be able to prevent its use in the brewing of that beverage which we claim as our national drink.
The advantages arising from the use of invert sugar in brewing are many, the principal of which may be summed up as follows:—Speediness of clarification and increased brilliancy, and hence increased stability, for it may be taken as a general rule that a beer which tends to become spontaneously bright, or clarifies quickly, keeps well; although the addition of invert-sugar diminishes the percentage of soluble albuminoids, as one would naturally expect, it has also the properly of dissolving up certain of these bodies which would otherwise probably remain in a state of semi-solution, but which, by entering completely into solution, disappear to a greater or less extent during the primary fermentation along with the sugar, and hence the result is a readily brightening beer.
Early conditioning is also favoured by the use of invert-sugar, and there can be no question but that, for some reason which it is difficult to explain, a certain amount of fulness is imparted to the finished product. Here, then, are all the qualities which characterise the light beers of the present day, and which every brewer strives to attain in his own particular brewery.
A beer in the brewing of which invert-sugar has been employed has a certain pleasant, clean, agreeable taste about it which is not to be obtained by the use of any other malt substitute, reminding one to a very large extent of the pleasant taste produced by honey, and which properly made invert-sugar from the sugar-cane always possesses. It seems to be in some way called into existence, or at, any rate intensified, by the combined action of acid and heat, for curiously enough, this peculiar flavour is not to be found in sugar inverted by the action of yeast, neither is it present in invert-sugar derived from beet-sugar.
A paper on such an important subject as the one I have chosen would hardly be complete without some reference to the analysis of commercial invert-sugar. Time will not permit me to enter fully into this question just now, so that I can only refer to some analyses of the three qualities of invert-sugar at present on the market. I do not regard these as typical, or infer that they should be taken as standards, but at the same time they will serve to give some idea as to what the composition of the various qualities of properly prepared invert-sugar should be.

All three qualities when liquid ought to exhibit a clear transparent appearance, and on dissolving in water, and making up to a sp. gr. 1040 ought to give a clear transparent solution, which, when raised to boiling, should throw no deposit, and should show the following reading at 60° F., in a 200 mm. tube on Laurent’s polarimeter, on what is known as the sugar scale of that instrument, Nos. 1 and 2 from —11 to —15; No. 3, —6 to —11. That is to say, no sample at a gravity of 1010 should give a reading of less than —6·0 divisions on the sugar scale of Laurent’s polarimeter.
No. 1 should show no traces of iron or acidity.
No. 2 only very slight traces of iron. No acidity.
No. 3, slight traces of iron. Very slight acidity.
I am fully aware of the fact that 1 have only been able to give you a very hurried and imperfect sketch of the manufacture of invert-sugar. I can only excuse myself by saying that pressure of business has encroached to a very great extent upon the little spare time which I was able to devote to the preparation of this paper.
In conclusion I desire to thank my friends, Messrs. Newlands Bros., of London, and Mr. James Buchanan, of Liverpool, for their kindness in lending mo the diagrams and photographs which have served to illustrate this paper, as well as for some of the descriptions of portions of the plant employed in the manufacture of invert-sugar; and also those firms who have kindly sent the various samples of invert-sugar which are on the table.
Discussion
The President expressed the indebtedness of those present to Mr. Heron for his able and interesting paper. Owing to his long experience in the manufacture of invert-sugar, the author was able to give them a very complete account of nil the processes adopted—such information, in fact, as they were unable to obtain from text books. With regard to the decomposition products of levulose which -were usually designated as “other carbohydrates,” he believed that it was to these substances that the luscious taste and additional fulness which characterised beers brewed from invert-sugar was due. In this respect, he thought it probable that the lower grade invert-sugars No. 2 and No. 3 would be superior to No. 1, especially when colour was not of great importance. The presence of these substances in invert-sugar prepared by the action of acid made the latter product preferable to that prepared by the action of yeast, which was quite free from them, and certainly did not give the same flavor and fulness. Mr. Heron mentioned that the inversion of the sugar was controlled by the polarimetcr. The author had given the maximum rotation as 15 divisions of the Laurent sugar scale. He (the speaker) took it that allowance bad been made in this value for the fact that invert-sugar, as manufactured, always contains an excess of dextrose. He should like to have some information as to the quantity of acid used for inversion. He believed that, as a rule, something like 1 to 2 lbs. per cwt. was used. He gathered, however, from the paper that the author had devised a method by means of which he was able to find the exact quantity of acid required for the inversion of raw cane-sugar. Mr. Heron had stated that it was wrong to judge from the amount of ash found in a sample of invert-sugar, whether it had been made from a high class sugar or otherwise. He (the speaker) thought, however, that it would be admitted that a high percentage of ash in a sample of invert-sugar was an indication of bad quality. He also thought that the percentage of phosphates and of potash gave some idea as to whether beetroot-sugar had been employed in the manufacture of the sample.
Mr. T. May Smith, Jun., wrote regretting his absence. He wished to ask the following question:—Was the author acquainted with any method of proving if a finished beer had been primed with sugar, more particularly with “invert-sugar”? His own idea was that invert-sugar was going out of use, and he thought that the use of American glucose, and the change in the taste of beer drinkers had much to do with it.
Mr. J. Grant said he should be glad if Mr. Heron would answer the following questions:—1. “What was the strength of sulphuric acid employed in the inversion? 2. What other secondary substances besides levulinic acid were formed during the inversion, and what were their characters? 3. Did any oxidation occur by the use of animal charcoal, and if so what was the source of the oxygen? 4. Was not the composition of the ash of invert-sugar—especially the percentage of potassium salts and phosphoric acid—of value in judging the sources of the raw material used in its manufacture?
Mr. Hyde wished to know if Nos. 2 and 3 qualities of invert-sugar, the analyses of which were before them on the black board, were free from nitrogenous compounds. He also desired to know if such a high degree of refining as had been carried out with No. 1 quality was of any advantage to the brewer.
Mr. Heron, in reply, thanked the members of the North of England Institute, for the kind way in which they had received his paper. Dr. Hiepe was quite right in supposing that in the polarisations given, due account had been taken of the fact that in all commercial samples of invert-sugar there was an excess of dextrose, or, to speak more correctly, a deficiency of levulose, owing to the fact that the latter underwent further hydration and caramelisation so very readily during the process of manufacture. The quantity of acid used in manufacturing process varied from about 1 to 6 per cent, of acid calculated as anhydrous, H2SO4,; the quantity of acid employed would depend entirely upon the quality of the raw sugar. It certainly was not correct to say that because a sample of invert-sugar contained a high percentage of ash, it bad been manufactured from a low class sugar or syrup, for, as would be readily seen, one lot might be manufactured from a really first class sugar, and yet contain a high percentage of ash, whilst another lot originating from a low class syrup might turn out a very respectable invert-sugar. The question of ash depended to a very large extent upon the care bestowed upon it as well as the relative amount of char, &c, employed in the process of refining; as a matter of fact, only an expert in the manufacture of sugar could tell with any degree of certainty as to the quality of cane-sugar employed. Speaking generally, it would be quite right to condemn a sample of invert which showed a high percentage of ash. As regards beetroot-sugar, with that he had nothing to do, as his paper only treated of the manufacture of invert-sugar from cane-sugar. He was aware that beetroot-sugar was employed by some manufacturers. He, himself, did not approve of its use, but there were certain tests which in the hands of an export would show most conclusively if beetroot-sugar had been employed in the manufacture. In reply to Mr. May Smith’s query, he was not aware of any means whereby it could be proved that a finished beer had been primed with invert-sugar. He did not at all think that invert-sugar was going out of use; far from it, he knew for a fact that as much invert-sugar was at the present time being used in brewing as ever there was, probably more; whatever else American glucose was replacing, he did not think it was invert-sugar, indeed he did not see very well how it could, the class of beers brewed with invert-sugar was so widely different from that brewed with glucose, and he would say, of so superior a character all round that he did not see very well how a brewer accustomed to use invert-sugar could very well replace it with glucose. In answer to Mr. Grant’s queries, the sulphuric acid used for inversion would not be used in the concentrated form, but would be considerably diluted with water, generally from one-third to one-fourth. With regard to the organic bodies other than invert and cane-sugar present in a finished sample, he could not say much at present. He had done a lot of work on this subject, but want of time had prevented him completing his investigations, suffice it to say that the residue left after completely fermenting1 away the invert and cane-sugar, and purifying by means of alcohol, &c, possessed absolutely no optical activity, and a cupric reducing power of about 30. He did not think any oxidation occurred by the use of animal charcoal; he had made very many experiments on this subject with the object of ascertaining what the decolorising power of animal charcoal was due to, and he had convinced himself that contrary to the generally accepted theory it certainly was not duo to oxygen in the char. The composition of the ash of invert-sugar was of value in judging the sources of the raw material used in its manufacture. Replying to Mr. Hyde, Mr. Heron said that probably Nos. 2 and 3 might contain very small amounts of nitrogenous compounds, but he was not aware that they indicated much. (See Part II) The only advantage derivable from a high degree of refining, which meant less colour, was that the use of such an invert-sugar enabled a brewer to use in the brewing of his pale ales a more highly kilned, and consequently a sounder, malt than could be done with a lower quality sugar.