MEETING HELD FRIDAY, 15th MAY, 1896, AT THE CRITERION RESTAURANT.
Mr. Arthur C. Tanqueray (President), in the Chair.
The following paper was read and discussed:—
INVERT – SUGAR Part II
by John Heron, B.E.. F.I.C., F.C.S.
In a former paper on this subject (INVERT-SUGAR Part I) I referred very briefly at the close to the analysis of commercial invert-sugar, but time did not then permit me to go very fully into so important a matter, and, as a paper on such a subject as the one referred to could not be considered as complete without entering fully into the question of analysis, I thought the best thing I could do would be to read another paper on invert-sugar dealing exclusively with this question. It may perhaps be considered by some members of this Institute that a paper on such a subject is unnecessary and uncalled for, considering that the subject has already been treated of in many text-books, and that methods have been in use for many years in the laboratories of brewers’ analysts which are supposed to be fairly correct. However, if I can prove that such methods are more or less incorrect and unreliable, I consider that I shall be fully justified in bringing such a subject forward for further consideration, and I cannot introduce the subject better than by referring to a matter which came under my notice a few years ago.
A parcel of invert-sugar, No. 3 quality, was sent by a certain manufacturing firm to a brewery where at the time the brewer in charge happened to be in trouble with his beers, and, as is often the case at such times, samples of the various brewing materials are sent to the brewers’ analyst for examination, and, amongst other materials, a sample of this particular lot of invert-sugar was sent; the report came back that the sample in question won made from a low class beet-root syrup, and was unfit for brewing purposes. How the analyst in question arrived at such a conclusion I am still at a loss to understand. Samples of the same parcel were then sent to no less than four other brewers’ analysts for examination with the following results:—

No. 1 reported a good sample of No. 3 quality.
No. 2 reported a low class sugar only suitable for running beers.
No. 3, inferior sample of invert-sugar, should be used only for quick-running beers and common stouts.
No. 4 was analysed by the writer, and as a matter of fact the sample in question was made from partially refined Jaggery syrup from which the first crystals had been extracted for the manufacture of No. 1 quality.
Now, very little consideration will lead one to the conclusion that the differences observable in these analyses were due to either varying methods of analysis being used, or to certain errors of experiment which may have crept in somewhere, or to n combination of the two. These points will be referred to later on. It is with the object of bringing under your notice a method which I have every reason to believe is more accurate, and at the same time more rapid than any one at present in use, and by the practice of which such a deplorable state of things as depicted in the preceding table may be obviated, that I have decided to read a paper on such a subject.
In the analysis of a sample of commercial invert-sugar the following determinations are usually made:—
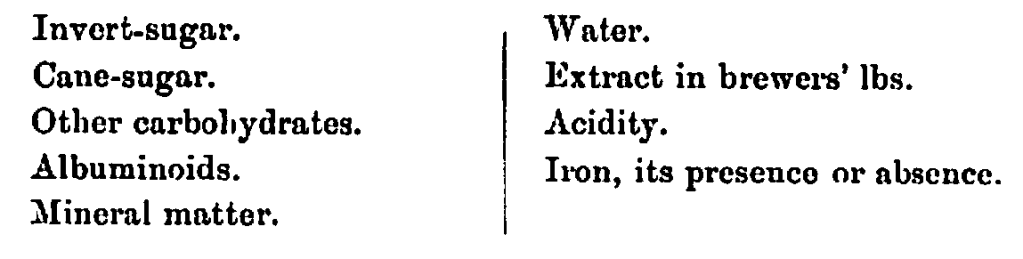
(a.) The Estimation of Invert-sugar.
At first sight the estimation of a reducing sugar by means of Fehling’s solution looks a comparatively simple and easy operation. An experience of over 20 years has convinced me that the correct estimation of a reducing sugar by means of Fehling’s solution is an exceedingly delicate, as well as difficult, operation, and in which proficiency or accuracy can only be obtained after a long apprenticeship.
The methods of estimation are generally determined nowadays gravimetrically either by weighing the copper reduced as cupric oxide (CuO) or as metallic copper.
As it was to me a matter of much importance to ascertain the most accurate and reliable method for the estimation of reducing sugars, 1 have given much time and attention to this matter, and, after a careful and critical examination of every method that has been introduced up to the present for the determination of reducing sugars by means of the copper test, I have come to the conclusion that the most accurate and reliable of any is the gravimetric method described by O’Sullivan in 1876 (J. Chem. Soc, 1876, 2, 130), provided that the conditions of experiment be strictly adhered to. Working under the conditions laid down by him, O’Sullivan found that 1 gram of dextrose reduces 2205 grams of cupric oxide (J. Chem. Soc, 1876,1, 479).
Later on Soxhlet found that varying results can be obtained with the same sugar solution, according to the state of dilution of the Fehling’s solution. This is now fully confirmed by all chemists who have given any attention to the subject, hence, before starting on the analysis of reducing sugars, each analyst must first determine for himself under what conditions he is going to conduct his experiments: whether his Fehling’s solution, after the addition of the sugar solution, is to show a dilution with one, two, or three volumes of water. For example, O’Sullivan dilutes his Fehling solution with two volumes of water to one of Fehling solution; under these conditions ho finds that 1 gram of dextrose reduces 2·205 grams of cupric oxide. I find it more convenient to use only one volume of water to one volume of Fehling’s solution; under these conditions I find that 1 gram of dextrose instead of reducing 2·205 grams of cupric oxide, reduces 2·26 grams of cupric oxide. Dextrose being the type of reducing bodies, and that carbohydrate, for which the amount of cupric oxide reduced was first determined, O’Sullivan takes this body as the standard to which to refer all other reducing carbohydrates or mixtures of reducing with non-reducing ones. Therefore, according to him, the cupric oxide reducing power of a body is the amount of cupric oxide calculated as dextrose which 100 parts reduce. This power, then, for dextrose would be 100, and, for shortness, is written K = 100, and if an analyst, on calculating out results of his experiments working with a solution of pure, dry dextrose, does not obtain this value for dextrose, he may very safely conclude there is an error somewhere in his method of working which has got to be set right.
Working on similar lines, both C. O’Sullivan and J. O’Sullivan have found that pure invert-sugar possesses the same value for K, namely 100; this I have fully confirmed over and over again. I am aware that Herzfeld and some other continental workers state that the reducing power of invert-sugar is somewhat lower than that of dextrose, and, following them, Moritz and Morris give the value of K for invert-sugar as 96·6. There can be no doubt, however, but that the cupric reducing powers of invert-sugar, levulose and dextrose are all alike, namely, 100.
One would think that in these days it was hardly necessary to denounce the volumetric method of estimating reducing sugars with Fehling’s solution as being most unsatisfactory and unreliable; indeed, one would suppose that any analyst who knew his business would never dream of using such a process, and yet, in a text book of quantitative analysis, the third edition of which has only recently been published, the old primeval method for the analysis of sugar, which was practised in the good old days of long ago, before polarimeters were used, is still given, and, amongst other obsolete processes, our old friend, the volumetric method, crops up, along with the digestion of the cane-sugar, with excess of acid under pressure in a corked and wired flask, for half an hour, at temperature of boiling water, so as to ensure complete inversion of the cane-sugar, or rather, I should say, destruction.
In order to obtain accurate results with Fehling’s solution great care must be bestowed upon its preparation.
It is most important that the sulphate of copper should be as pure as it is possible to obtain it. I have found it is impossible to buy sulphate of copper in a state of sufficient purity for this purpose, so that one of two things must be done, either to prepare the pure salt from pure metallic copper, such as copper deposited electrolytically, or to purify the commercial salt; in the latter case commercially pure sulphate of copper is dissolved in distilled water, to which a few drops of pure sulphuric acid have been added, filtered bright, then well boiled, concentrated, and allowed to crystallise; the crystals so obtained are well drained and re-crystallised twice after this from distilled water, the last time the solution being kept constantly agitated with a glass rod so as to obtain the crystals in a state of fine powder. The Rochelle salt can be obtained commercially pure enough for the purpose.
The sodium hydrate, I prefer, is that known commercially as “pure by alcohol.”
I prepare Fehling’s solution as follows:—
Thirty-five grams of pure sulphate of copper are weighed out and dissolved in 500 c.c. of distilled water; as soon as completely dissolved the solution is quickly raised to boiling, and boiled for about 10 minutes, during which time a green, insoluble, basic sulphate of copper is thrown down, the solution is filtered from this, cooled down to 60° F., made up to 500 c.c., and put away for use.
One hundred and seventy-three grams of Rochelle salt are next dissolved in about 200 c.c. of distilled water, and filtered if necessary.
Sixty grams of sodium hydrate purified by alcohol are dissolved in about 150 c.c. of distilled water, and as it is impossible to obtain any sodium hydrate absolutely free from iron, I find it the best plan to allow the sodium hydrate solution to stand for a few days by itself, during which time most if not all the iron, which at best is only present in a very small quantity as an impurity, settles out; the solution is then filtered through n clean asbestos filter by means of the air pump, then added to the solution of Rochelle salt, made up to 500 c.c. with water, and bottled up for use.
I have no doubt that in many instances where errors have occurred, errors which are generally on the plus side, many, if not most of them, are due to the use of more or less impure chemicals, or to want of attention to details in the preparation of the Felhing’s solution.
Having, then, determined the necessary conditions of experiment, we are now in a position to estimate the amount of invert-sugar present in any given solution; I find the following steps in the process must be rigidly adhered to.
The quantity of solution taken must be weighed out carefully in a tared beaker, and its true volume afterwards determined by multiplying by 1,000 and dividing by the sp. gr. of the solution.
The water in the water-bath must be boiling vigorously, as well as the water in the wash-bottle, which must have been previously well boiled so as to expel any atmospheric air.
The digestion with Fehling’s solution must not be carried on longer than 12 minutes, the solution must be filtered as quickly as possible, and as soon as the last drop of the Fehling’s solution has been poured on the filter, the filter must be kept constantly washed with boiling water until the blue colour has completely disappeared; the rest of the precipitate of cuprous oxide is now transferred to the filter, and the washing carried on until the wash water ceases to give any blue reaction with delicate litmus solution. I find that, do what one will, a very small portion of some of the constituents of the Fehling’s solution are persistently retained by the filter paper, be that it is always advisable when starting with a fresh lot of filter papers to make some blank experiments so as to ascertain what increase in weight the filler ash gives; I find this generally amounts to about 2 milligrams, a correction for which must accordingly be made. There is no necessity whatever to treat the cupric oxide with nitric acid in the crucible; if the cuprous oxide has been properly detached in the first instance from the filter paper, and the filter paper burnt by itself ns should always be done by every careful analyst, simply heating over a Bunsen burner in a porcelain crucible with the lid off, for 12 to 15 minutes, is quite sufficient to completely convert all the cuprous into cupric oxide. It has amused me very much, on looking over the large number of papers which have been published in the Continental journals during the last two or three years on this subject, to see the various arguments for and against the method I have advocated, the straining after the poor, miserable gnat, and the enormous number of camels that have been swallowed in doing so. It would take far too much of my time and weary your patience to enter fully into a discussion of all the arguments which have been advanced, suffice to say that if the instructions laid down by O’Sullivan, be strictly adhered to, and that each analyst determines what his own personal factor of manipulation is, and takes this into account, I say most emphatically no method is so accurate or so reliable as the one I have mentioned.
(b.) The Estimation of Cane-sugar.
In the case of raw and refined sugars, where only comparatively small proportions of invert-sugar are present, the method usually adopted for the estimation of cane-sugar is the one introduced by Clerget several years ago, and known as the double polarisation method, and which has since been slightly modified and rendered more correct; but where the proportion of invert-sugar is large, and more especially where the amount of cane-sugar is very small, as in the case of commercial invert-sugar, where it rarely exceeds 5 per cent., no accurate estimation of the cane-sugar can possibly be made by this method, for it has been found that mineral acids, hydrochloric and sulphuric (Jungfleisch and Grimbert, Comptes rendus, 108,144), exercise a peculiar modifying influence upon the optical activity of levulose: for instance, the addition of a small quantity of hydrochloric or sulphuric acid to a solution of pure levulose at ordinary temperatures instantly increases its optical activity, which action becomes intensified on heating the solution, wherefore, in the analysis of sugars containing an appreciable amount of invert-sugar, such as syrups, molasses, invert-sugar, and such like, Clerget’s method of double polarisation cannot be applied, since the addition of the acid necessary to bring about the inversion of the cane-sugar present increases the levo-rotation of the invert sugar already present in the sample, so that the polarisation after inversion would apparently show more cane-sugar to be present than there really was. It is therefore best to proceed as follows :—
Determine the total amount of reducing sugar after inversion by means of Felhing’s solution gravimetrically, from this subtract the amount of reducing sugar similarly determined before inversion, multiply the remainder by 0·95; the result will be the amount of cane-sugar present in the solution.
In the inversion of the cane-sugar by means of acid, care must be taken that too much acid is not used, and also that the proper temperature be observed. In all the text books in which methods for the analysis of sugars are given, a much larger quantity of acid than is really required is always recommended. Now, when we consider that 0·5 c.c. of pure concentrated hydrochloric acid is amply sufficient to completely invert 1 gram of cane-sugar, and that in a 10 gram per 100 c.c. solution of invert-sugar we rarely have more than 0·5 gram of cane-sugar, more generally 0·2 gram, or even less, 0·5 c.c. of acid is quite enough.
For all practical purposes I find the following method of procedure answers excellently:—100 c.c. of a 10 gram per 100 c.c. of invert-sugar solution is taken in a flask having two marks on the neck, 100 c.c. and 110 c.c.; to this 0·5 c.c. of pure concentrated hydrochloric acid (sp. gr. 1150) is added; the neck of the flask is then twirled rapidly between the fingers so as to mix as thoroughly as possible the acid with the sugar solution; a small thermometer is next placed in the flask, which is then plunged in a water bath at a temperature of 70° C. In about five minutes the temperature of the solution in the flask will have risen to 68° C, at which temperature it is maintained for five minutes longer (during which time the cane-sugar present will be found to have been completely inverted), taken out, cooled down to 15·5° 0. (00° F.), the thermometer taken out, and in doing so washed with a little water into the flask, the volume made up with water to the 110 mark, and well shaken up. The total invert-sugar is then determined gravimetrically with Fehling’s solution, about 1 c.c. being taken for the purpose, and the result multiplied by 11/10 to correct for the dilution.
(c) The Estimation of the other Carbohydrates.
These bodies, to which the very inappropriate terms “inert bodies.” “inert carbohydrates,” “intermediate carbohydrates,” have been variously applied, cannot very well be determined directly, and are usually estimated by difference. The way in which this difference is generally arrived at is as follows :—
The specific gravity of the solution of invert-sugar is taken at 60o F.; the number so obtained, less 1000, divided by 3·86 is said to give the amount of solid matter in solution, from which the percentage of solid matter in the sample under examination is arrived at.
The sum of the percentages of invert-sugar, cane-sugar, ash, and albuminoids is subtracted from the percentage of the solid matter, and the difference is put down as so much “inert bodies.”
Now, one very important fact is overlooked in such a proceeding, and that is that the specific gravity of sugar ash in solution differs very considerably from the specific gravity of the sugar itself, so that in samples of low quality sugar where the ash is comparatively high, a serious error may creep into the analysis.
As the result of a very large number of determinations of the specific gravity of the ash in solution of invert-sugar, made from sugars of widely different sources, I find that one gram of ash dissolved in water and made up to 100 c.c. at 60° F., gives a sp. gr. of 1008. On the other hand, it is now a well established fact that a 10 gram per 100 c.c. solution of pure sugar gives a sp. gr. of 1038·6, so that a 1 gram per 100 c.c. of such a solution would show approximately a sp. gr. of 1003·86, therefore, in order to obtain the true amount of organic solid matter or of carbohydrates in a solution of commercial sugar we must first determine the amount of ash present, then correct the specific gravity of the solution for the gravity due to ash before we divide by the factor 3·86; then, having divided the corrected specific gravity less 1000 by this factor, we obtain the true amount of total carbohydrates in solution, from which, if we subtract the sum of the invert- and cane-sugars, we obtain the real amount of the other carbohydrates which may be present.
1 say other carbohydrates, for those bodies other than cane- and invert-sugar which are present to a greater or less extent in most samples of commercial invert-sugars are in every sense true carbohydrate bodies. I have made a careful study of some of the properties of thesis bodies, but several circumstances have occurred which have prevented me completing my observations, the result of which I hope to have the pleasure of laying before the Institute in the near future. Suffice for the present to say that this body, for I believe only one exists, possesses no specific rotatory power, but has some action on Fehling’s solution, showing a value for K of about 30.
Estimation of the Albuminoids.
This determination shows very little, and although I at one time estimated the amount present by Kjeldahl’s method, of late years I have given up doing so. In all properly made samples of saccharum the nitrogen is exceedingly low or absent altogether; of course in a notoriously bad sample it may be thought useful by some analysts to determine the nitrogen, but I have never found its indications of much value.
Estimation of Mineral Matter.
The mineral matter or ash is best and most speedily estimated as follows:—5 grains of, the sample are weighed into an accurately tared platinum dish of about 100 c.c. capacity, then gently heated for a few minutes over the water bath until the thick syrup or solid mass melts, then about 20 drops of concentrated sulphuric acid (sp. gr. 1600) added. Immediately the acid is added the syrup swells up into a porous carbonaceous mass, which is then ignited over a Bunsen flame,
gently at first and strongly afterwards, until only a while or grayish-white dry powder is left. The dish and contents are then transferred to a desiccator, allowed to cool, and weighed; the dish is next washed clean of the ash, heated gently, cooled, and again weighed, the difference between the two being the weight of the sulphated ash.
The addition of sulphuric acid converts the various mineral bases into sulphates. When this process was first introduced, the sulphated ash was considered to weigh heavier than the ash obtained in the ordinary process of carbonisation, so that it was, and is, still usual to deduct 10 per cent, of its weight from the sulphated ash in order to obtain a more correct result. This may apply, perhaps, to beet-sugars, but it certainly does not to cane-sugars, and from an extensive, series of experiments which I have made on this point, I have come to the conclusion that much more accurate results are obtained by taking the weight of the sulphated ash, without any deduction, as representing the true percentage of mineral salts in the sample under examination; recently the results of other chemists fully confirm this view.
Estimation of Water.
The amount of water present cannot be determined in the ordinary way, namely, by drying in the hot-air bath or water oven, and weighing from time to time until the substance ceases to lose weight, owing to the fact that when any substance containing levulose is heated, for even only a short time, much above 140° F, the levulose is very liable to undergo decomposition, and hence the results will invariably be inaccurate; the water is, therefore, best and most accurately determined by difference; thus the percentages of invert-sugar, cane-sugar, other carbohydrates and ash having been determined, the sum of those subtracted from 100 gives the water.
The following example will serve to illustrate the method of analysis as discussed in the previous pages.
Analysis of Sample of No. 3 Quality Invert-sugar.
Twenty-five grams were weighed out in a carefully tared beaker, dissolved in boiling water, transferred to 250-c.c. flask, beaker washed out with distilled water two or three times, and washings added to solution in flask, which was then cooled down to 60° F., and made up to containing mark, and the flask well shaken, and the usual determinations being made, the following results were obtained:—
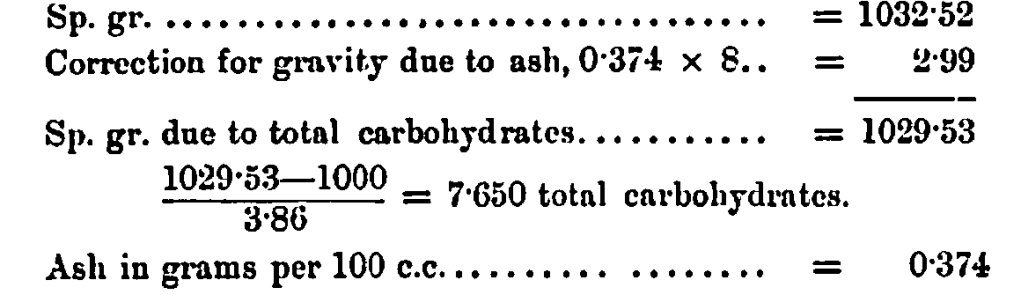

adding the invert-sugar and cane-sugar together, and subtracting from the total carbohydrates as found from sp. gr. corrected for ash, thus—7·65 — (7·229 + 0·154) = 0·267 gram per 100 c.c. other carbohydrates.
I might here draw attention to a very useful check upon the invert and cane-sugar estimations which is afforded by this proceeding, for it will be at once seen that, if the sum of the invert- and cane-sugar exceeds the amount of the total carbohydrates deduced from the sp. gr. some error must have crept in, by means of which a larger redaction is shown for the Fehling solution than could possibly have taken place were the assay correctly performed.
The analysis so far stands thus—

adding all these together, and subtracting the sum from 10, we arrive at the moisture, thus 10 — 8·024 = 1·976 grams per 100 c.c. water; multiplying each of the above results by 10 we at once get the composition in percentages.
Let us now refer back to the different analyses of the same sample of invert given on previously and apply the check I have just mentioned to these analyses, and see what we can learn from it.
We can easily calculate what the specific gravity of the solution which was used for analysis was from the extracts given. Hence the total carbohydrates in grams per 100 c.c. amount to 7·6217, but the sum of the invert-sugar and cane-sugar amount, to 8·045, which shows 3·98 per cent, more sugar than really exists in the sample, so that some error was introduced in the sugar determination by means of Fehling’s solution. In Analysis No. II a similar error to the extent of 1·72 per cent, occurs, and in No. III to the extent of 2·10.
I have no doubt whatever but that the differences observed in these analyses can be accounted for by the different methods of manipulation of the Fehling’s solution pursued by the different analyses, and I think tends to show how extremely desirable it is to have some kind of check such as the one proposed, whereby greater uniformity in the results of analyses of invert-sugar may be obtained.
Discussion.
The President said brewers were much indebted to Mr. Heron for placing the details of his methods so clearly and practically before them, and when they looked at the great difference between the four analyses given, it was evident that the sooner some standard analysis, which would yield uniform results, was fixed upon the better. He was very much interested in the method Mr. Heron had proposed of estimating the “other carbohydrates,” and he was glad to hear that the author intended to give a further communication on the same subject at a future meeting. It was a subject of great importance to brewers, and one of which they knew very little.
Mr. J. L. Baker said he did not quite understand why Mr. Heron determined the moisture in invert-sugar syrups by difference when there were ready means at hand of directly estimating it. The syrup might, for example, be mixed with kieselguhr and dried in a water oven at a temperature of 98° or 100° C. He should also like to know why the author added sulphuric acid to the solution of copper sulphate in the process of purifying that salt by re-crystallisation.
Dr. G. Harris Morris inquired whether the value, 1008, given by the author as the specific gravity of a solution of 1 gram per 100 c.c. of ash was determined by direct experiment, and, if so, what allowance was made for the insoluble portion of the ash. The author had given no experiments on the reducing power of invert-sugar in support of his statement that it was the same as dextrose, but he (Dr. Morris) was bound to say that the consensus of opinion was decidedly opposed to that view. In the recently published work of Kjeldahl, in which the question of reducing power was gone into more thoroughly than had over been done previously, the reducing power of invert-sugar was shown to be distinctly less than that of dextrose. He could not recall any work of C. O’Sullivan in which mention was made of the reducing power of invert-sugar. But with regard to James O’Sullivan’s work, his recollection was that the values ho obtained in his determinations did not altogether support the view he took as regards dextrose and invert-sugar. Mr. Horace T. Brown and himself had just completed a very exhaustive examination of the reducing powers of different sugars, and they had certainly found a difference between levulose and dextrose.
Mr. Daniell asked if it were a fact that in the manufacture of invert-sugar a small amount of glucose from starch was added, in order that the product might set rapidly. If that were so, it would be well to know the percentage of glucose added, since it was evident that at present prices the more glucose a manufacturer could add without its being detected the more it would be to his profit.
Dr. Morris said ho should like to supplement his remarks. Mr. Heron had apparently not used the polarimeter, but had taken the whole of his initial reduction as due to invert-sugar. The point raised by Mr. Daniell brought to his mind the absolute necessity of determining the opticity, as well as the reducing power, of commercial invert-sugars, and deducing from these two values the relative proportions of dextrose and levulose. In some samples delivered as invert-sugar he had found as much as 60—70 per cent, of added glucose, which would not be detected unless the polariscope were used.
Dr. W. J. Sykes called attention to a not generally known method for estimating levulose, which was effected by taking advantage of the difference in the rotatory power of a solution of this sugar at a high and low temperature. The method had been recently applied by Wiley (J. Amer. Chem. Soc, 1896,18, 81; see this Journal, 1896, 2, 293), to the estimation of levulose in honey.
Mr. Heron, in reply, said he preferred to determine the moisture in the way he had described. It was impossible to heat levulose above 140° F., either in the solid state or in solution, without its undergoing decomposition, and the longer the duration of heating, and the higher the temperature, the more decomposition occurred.
Mr. Arthur R. Ling said the moisture could be got rid of below 70° C. if the sample were mixed with alcohol and sand.
Mr. Heron said that method was not reliable at all, especially if a large quantity of levulose were present, as was the case with invert-sugar. Where there was only 2 or 3 per cent, of invert-sugar the approximate percentage of moisture present could be estimated in that way, but where it was more than this it could not. He added sulphuric acid to the copper sulphate because he found that most copper sulphate contained small traces of nitric acid and other impurities, and the addition of sulphuric acid certainly exercised a considerable purifying effect on the salt. The specific gravity of the ash solution was determined by direct experiment with the ash of sugar from various sources, and from the published results of the specific gravities of the salts of which it was composed; the average came out as close as possible to 1008. With regard to the cupric reducing power of levulose, it had certainly been taken as identical with that of dextrose by Cornelius O’Sullivan, and James O’Sullivau had definitely stated that the reducing power of levulose was K = 100. He had determined the reducing power of invert-sugar in a variety of ways, and had always found it to give an average value of K = 100. He had no experience of adding glucose to invert-sugar, either for the purpose of graining or for adulteration, and any manufacturer who did so would be acting dishonestly. In many cases brewers demanded solid invert-sugar, and in order to expedite solidification a substance known as “graining” was sometimes added; this substance consisted of invert-sugar which had previously solidified. With regard to the optical investigation, he had referred to that in the first part of the paper, read before the North of England Institute of Brewing. He had there stated the rotatory power which ought to be exhibited by a sample of invert-sugar properly made. Dr. Morris was mistaken, however, if he imagined that the actual amount of dextrose and levulose in a sample of commercial invert-sugar could be determined by the polariscope, for during the process of inversion a certain amount of levulose was caramelised, and the true polariscopic value would not be observed; the purest sample that could be prepared on a commercial scale would never show the rotatory power of pure invert-sugar as prepared in the laboratory.
Dr. Morris said that was not quite his point; the fact was that when the opticity was determined in addition to the reducing power, some indication was obtained as to whether glucose had been added.
Mr. Heron agreed, and said the polarimeter was most useful in the hands of an experienced operator in detecting the addition of glucose. The mere fact that a dextro-rotation was obtained was not sufficient, however, to prove the presence of added glucose, as this dextro-rotation might be occasioned by the presence of cane-sugar, due to under-inversion. Anyone interested in the matter would find papers on the subject by Casamajor in the Chemical News (see that Journal, 41,221; 42,326; 44,265).
The President then proposed a vole of thanks to Mr. Heron for his paper, which was carried unanimously.