Journal of the Institute of Brewing – August 1922
INVERT SUGAR
Invert sugar, as is generally known, owes its name to the fact—first observed by Biot in 1836—that a solution of it rotates a beam of polarised light to the left, i.e., in an opposite direction from that observed in the case of its parent substance, cane sugar. This behavior is due to its consisting of a mixture in equal proportions of two sugars —dextrose and lævulose—the latter of which rotates polarised light more strongly to the left than does dextrose to the right, the resulting mixture having thus a left-handed rotation.
Invert sugar occurs widely in nature—in honey, molasses and many fruits, while immense quantities are made artificially from cane sugar for the use of brewers. The inversion of sucrose (as the chemist calls cane sugar) on the manufacturing scale is a flourishing industry, and, although it is, of course, quite possible to invert sugar in the brewery by the use of yeast (F. W. Thompson, Eng. Pat. 8686, 1884) most brewers prefer to buy the manufactured article. Commercial invert sugar is never met with free from water but it readily sets hard, especially if of high grade quality and of low ash content; it usually contains about 18 to 20 per cent, of water whether in the syrupy or solid state. When syrupy invert sugar sets, it usually does so in a homogeneous mass, but under some conditions the dextrose first separates, since this sugar is more readily crystallisable. A sample which was kept a year and had separated into a clear syrup at the top of the jar and a solid mass below showed, on analysis by the writer, the following composition:—
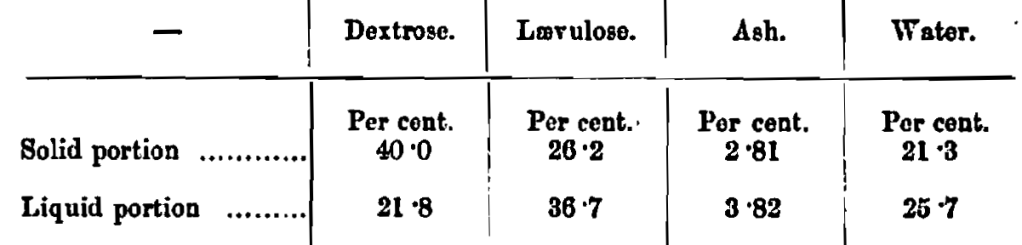
It will be seen that although considerable fractionation has occurred, the separation is still by no means sharply defined.
Honey, which is practically a mixture of invert sugar and water, shows a similar behaviour, in fact, many bee-keepers believe that a capacity to set is a mark of a genuine unadulterated honey. Honey will sometimes crystallise to a certain extent in the comb, with the result that the run honey is richer in lævulose than in dextrose, although even freshly run genuine honey nearly always in the writer’s experience shows when carefully analysed, a slight but constant lævulose percentage a trifle in excess of the dextrose value—a feature never mot with in a commercially prepared invert sugar. The inversion of the cane sugar collected by bees from the nectar of flowers is brought about by an invertase secreted by the bee.
The inversion of cane sugar may be brought about by two separate agencies, both of which result in the formation in equal proportions of the same products—dextrose and lævulose. These two agents are the enzyme invertase and acids. Commercial invert sugar as sold to brewers is made by the use of mineral acids—hydrochloric or sulphuric —though a few brewers employ the invertase present in yeast for the inversion of cane sugar in the brewery (see this Journ., 1902, 8, 270).
If this last method is employed, it is usual to boil the mixture of inverted sugar and yeast in the copper, and opinions differ as to the advisability of having boiled yeast in the wort. Thus Heron (this Journ., 1896, 2, 478) considers such a practice “dangerous and very objectionable,” while some brewers deliberately boil a little yeast in their coppers in order to augment the supply of nitrogenous yeast food for their pitching yeast. The action of yeast on cane sugar is twofold; it both inverts the sucrose by virtue of its invertase and ferments the invert so produced by means of the zymase present in the yeast cells. Under ordinary conditions of fermentation both reactions occur simultaneously, but if yeast be added to cane sugar at a temperature too high to admit of fermentation (such as 135° F.) inversion alone will take place, while even at ordinary air temperature yeast will more slowly invert cane sugar without fermenting it if some substance which inhibits fermentation, such as chloroform or toluene, is present. It should be borne in mind that yeast is unable to ferment cane sugar without first inverting it, and this inversion the yeast can readily bring about by means of its invertase, which hydrolyses the cane sugar with the formation of dextrose and lævulose. This latter sugar as shown by Hiepe (this Journ., 1895, 1, 288) disappears somewhat more slowly than dextrose when yeast ferments invert sugar.
Since yeast can thus readily invert cane sugar while fermenting it, it may be asked why the brewer who uses sugar as part of his grist does not use cane sugar itself, and thus save the difference in price between the two materials.
Several more or less plausible answers may be given to such a question. The one most often given perhaps is an expression of the belief that in asking the yeast both to invert and ferment cane sugar too great a demand is being made on the yeast’s activities, with a resulting weakening of its fermenting power (see Heron, this Journ., 1896, 2, 477; also Hiepe, loc. cit., p. 307). A writer who has recently dealt with the question of the presence of cane sugar in brewing sugars (Brew. Trade Rev., 1922, 36, 168) alludes to this belief as a tradition unsupported by real scientific evidence, and gives various reasons for thinking it is exaggerated. It should be remembered that, after all, the two processes, inversion and fermentation, are mediated by quite distinct enzymes, and it is unlikely that the action of one of them would result in a weakening effect on the other. Certain it is, however, as shown by Baker and Hulton (this Journ., 1914,20, 241) that even when added as priming to finished beer both cane and invert sugar disappear at the same rate, and there is no evidence that the fermentation of the cane sugar lags behind that of invert under these conditions. A far more probable explanation of the dislike of brewers for raw cane sugar used as priming is the fact that its use is occasionally accompanied with the appearance of ropiness in the beer, and there is evidence that this is sometimes the case. Cane sugar is occasionally infested with leuconostoc and other organisms, and if inverted with acid at a high temperature will by such treatment be sterilised. A further point is sometimes raised as to the difference in flavours, but it is doubtful whether any such distinction is recognisable in a finished beer after the cane or invert sugar—as the case may be—has fermented as it would be when such sugars are employed as part of the grist, although when used as priming the extra lusciousness and sweetness of invert sugar is of value. Even as regards the relative sweetening powers of cane sugar and invert sugar opinions have differed considerably. Thus Paul (this Journ., 1921, 27, 592) found that dextrose was only half as sweet as cane sugar, and lævulose sweeter than cane in the proportion of 103 to 100. This would make invert sugar only 75 per cent, as sweet as cane sugar, assuming that dextrose and lævulose each produced its proportionate sweetening effect when in presence of the other. Sale and Skinner, in some more recent work (this issue, p. 624), assign to invert sugar a value of 85 as against 100 for sucrose—a result fairly close to Paul’s findings. It must be remembered, however, that these workers were using absolutely pure invert sugar, whereas the brewer employs invert sugar of only commercial purity, containing ash, and the marked effect of the presence of salts in modifying the apparent sweetness of sugar is well known. It would be of interest if the sweetness of pure cane sugar were compared with such commercial invert, as this might disclose corroborative- evidence for the general belief held among brewers.
When a perfectly pure cane sugar is inverted with acid, the merest trace is sufficient, but if any ash is present, as is always the case with all but highly refined sugars, more acid is required, with the result that a certain amount of the lævulose formed is decomposed with the formation of unfermentable bodies, and for this reason commercial invert sugar, when analysed, never shows a percentage of dextrose quite as high as that of lævulose. Moreover, it is generally found that in such sugars a small amount—varying from 1 to 5 per cent.—of cane sugar remains un-inverted, and the presence of such cane sugar may be looked upon, the writer in the Brewing Trade Review, loc. cit., suggests, as evidence that the cane sugar employed was not of the highest grade, since the maker in an attempt to limit the decomposition of the lævulose and the production of colour and unfermentable substances will sometimes restrict the degree of inversion to a point which leaves a little cane sugar un-inverted. There is however, no objection whatever to the presence per se of this cane sugar, even in quantity, for, as is well known, malt itself contains some 5 per cent, of sucrose among the ready-formed sugars.