MEETING HELD FRIDAY, 29th NOVEMBER, 1895.
Mr. Harold Trinder in the Chair.
Nitrates in Brewing Waters
by R. E. Evans.
Having been asked to read a paper before you on very short notice, I have selected the above subject, as some time ago I did some work in this direction to find out, if possible, the exact influence which nitrates exerted. Many of the experiments which I tried then I have been able to repeat; but, unfortunately, I have not been able to make any trial brewings on a fairly large scale, and so have collected the experiences of others which I have become acquainted with from time to time.
The presence of nitrates in a water supply is undoubtedly due, in the first place, to the presence of organic matter, which, by decomposition, yields ammonia, this being, tinder certain circumstances, converted by a living organism into nitric acid which combines with any base that may be present. There seems to be some doubt whether the ammonia is directly oxidised to nitric acid, or whether nitrites are found in an intermediate stage. The latter seems the most probable, but cannot be absolutely proved, as, in certain cases, considerable quantities of nitric acid have been produced, yet during the process no trace of nitrites have been detected. The actual process in nature may be the conversion of ammoniacal salts to nitrites by one organism, while the further oxidation to nitrates is effected by another, and, of course, if a large excess of the latter wore present the nitrites would be converted to nitrates immediately on their formation, and so would not be detected.
Alkalis, such as sodium or potassium carbonates, very greatly check the formation of nitrates, whilst magnesium and calcium carbonates arc very favourable to their production, probably byneutralising the nitric acid as soon as it is formed. Gypsum is also said to be favourable to nitrification. It is these circumstances that render the proportion of nitrates of little value in determining the purity of a water, unless at the same time a complete analysis of the salts present be made. With waters containing over 15 grains per gallon of chalk and a little gypsum, the amount of nitric acid present may be taken as an indication of past contamination, but waters containing soda are almost sure to be free from it. As the nitric acid is derived from ammonia, which in turn must have been derived from some nitrogenous material, if it were not for this action of the salts, it would give very valuable indications of contamination occurring at some distant point, but which is oxidised and rendered harmless (in a sanitary sense) by the action of micro-organisms. A small quantity of nitric acid is found in the purest rain water; it is fairly constant, and is about 0·0424 grain per gallon of NO2. Its presence is duo to discharges of electricity in the air whereby the oxygen and nitrogen directly combine to form NO2, which is subsequently oxidised to nitric acid, and dissolved by the rain. This amount might, therefore, be looked for as constant in all waters were it not that vegetation has the power of removing it, and it is doubtless this fact that accounts for the cases in which no traces have been found in waters by the most careful testing.
Recently I have examined the results of the last 105 analyses of various waters from breweries in all parts of the country and analysed in our laboratory. As all these analyses have been made for the purpose of supplying hardening mixtures, when the waters have been deficient in the necessary salts, they are complete ones comprising the estimation of free and albumenoid ammonia, total soluble matter (organic and inorganic), chlorine, nitric, and sulphuric acids,and the bases, usually soda, magnesia, and lime, with qualitative tests for nitrites, iron, lead, and copper, and microscopical examination of deposit, if any. I have not included analyses mode for domestic purposes, for which such a complete examination is not usually performed. Of these 105 analyses, the average amount of nitric acid (reckoned as NO2) is 0·505 grain per gallon, or excluding one very high result showing 6·18 grains per gallon, the average is 0·45 grain per gallon.
Examining the results farther, 47·6 per cent, of the waters contain 0·2 grain and under, 32·4 per cent, contain from 0·2 to 1·0 grain, and 20 per cent, of the results show over 1 grain per gallon. The highest amount found is 6·18 grains per gallon, the next 2·2. Where the amount of nitric acid is not greater than about 1·2 grains there is usually found enough alkali, sodium or potassium, to account for its combination as sodium or potassium nitrates, but when the amount is greater than this, it is usual to find some of it combined as magnesium or even calcium nitrates.
On examining a few cases from those, containing the greatest amounts of nitrates, the conditions under which they are found are well borne out. This first is a water containing 0·81 grain per gallon of sodium nitrate, and it also contains 26·2 of chalk, and 6·9 calcium sulphate, in another case 1·74 grains per gallon of sodium nitrate are present with 18·8 chalk, but no gypsum. In both cases there is no excessive quantity of free or albumenoid ammonia. The third case is a curious one. The water contains sodium, magnesium, and calcium chlorides, the nitric acid is all in combination with calcium, and the other malts present are 2·82 grains of calcium sulphate (gypsum) and 22·01 grains of chalk; the ammonia both free and albumenoid is very low. I think this is the only case of this type of water we have met with, though we have many cases where the nitric acid is at least partially combined with magnesia.
In the case of the water containing the highest proportion of nitric acid, i.e., 6·18 grains per gallon, equal to 8·34 grains of sodium nitrate, the other salts present were calcium sulphate 1·75, calcium carbonate 13·05, and magnesium sulphate, 8·34 grains per gallon. I regard this as an exceptional case and, therefore, prefer not to reckon it when taking the average, as the water comes from a district where an enormous amount of sodium nitrate is every year added to the soil, and no doubt that a proportion is so far washed in by the rain that the plants are unable to remove it, hence the large amount of nitrates in the spring and well waters. This case shows the great importance of the analyst being informed as fully as possible of all details relating to a sample, so that a good and clear judgment of the analysis may be made.
It is rather curious to remark that in general the north country waters contain less nitrates than those from the south, as far as our experience goes samples coming from under a line drawn across the country about 14 miles north of Birmingham will on an average show a greater proportion of nitrates than those from above it.
Regarding nitrates as evidence of previous contamination a simple calculation will show that 1 grain per gallon of nitric acid (HN03) is produced by the oxidation of 0·27 grain per gallon of ammonia, or 0·38 part per 100,000, as it is more commonly expressed. This ammonia may either have existed as free, or “albumenoid,” both being convertible to nitric acid, the former directly, the latter in directly, through the agency of other organisms. An amount of nitric acid corresponding to 1 grain per gallon would therefore indicate a rather serious amount of contamination to have occurred, though at the same time this amount would have no influence on the brewing process.
It is not until the amount of nitric acid rises to about 2·0 grains per gallon that its effect is noticed. The general effect may be said to be a deeper colour, sluggish fermentations and peculiar flavours, but some brewers using nitrated waters complain of only one effect, and the others are not noticed, and there are yet others who, although they are employing waters with considerable amounts of sodium nitrate in solution, do not notice any effect at all.
As might be expected the influence of nitrates is much modified by the condition and character of the yeast. With a strong healthy yeast, the effect is small, whilst a weak yeast with the same water would give very bad results. The action seems something approaching a bad antiseptic, i.e., the development of yeast is checked whilst bacteria are not affected, this would of course account for the bad attenuations and flavours.
The colour complained of is often put down to the oxidation of the saccharine, or albumenoid bodies of the wort, but it is difficult to see how dilute solutions of a nitrate (a neutral salt) can possess any oxidising powers. I have made most careful experiments in this direction, but even when employing up to 10 grains per gallon of potassium nitrate and boiling for four hours, no increase in colour could be noted.
In the Journal of the Society of Chemical Industry, 1894, pages 367—375, a very interesting paper is communicated by Mr. J. B. Headman, showing the effect of various salts in brewing waters. Among the various salts used in these experiments was potassium nitrate which was employed at the rate of 50 grains per gallon, a rather large quantity, but one calculated to show fully the effects produced. Even this largo amount had no appreciable effect on the composition of the resulting wort when employed for mashing, nor did it produce the darkest wort of the series, by a long way. It is when we come to the fermentation stage that its effects come out, and in this case it gave the worst attenuation of all the series, thus showing the powerful action of the nitrates on the living organism, the yeast. The resulting beer had the least alcohol of any, and possessed a heavy smell and unpleasant taste.
Estimation of Nitrates and Nitrites
A great number of processes have been proposed for the estimation of these salts in waters, four or five of which are of sufficient accuracy to give reliable results. The direct estimation of nitric acid is impossible, as the nitrates are all soluble, the aim of the bulk of the processes being to convert it into some body, of which the most minute trace can be estimated. In the Frankland-Crum process, chlorides are first removed from the water by the addition of silver sulphate, the water is reduced to small bulk by evaporation, and the concentrated residue rendered strongly acid with pure sulphuric acid, is shaken with mercury in a graduated tube. By this means the nitric acid is converted into nitric oxide, the volume of which is measured, and from this the amount of nitric acid in the water is calculated. The method is accurate when dealing with large quantities of nitric acid, but with minute traces it is not sensitive enough, and I do not think it very largely employed.
The next general method depends on the fact that nitric acid may be converted in ammonia by powerful reducing agents, and, as the minute quantities of ammonia can be estimated by the Nessler test, it becomes a most sensitive reaction. The two reducing agents employed are metallic aluminium or zinc coated with finely divided copper, commonly known as the copper-zinc couple. We always employ this latter method, finding it to be quick and very accurate,
and think it is the most used of any. A brief outline of the process is as follows: 250 c.c. of the water are evaporated to a small bulk, a small piece of lime being added to decompose any ammoniacal bodies present, though in many cases this is not necessary, especially where the water contains chalk. Zinc in the form of thin foil is torn into strips and introduced into a conical flask of about 300 c.c. capacity, fitted with a tap, funnel, and connected with a good condenser.
Tearing the zinc produces curved irregular strips, which cause good circulation of the liquid, whilst, if cut, it lies flat on the bottom of the flask, and does not afford so much surface for action. It is now necessary to clean the zinc, which is always greasy, probably from oil used to assist the rolling, and this is best done with a mixture of caustic potash and methylated spirit. The flask is warmed, and in a few moments all grease is removed and the surface of the zinc left quite pure; the cleaning fluid can be used many times. The zinc in the flask is now washed with tap water till free from alkali and then covered with a dilute and slightly warm solution of copper sulphate. Almost immediately the copper begins to be deposited on the zinc ns a black powder, and in a few moments the red colour of the copper begins to show. The solution is then quickly poured off and the copper-zinc couple well washed with tap water, a final washing being given with water free from ammonia. A little care is necessary at this part of the process, for, if the copper solution acts for too long a time, the deposit is very loose and all washes off; for the same reason, the washing waters should be poured on and decanted very gently. The flask is then fitted to the condenser and the water residues added, together with about 1c.c. fairly strong sodium hydroxide solution. The washings of the dish are added and the contents of the flask distilled from a very gentle flame into a 100 c.c. flask; when about 80 c.c. have been collected, the flask is removed, made up to mark with ammonia free water, and a portion (usually 10 c.c.) taken for the Nessler test. Seventeen parts of ammonia (NH3) equal 62 parts of NO2 or, in other words, multiply the ammonia found by 3·65.
The aluminium method depends on the same reduction, and is performed in exactly the same way, and as sheet aluminium is now so cheap it may be found more convenient than the zinc.
Another method for the estimation of nitrates depends on the oxidation of indigo, whereby a colourless body is formed. The water is mixed with its own bulk of pure strong sulphurio acid, and standard indigo is added till a permanent colour is produced. The method is quick, but it does not give very accurate results, the employment of so much sulphuric acid probably leading to this, for, as is well known, it is difficult to obtain sulphuric acid absolutely free from all traces of the oxides of nitrogen.
It is seldom that a quantitative estimation of nitrites in a sample of water is required, the usual custom being to apply a qualitative test for their presence, which generally does not exceed a very minute amount. In most processes for the estimation of nitrates, any nitrites present are estimated at the same time, and, though the calculation finally is made into nitric acid, the error is very small. I have made careful experiments with the copper-zinc method, and find it decomposes both nitrites and nitrates, exactly the calculated amount of ammonia being obtained.
For the detection and estimation of nitrites two chief reagents are employed, being either potassium iodide or metaphenylenediamine. In the first case, the potassium iodide and a few drops of starch solution are added to the water to be tested, which is then rendered acid with dilute sulphuric acid. This liberates nitrous acid from the nitrite, which in turn sets free iodine from the potassium iodide, and this then combines with the starch present to give the usual blue colour.
In the other method, the water is rendered acid, and a few drops of a solution of metaphenylenediamine is added. A yellowish-brown colour is produced, the depth of which is proportional to the amount of nitrous acid present. The test is a very sensitive one, and will indicate 1 part of nitrous acid in 5,000,000 ports of water. By working in Nessler tubes and matching the colour with a standard solution of nitrite, the process is rendered quantitative, and the nitrous acid found is combined with the proper base.
The removal of nitrates from a contaminated water is a matter of considerable difficulty. I have made some experiments on this, but have never obtained satisfactory results, and time has prevented others being attempted. Any method of precipitation is, of course, out of the question, the only practical way being to decompose them either to free nitrogen or to ammonia, which would be removed by subsequent boiling. As sulphites are sometimes employed for this purpose, I have carefully tried them on the small scale. In the first place, 250 c.c. of pure water, containing 10 grains per gallon of potassium nitrate, were boiled for four hours with enough bisulphite of lime to give a smell of sulphurous acid to the water. At the end of this time the water was cooled, made up to the original bulk, and tested. The total amount of nitrate was very slightly diminished, and a very faint trace of nitrite was found, bat no trace of ammonia. The experiment repeated only with sodium sulphite gave exactly the same amount of nitrate after as before, and no trace of nitrite was found.
It will thus be seen that sulphurous acid, free or combined, cannot practically be used to decompose nitrates in a water, though I believe they are of service indirectly, and when a water contains much nitrate I always recommend the use of a little bisulphite of lime, not to try and decompose the nitrates, but to help to keep the yeast pure and strong.
For the some reason I recommend the use of a little organic yeast food, not constantly, but in, say, alternate brews. There is little doubt that the action of nitrates is on the yeast itself, and therefore the more steps we take to keep this healthy and vigorous the less the effect of the nitric acid will be.
I had intended to try the effect of a spongy iron filter, prepared by soaking coke in a solution of a salt of iron and then igniting at a white heat. Unfortunately, time has not permitted mo to finish these experiments, but I believe the process might be of service in case of highly nitrated waters, provided means could be adopted for preventing any of the iron from going into the solution.
I have also made some careful experiments as to the effects of nitrates in the brewing process. In the first place, wort containing 20 grains per gallon of potassium nitrate was fermented with ordinary yeast and tested from time to time for nitrites, but none were ever detected. This would prove that yeast is not capable of obtaining oxygen from nitrates and so reducing them to nitrites.
The question then arose whether yeast was capable of assimilating nitrates in the same manner as the higher plants can. To test this, wort was made containing 10 grains per gallon of potassium nitrate, and pitched with the usual quantity of yeast. A little was filtered at once, boiled with sodium hydroxide to remove free and combined ammonia, diluted, and the nitric acid estimated by the zinc-copper couple method. The other portion was allowed to ferment for four days, and the nitric acid again estimated, with the result that the same amount of nitric acid was found, which, I think, proves that the yeast itself is without any action on the nitrates.
Another series of experiments was made to find the exact effect of nitrates on the vigour of the fermentation. A quantity of pitched wort was divided into four equal parts, the first being fermented without nitrates, the others with 5, 10, and 15 grains per gallon of potassium nitrate respectively. They were all fermented in the same shaped vessels, and everything was done to render the results exactly comparative.
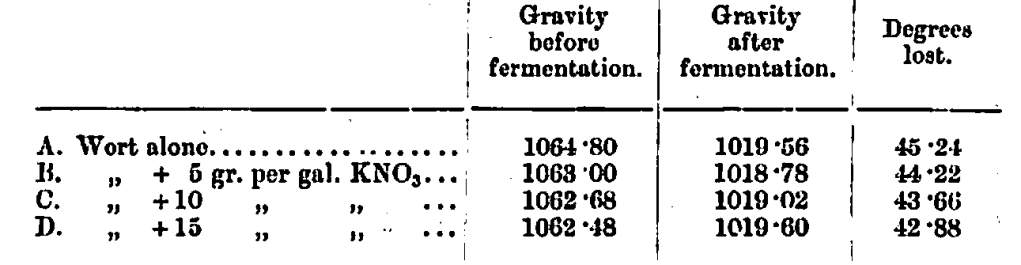
I think that the effect of nitrates on the fermentation can be well seen from these figures, and I have no doubt but that, on keeping, the beers would have shown corresponding acidities.
It is, of course, impossible to determine the exact effect of nitrates or really of any other salts, in the laboratory; actual practical experience of brewing for some time with such waters is necessary, and each experience is of much greater value than laboratory tests. It is impossible in the laboratory, without employing enormous proportions, to detect the slight differences in fermentation, yeast crop, attenuation, brilliancy, condition, and soundness, all of which an experienced brewer, working on an infinitely larger scale, would detect in an instant, and I hope to receive the benefit of such practical experience in the discussion which will follow, and which, I hope, will be a good one.
Discussion
Mr. Rogers remarked that he thought brewers would derive much benefit from the paper, because the subject of nitrates in brewing waters was little understood. It had been stated by certain brewers that good ales could not be brewed with waters containing nitrates. Personally, he did not think that nitrates had any harmful effect in brewing. He believed that the production of dark worts, which had been ascribed by some to the presence of nitrates in the waters, was caused by the attenuations having been stopped.
Mr. Hoyle described a case in which a certain Yorkshire water, when used for brewing, gave rise to worts of a “foxy-red” colour. An investigation by scientific experts having been instituted, it was found that the water was contaminated by flowing through some marly measures, into which nitrates had been put. He believed that the water in question contained 6 grains per gallon of nitrates. The question of nitrates in water had engaged the attention of the Wakefield Corporation, which had spent a considerable sum of money on the erection of filters containing carbide of iron; the filters had the effect of reducing the nitrates. He should be glad if the author could inform him what was the maximum amount of nitrates that could be present in a water used for brewing pale ales without danger.
Mr. Mitchell inquired if the brewing waters of Burton-on-Trent varied much in composition.
Mr. Evans, in reply, said that it was commonly supposed that nitrates caused darkening of the wort during boiling; personally, however, he had never observed this. The time at his disposal did not admit of his trying the effect of the various open filters. When more than 3 grains per gallon of nitrates was found in a water, it was his experience that brewers complained of poor attenuations. He had not had much experience in the analysis of Burton waters, but, so far as he knew, these waters came from the same strata, and therefore closely resembled one another. Most breweries possessed other wells, the water of which was employed solely for cooling, cask washing, &c.
A vote of thanks to the author brought the proceedings to a close.