MEETING HELD AT BREWERS HALL ON I0th APRIL, 1900.
Mr. JOHN M. HANBURY in the Chair.
The following paper was read and discussed :-
On Caramels
by JOSEPH W. LOVIBOND
The term caramel is usually applied to those soluble-coloured substances which am formed by the action of heat on cereals, sugars, and their allied products, and for which no satisfactory formula has yet been found, nor is it easy to realize how a single formula could describe the composition of all the substances now sold under the term; for instance, whatever properties the high-dried kiln and amber malts have in common with the roasted malts and darker caramels, it is obvious that there must also be important differences.
Users of caramel know to their cost of the wide differences in colour, flavour, and stability between caramels of apparently similar manufacture. No means at present exist, other than the one to be described, by which he can estimate these differences, although it is of importance that he should have information on the points before the cost of purchase and manufacturing expenses are incurred–to say nothing of the loss of public appreciation which always follows the use of variable material.
Of these three factors–colour, flavour, and stability, colour is the only one which can at present be measured and stated in definite terms; and the object of this paper is a preliminary attempt to find a method of correlating the two latter through the former by establishing the specific colour of each sample.
Specific colour is a new term which requires explanation; it means the rate at which the colour of a substance increases for regular increasing densities. A common impression has prevailed, that if the density of a substance was doubled, the colour also would be doubled; but this is never the case. The power of colour measurement has now demonstrated that each substance has a specific rate of colour increase of its own which is constant so long as the substance itself remains unaltered; the smallest change, either of kind or of quality, alters the rate of colour development, so that the original constants no longer apply – the altered substance acquires constants of its own. The colour constants can be expressed numerically in terms of standard units, or graphically by means of curves.
The method of establishing specific colour curves of such a substance as caramel, for instance, is quite simple. A percentage solution is first made, then colour measurements are taken at known increasing densities. These are plotted on a chart at those points where the ordinates, which represent colour units, intercept the abscissa which represent strata thicknesses, or densities; then by connecting these points curves are established which are specific for the substance in question.
I need not occupy your time by a detailed description of the apparatus, which has already been published, and is on the table for inspection. The results have proved to be so reliable in practice, that when the colour constants of a substance have been once established, the identification of a similar substance can be determined by their similarity in the rate of colour development; the smallest change alters the rate, and is made evident by alteration in the shape of the curves.
It will be at once seen that the value of the method depends on the accuracy with which the colour measurements can be made; the method has now been in use in many laboratories for a variety of purposes for nearly 10 years without serious question arising, either as to the soundness of the principle or accuracy of detail.
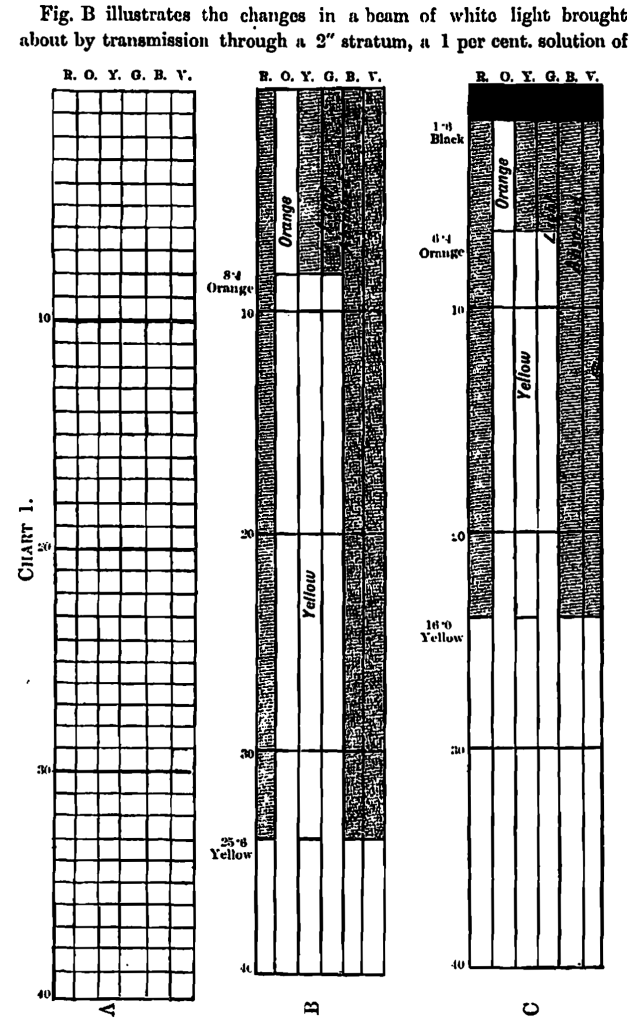
The colours of every-day life are derived from white light, and the constitution of an ideal white light is represented by Fig. A, Chart 1, which illustrates the ray composition of a white light of 40 units of luminous intensity as being made up of the six colour rays, each of 40 units colour intensity; the horizontal divisions represent white light units, whilst the perpendicular divisions represent the colour rays: this equivalence of the six rays cannot be broken without developing colour.
The conditions which govern the colour development in an abnormal beam are defined in Law 2 of our code (this Journal, 1897, 3,107) as being that “of the one preponderating ray, if the colour be simple, or of the two preponderating rays if the colour be complex; the depth of colour is in proportion to their preponderance.”
Fig. 13 illustrates the changes in a beam of white light brought about by transmission through a 2” stratum, a 1 per cent. solution of cararmelised sugar (No. 2 on our list), showing the darkened portions of the red, green, blue, and violet rays as absorbed, leaving 25∙6 units of yellow and 8∙4 units of orange in preponderance, and colouring the whole beam in these proportions.
The only two colours which enter into the composition of the caramels here dealt with are yellow and orange, except in the case of the charred grain from the Swiss Lake dwellings, when yellow is replaced by red; but as this has been under exceptional conditions of time, and is obviously overheated, it may be considered as an exception. This sample was kindly given me by Mr. Beavan, of Warminster, and, in order to make some comparisons with overheated wheat, I have contrasted it with a small variety of wheat (Lincoln square head) which has been submitted to different temperatures, beginning at 210o C., when in this instance colouration by heat begins, and ending at 250o C., when the grain appears to be wholly carbonized in so far that it then contains no soluble colour. The laboratory-heated sample is not distorted so much as the Swiss Lake dwelling sample; but as we know nothing of the conditions under which the latter was heated, it would be unsafe to draw conclusions. The colour of a chalk well water in a 12-inch stratum has been added as a contrast to show how little soluble colour is now in the grain.
Some caramels, notably those of the higher priced roasted malts, have a black factor in addition to the yellow and the orange. As those three terms comprise all the visual sensations distinguishable in caramel solutions, it follows that a caramel colour can be qualitatively described by the use of the three terms – orange, yellow, and black, and quantitatively described when these terms are associated with their unit value.
The term black is here used in a relative sense as one of the factors influencing colour depth. It means the degree of lessened luminosity caused by the absorption of white light, in this case by a caramel solution of roasted malt, No. 40 in the list; the colour composition is quantitatively represented in Diagram 1, Fig. C, by 1∙6 black units, 6∙4 orange units, and 16 yellow units.
It must be explained that the black factor may arise from two causes – it may be an inherent property of the caramel itself, as in this case, or it may arise from loss of light caused by the scattering effect of solid matter in suspension. The instrument itself has no means of discriminating between them, it simply registers loss of light; therefore, in measuring turbid caramels for specific colour, they must be first filtered: the proportion of loss of light due to each cause is then found by comparing the measurements before and after filtration.
COLOUR DEPTH
The difference in visual effect between the orange, yellow, and black factors may be described as follows :-
The orange factor governs the colour depth when no black is present; therefore, in such cases, when quoting colour depth only, the unit value of the orange factor is sufficient for general purposes. The yellow factor influences colour character without much affecting colour depth. The black factor, as already explained, has a direct influence on colour depth, and in order to contrast the difference in visual effect caused by orange alone and orange combined with black, I have put the 14o orange measuring glasses used for No. 4 (a treacle with no black) in a frame with the measuring glasses used for matching black malt, No. 40, which contains 1∙0 units of block in addition to 6∙1 units of orange. You will observe that they are about equal in colour depth, but No. 4, without black, is of greater colour purity.
BEST CARAMELISING TEMPERATURES
As the results of investigation accumulated they were found too complicated to be dealt with in a single evening; it was therefore decided, for the purpose of this paper, to limit the term “caramel” to those products formed at the high temperatures just preceding the temperature of carbonization.
It may appear at first sight that this definition is vague, but as we proceed it will be found that there is a wide variation in the best caramelizing temperatures for different materials; it will also be found that the limits of temperature for each material vary considerably, but that the temperatures at which caramelisation begins and carbonization takes place are easily defined.
In making this set of experiments it was necessary to have some authentic samples of different sugars as a basis of comparisons; these, together with their chemical analyses, were kindly furnished me by Messrs. Lyle & Sons.
The object of the first act of experiments was to find the particular temperature which gave the highest tinctorial results for each material; in order to obtain this, a number of 1 gram samples of each material were simply heated in a hot-air bath, the temperature of which could be easily regulated; samples were taken from time to time at increasing temperatures, and measured for colour until that temperature was found which gave the best colour results. Separate heatings were then made of each material, beginning at that temperature which induced the first sign of colourisation, and carried on through all the degrees up to the point of total carbonisation. In preparing the samples for measurement, 1 gram of the raw material was made up to 1 litre with water, and this proportion was maintained in all cases.
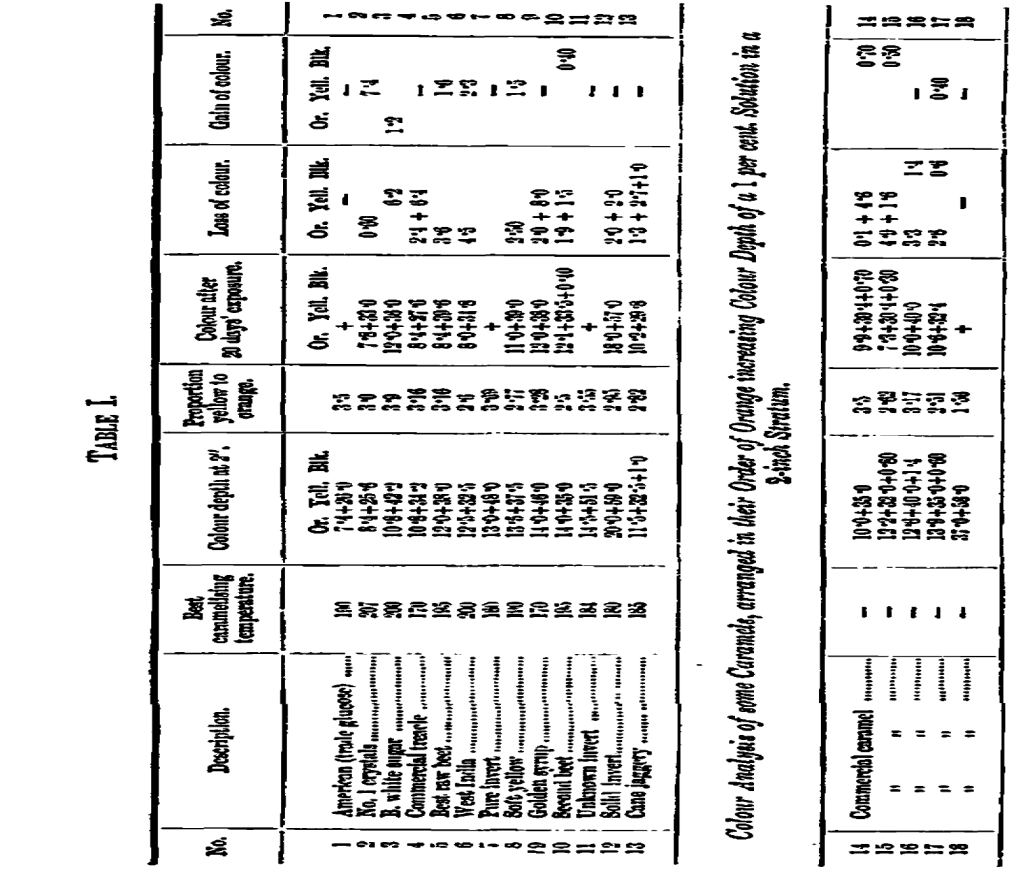
The measurements of the verified sugars are collected in the first division of Table I, where the results are arranged in numerical order according to the unit value of the orange factor; orange, as already explained, being the factor which governs colour depth when no black is present.
Assuming that colour depth is of commercial value, we have in this column of orange units the relative value of the different samples for this particular merit, and in odor to obtain the cost per unit of colour depth, we have only to divide the orange units into the cost of the material in each case.
A general feature in the caramelisation of these sugars in the lower temperatures required by the inverts compared to the cane-sugars, the range for the inverts being from 160 to 190 Centigrade and for the cane-sugars from 190 to 207 Centigrade.
The differences in orange colour depth between the samples are considerable, ranging as they do from 7∙4 to 20 units; they are, however, too irregular and too few in number for classification now, and in future experiments chemical analysis will be of great assistance for this purpose. A similar condition of things applies to the comparison of yellow proportions to orange.
In addition to finding the test caramelising temperature and the cost per unit of colour depth, the same set of measurements supply some information which may be of interest, as it defines the loss arising from temperature fluctuations during manufacture; this is obtained by plotting the set of measurements used in obtaining the best caramelising temperatures on a chart, where the ordinates represent the colour units and the abscissa the temperatures which produce them; the curves so obtained show the loss of colour depth for each degree of variation of temperature from the best possible.

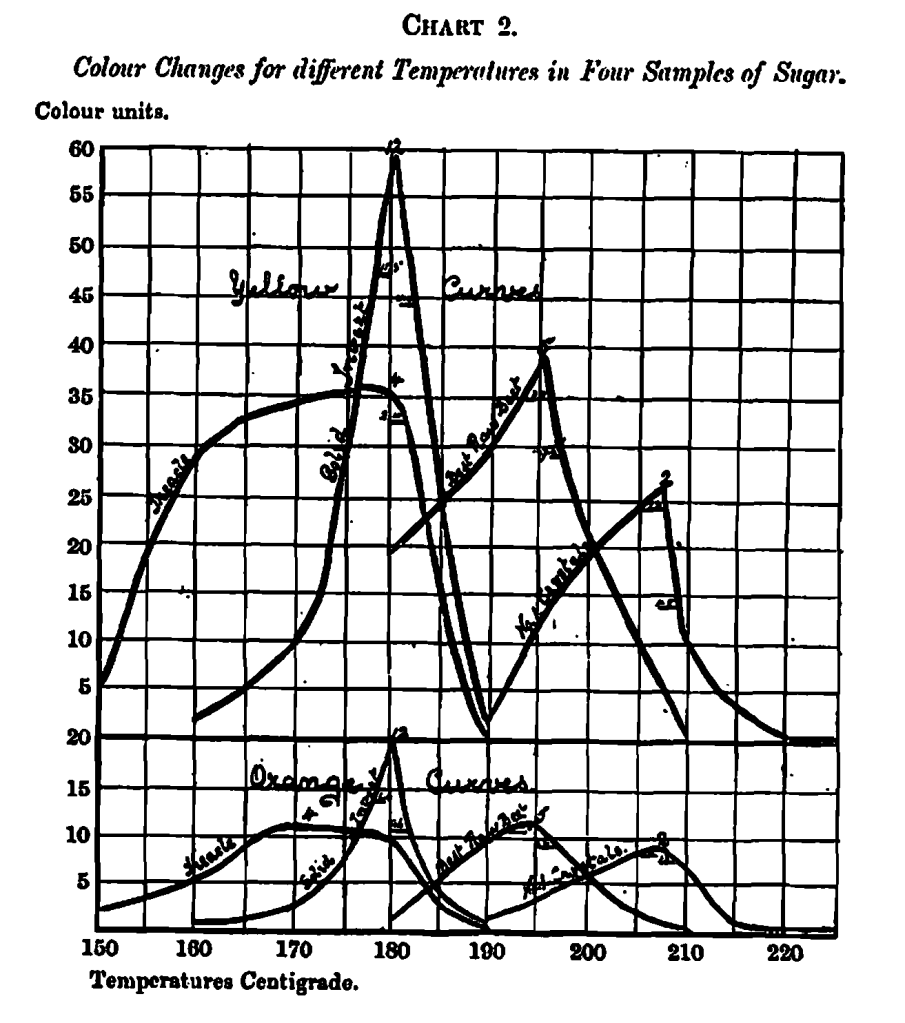
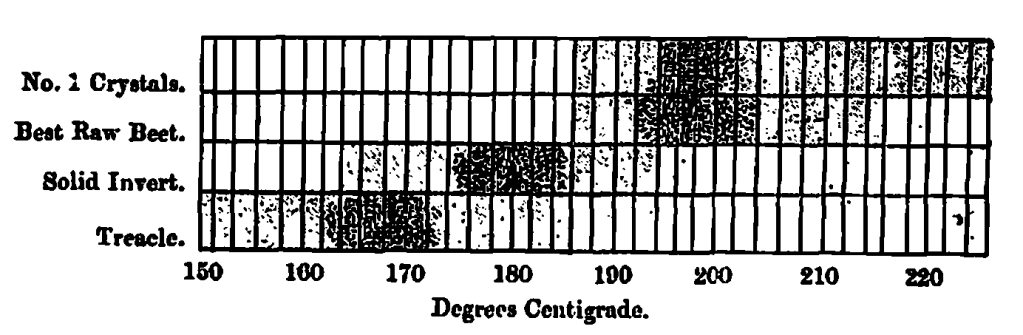
In illustration, four such curves are plotted on Chart 2, where the loss of colour arising from fluctuations of 2o on each side of the best caramelizing temperatures are indicated by black lines across the curve to a perpendicular from the highest point. If the fluctuations are on the right of the perpendicular, that is, in degrees higher than the best temperature, the loss is total by carbonization; but if on the left, that is, on the lower temperature side, the loss is more or less compensated for by an incomplete caramel. In the interest f the user some means is desirable for determining whether a caramel has been overheated or underheated. In order to avoid confusion of lines the orange and yellow curves are shown on separate charts; the similarity of numbers shows their association.
An examination of thee curves show their relative loss of colour depth; the percentages of loss appear in the following table :-
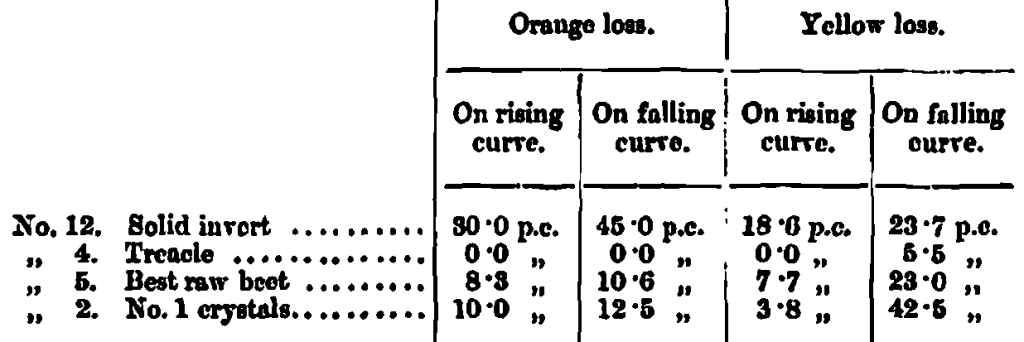
In order the better to realise the true colour sensation represented by these curves, the actual measuring glasses are arranged in a transparency in the same order of temperature which they hold as shown in Chart 2a.
Division 2, Table 1, Commercial Caramels
The measurements in the second division of Table 1 are those of five commercial caramels, and in making comparisons with the first division it must be pointed out that, in the case of the commercial caramels, the proportions are 1 gram per litre of the manufactured article, whereas in the laboratory sugar experiments, the proportions are 1 gram per litre of the raw material.
Even after making allowance for this difference in proportion, the commercial caramels do not compare favourably with the laboratory-prepared examples from sugars, except in the case of No. 18, which shows a much higher value in orange colour depth than the others; but this is coupled with a low proportion of yellow to orange. No definite conclusions can, however, be drawn, as neither the materials from which the commercial caramels were made, nor the conditions of their manufacture, were disclosed.
Division 3, Table 1, Roasted Malts
In comparing the colour measurements of the roasted malts and barleys with the sugar and commercial caramels, it must be noted that the latter two were wholly soluble, whilst the roasted malts contained insoluble matter.
Division 3 contains the measurement of a number of samples of roasted malts. It would be unsafe in our present state of knowledge to attempt the classification of these in any odor of merit; the most that should now be done is to draw attention to salient differences, leaving it to the experts to define what property, or combination of properties, they most value. It appears evident that the following have some bearing on the question :-
Stability of Colour
This has long been considered a merit in caramels and in roasted malts, and various devices are used to determine it. The method here employed was to expose 0∙1 per cent. solutions in clear glass vessels to a north light for 20 days, taking measurements from day to day. It was originally intended to make the period 30 days, but in some, cases organic changes took place towards the end of the longer period which created a different set of phenomena to those of the shorter period.
The results are shown in the three last columns of Table 1, the first column containing the measurements on the twentieth day, the other two columns showing the difference between these and the freshly prepared solutions; as there was a positive gain in colour in some instances it was necessary to provide two columns – one showing gain and the other showing loss of colour. The acquirement of this knowledge has been too recent to allow of an exhaustive classification. I have, however, selected five types of colour change by exposure, and plotted their fading curves in Chart 4.
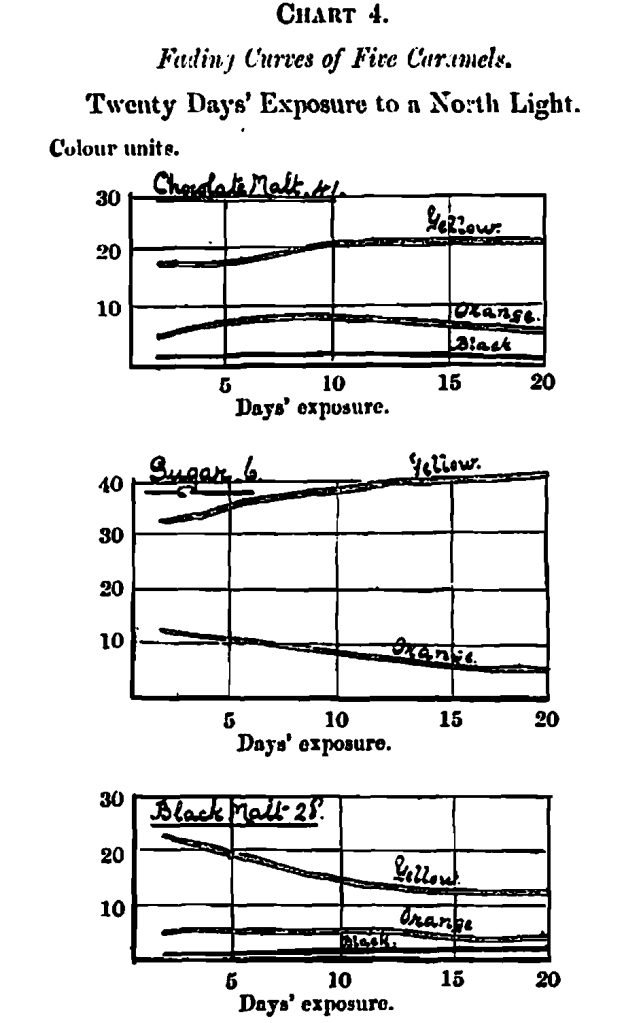
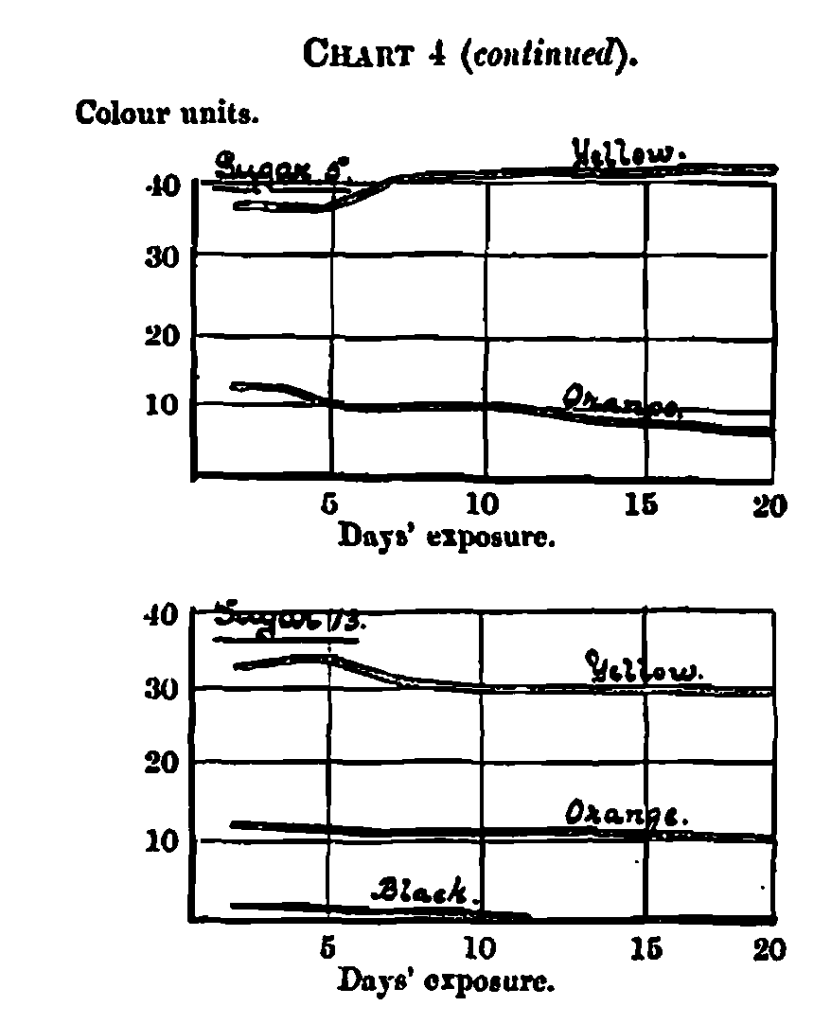
The first of these (No. 41), a chocolate malt, represents a type which increases in colour depth before beginning to fade; the curves show an increase, both in yellow and orange, for the first 10 days, after which the orange fades very slowly, whilst the yellow remains stable for the remainder of the term. The black is constant for the first 10 days and then begins to fade slowly.
No. 38, a black malt, shows the smallest possible increase for the first seven days, and a similarly small fading, both in yellow and orange, for the reminder of the term. The black increases slightly for the whole term. Considering stability of colour as a desirable property, there is little question but that No. 41 is the best in this respect.
No. 6, a caramel from West Indian sugar, shows an increase in yellow and a fading in orange during the whole period.
No. 5, from best raw beet, is similar to No. 6 in character; but varying in rate.
No. 13, from cane jaggery, shows a slight increase in yellow for the first four days, on increased rate of fading for the next four, and practical constancy for the remainder of the term. The orange shows a slight fading for the whole 30 days. I much regret the incompleteness of this section.
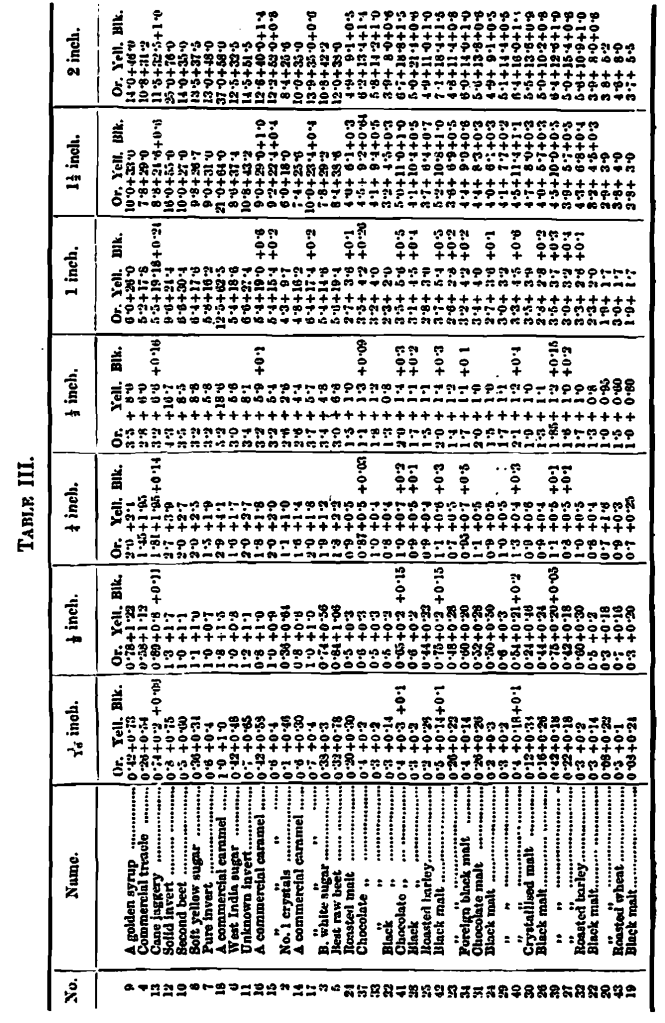
Specific Colour Curves
An examination of all the colour measurements here dealt with shows a wide difference between the proportion of yellow to orange, if the extremes are taken; but when their specific colour curves are arranged in an order according to the similarity in curves, as in Table 3, the difference between any two adjacent samples is very small. The whole set of curves is too voluminous for reproduction, but Chart 7 contains a selection of five examples illustrating the progress, the series beginning with a golden syrup, where the orange and yellow lines start on their upward trend at some distance from each other; in the next example, a treacIe, the lines begin their career a little nearer each other, and so on, until at the fifth (second beet sugar) they nearly touch at the initial measurement; in the sixth the lines begin to cross, and in each succeeding instance the point of crossing recedes a little, until in the last, No. 19 (a black malt), it has receded to two-thirds the length of the whole curve. The changes are so gradual as to make it difficult to point to a distinctive difference between the curves of any two adjacent samples, although the extremes vary so much.
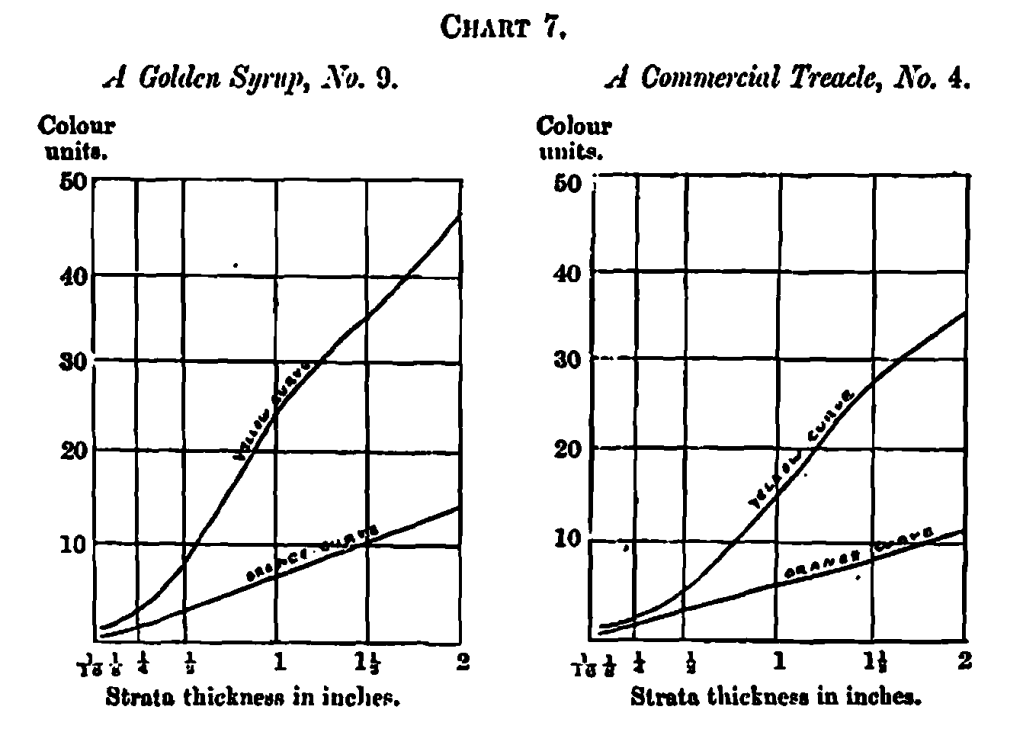
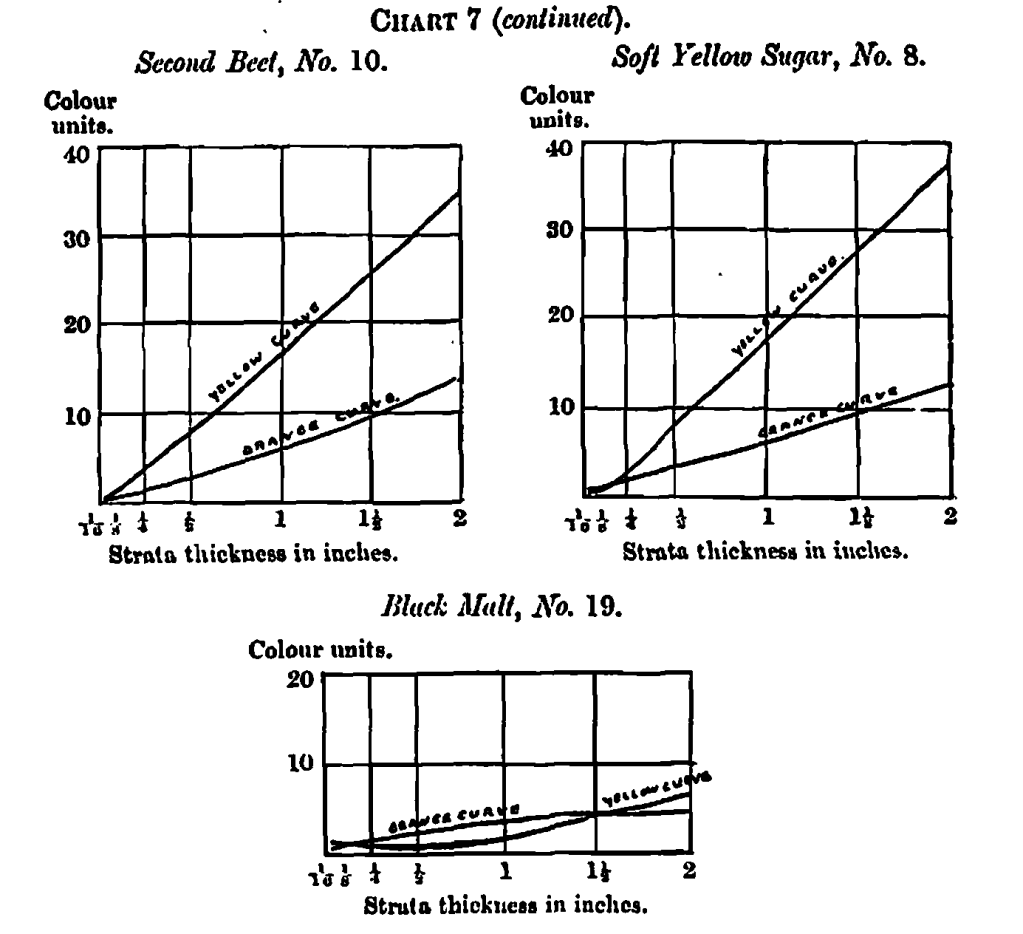
This gradual relation in the nature of the orange and yellow specific curves may indicate a gradual change in the nature of caramel itself, instead of sharply-marked varieties, or they may point to the possibility of a definite composition of caramel associated with some modifying conditions which, varying for different materials, alter the rate of absorption, and therefore the form of the specific colour curves. In some cases, complexity in chemical composition appears to induce corresponding complexity in these curves, as shown by causing greater deviation from straight lines. I do not here apply this to the caramel curves, but call attention to it as a fact to be remembered.
Two examples of each curve complexity are shown on Charts 5 and 6 – one containing the colour curves of nesslorised ammonia, arranged according to increasing percentages of ammonia, and the other the curves of a 1 per cent. solution of alkaline litmus. The ammonia curves are comparatively simple, whilst the litmus curves are complex, showing a pure blue only in the initial measurement, after which violet appears in increasing proportions, until at the 6” stratum the blue disappears entirely, being replaced by red. This change of colour from a blue-violet to a red-violet accounts for the difficulty in judging the neutral point between an acid and an alkaline solution when the solution happens to be strong, or what amounts to the same thing, a weak solution in a deep vessel, their visual effect being the same.
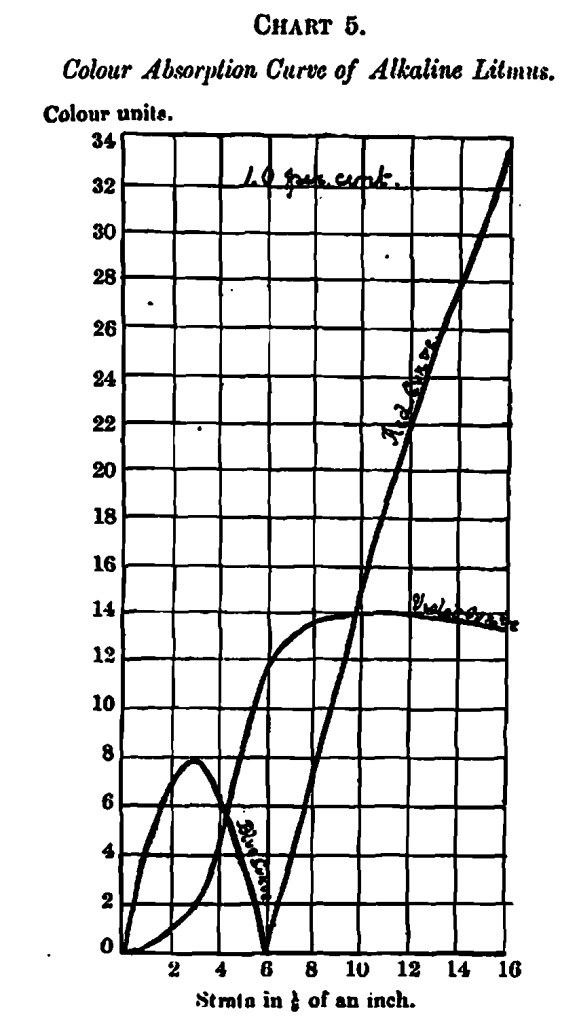
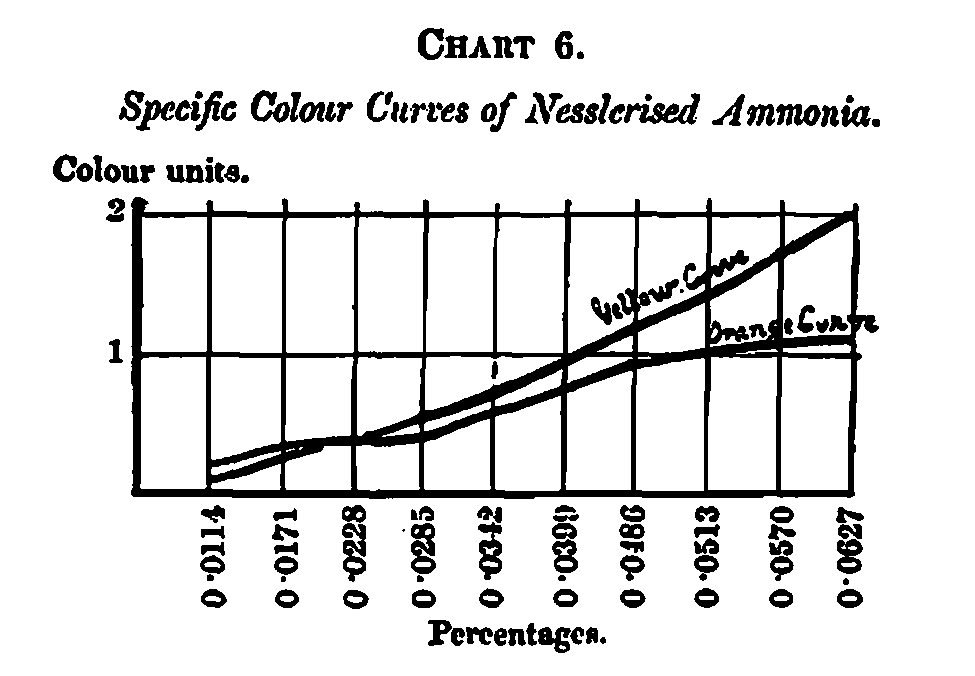
Any view which I may express concerning the odor of merit for the different properties dealt with must be considered as tentative only, being simply advanced as indicating lines for further research. Probably every variety of caramel in the market has its special manufacturing value; but it is obvious that the correlation of any merits or demerits, with their specific colour curves, can best be carried out by those exports who have an opportunity of watching the results in actual practice.
With these remarks I should place considerable value on a high proportion of yellow to orange, because it has already been demonstrated to be an element of intrinsic worth in pale malts, as well as in the baking value of flours. I also consider that colour stability should take a high position, bit have not yet sufficient data to venture on any expression as to the order of merit between the rate of fading, the proportion of yellow to orange, the colour depth caused by the orange factor, the colour depth caused by the black factor, and have no doubt but that an examination of those caramels and roasted malts which have proved their intrinsic worth by commanding the best prices in the market, would go a long way towards the construction of a scale of merit.
There is one important factor of value which I have not yet dealt with — I mean that of flavour; an examination of this was contemplated, but was found to require a wider range of experiments than could be dealt with in a preliminary paper of this character.
Whatever may prove to be the relation between the specific colour curve and quality differences, it is evident that by this method of examination any variety of caramel or of roasted malt can be identified with certainty; and, further, that when a user knows that variety which is best answers his purpose, he can make sure whether he obtains is or not.
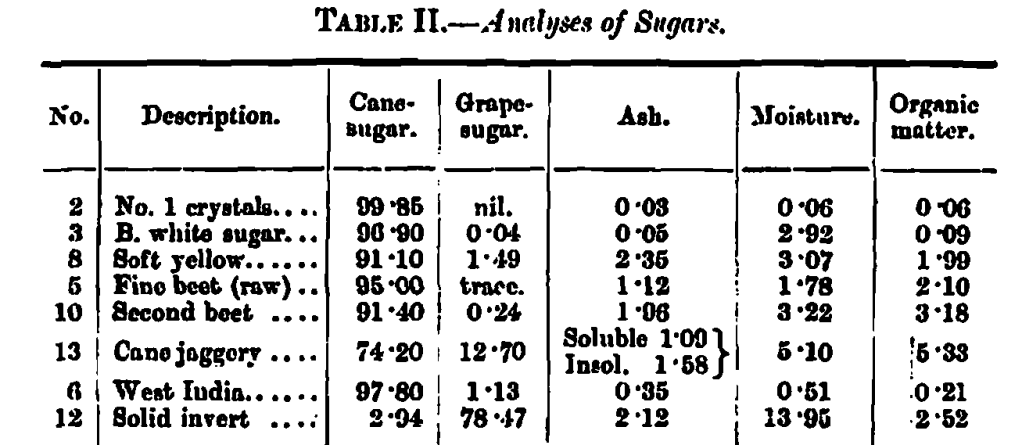
DISCUSSION
Mr. Chapman said whilst it was a great advantage to hear a paper from Mr. Lovibond on R subject which he had so thoroughly made his own, there was the slight drawback, that he was apt sometimes, perhaps, to make use of language that was not entirely intelligible to those who had not studied the matter so deeply, and he should like a little further information with regard to one or two expressions he made use of. He frequently used the term “black factor,” and he (Mr. Chapman) would like to ask precisely what was to be understood by the expression. He had the pleasure of hearing a paper which Mr. Lovibond read before the Society of Chemical Industry some years ago, in which he said that whenever this black factor was discoverable in the colour analysis of malt solutions, he considered it told against the malt in which it was almost entirely lacking was preferable to one in which the “black factor” was found. Did Mr. Lovibond mean by that simply the absorption of light owing to the presence of very small suspended particles, or had it some other meaning? One point had occurred to him in connection with the figures showing the best caramelizing temperatures. It seemed to him that the numbers given in the table lost to a great extent their scientific value, because one very important factor had not been taken into account, namely, the percentage of moisture. Every chemist knows perfectly well that a dry organic substance could be heated to a comparatively high temperature without the production of colour, whereas in the presence of moisture colour was formed at a much lower temperature. The temperature given on the diagram showed exactly what one would expect to see, that treacle, for example, with a high percentage of water caramelised at a much lower temperature than sugar crystals which had a small proportion of water. In other words, the optimum colour temperature was very largely inversely proportional to the amount of water contained in the samples. Therefore, in speaking of the temperatures at which the maximum colour was produced, some statement should have been made with regard to the water percentage present in the samples under observation. Then he should like to ask a question with regard to the actual tintometer difference between black malt and caramel solutions. They all knew that the colour-tone of a caramel solution was distinctly different from that of a black malt solution. Many brewers used caramels for that reason: the colour being richer, and the tinctorial power distinctly higher, and he would like to know if Mr. Lovibond could give any scientific explanation of this particular difference. With regard to the use of aniline dyes, he had himself come across one sample of caramel in which an aniline dye was present, but he thought the practice of employing those dyes for this purpose widespread. He could well understand that examination by the tintometer might in such a case afford valuable evidence, although the detection of such dyes by ordinary chemical means was not at all difficult. The whole thing seemed to him to resolve itself into this. Brewers who had to use caramels had to ascertain whether a certain sample submitted to them was one which would answer their requirements.
In the first place, of course, it was essential that the caramel should have a high tinctorial power and that it should be cheap, i.e., that the price charged per unit of colour should not be too high. In the next place the colour should be good. Then, again, it was desirable that the colour should be moderately permanent, that a very small proportion should disappear during fermentation, and that other influences, such as were brought to bear upon it during the ordinary brewing process should result in very little diminution in colour. Then, again, there was another point which had not be referred to in the paper, and that was the question of the permanency in solution of the colouring bodies of the caramel. Every brewer knew perfectly well that there was a tremendous difference between caramels in this respect. Some when added to beer at once caused a brown flocculent precipitate, whilst others caused very little or none. All these were points about which brewers wanted information, and he could not help thinking that the best plan was to apply in the brewing room or in the laboratory, or on a small scale, experiments similar to those which would have to be made subsequently on a large scale. With all deference to Mr. Lovibond, he thought that experiments in these directions would, perhaps, yield results of more value and interest to the brewer than complicated tintometrical observations. Still he thought Mr. Lovibond had done much towards placing the measurement of colour on a sound basis, and both scientific men and manufacturers must be extremely indebted to him for his labours.
Dr. Thorn said in this paper Mr. Lovibond had certainly broken fresh ground, which would be of very great importance and interest to those who had to examine and decide on the value of caramels. There was no doubt, although they had certain data to go upon at present, that this was a difficult task. The different samples which came into the market varied so enormously, and it was very difficult indeed to decide with present methods which was the best for the purpose in view. It seemed to him in this method they had one which would help very much in determining those values. To his mind, one of the most important points brought forward was the permanency. The author had spoken always of a 0∙1 per cent solution in water, and he would like to ask him whether he had yet been able to carry out any experiments on varying strengths of solutions, or on the caramels kept in the solid or dense state, because a very great change certainly went on in caramels in the dense or semi-fluid state in which they came into the market. Those who had kept caramel for any time know that they changed very much in that form, and whether that change took place in a similar direction to that in the weak solutions, was a point of great interest, although of course it was when in solution that the change was most important to the brewer. Again, he should like to ask Mr. Lovibond whether, in making these determinations of the tintometer value of the orange and yellow and black, he had attempted at all to differentiate between the different solubilities of the caramel. They knew that when used for brewing purposes there was always a certain amount of alcohol in the solvent medium and that different caramels differed very much in their behavior in solvents containing different percentages of alcohol. He should like to know whether the tintometer value varied according to the solvent, whether, for instance a pure aqueous solution showed the same as a solution made for a 0∙1 alcohol solution. At present very little was known as to the composition of these caramels; anything which would throw light on their formation and composition would be of great value. He thought the method Mr. Lovibond had brought forward would form a very important help in determining the differences of the different caramels, and be a very important method of investigation. Mr. Chapman had referred to the influence of water, and of course, that was the point which would have to be determined, and must have a very great influence, but one of the great difficulties which the caramel maker had to contend with was to get any means of determining as the caramel was being made the best conditions when not only the maximum intensity but the best quality of colour was obtained. It seemed to him that the curves which Mr. Lovibond had shown would help very much in that direction. It might also help very much in enabling chemists to differentiate between the different bodies present in caramels, and to isolate, if it were possible to do so, what might be called true caramel from different substances. If there were, as was likely, several definite caramels, it would enable them, he thought, to determine whether there were a small number of different definite compounds, or whether the variations went on by gradations that were numberless in character.
Mr. F. BOWER said he should like to ask Mr. Lovibond whether the percentage of sugar he had in solution had any effect on the specific colour absorption, and the rate of colour absorption. One might use a 5 per cent solution and examine that in 1-inch, 2-inch, 3-inch, or 4-inch cells, or one might use a 10 per cent solution and examine that every ½ inch, and it would be interesting to know whether there would be any difference in the curve produced under those two conditions. He put the question in consequence of something that occurred in his own experience. He was examining an invert syrup, and he found in a great number of cases, when he examined it for colour, in the form of syrup, that this was of a better colour, if he might so express it, than the standard glasses, but when he dissolved it and made a 10 per cent solution he found that the colour was no longer better than the glasses, but there was a black factor introduced; and he found that taking higher percentages than 10 per cent., according to the percentage so did this black factor appear or disappear. It would be interesting to know Mr. Lovibond’s experience on that point.
Mr. HERON said, as he was partly responsible for Mr. Lovibond reading this paper, he felt delighted to find that he had opened up a lot of new ground, and had given those who were interested a great deal of food for reflection and research. He quite agreed with the author with regard to the black factor, because for some years – in fact, since Mr. Lovibond had brought the value of the tintometer so prominently before the brewing world, in a paper read before the Society of Chemical Industry – he had taken a very active interest in it, and could fully confirm all that he then said, and the work he had done since in that respect. No matter what the malt appeared to be in other respects, if the black factor entered into the tintometric examination it invariably turned out to be a malt of inferior quality to that which did not show the black factor. The same also applied to the examination of black malts and caramels. One thing he was not quite clear was, What were the caramels which Mr. Lovibond had examined? If he rightly understood him, he took the different forms of raw sugars and heated them to a temperature of 212o C. He had a great deal of experience in the manufacture of caramel on a large scale in years gone by, and certainly the temperatures reached in the production of caramel were very much higher than that, and the caramels which resulted, even when the process was carried out to the verge of carbonization, had shown a higher tinctorial power than those manufactured at a much lower temperature. One of the first things the brewer would look for in caramel was the colour intensity. That which gave the highest colour intensity was generally the most valuable but apart from that, there were other factors to be taken into account. A caramel which would make a perfectly brilliant solution in water would answer all the purposes in regard to the tintometric observation, and might at the same time be one which, when added to beer, would give altogether different results to what the brewer thought. For instance, some were slightly alkaline in character, and when such were added to beer, it would in a short time lose a large quantity of its colour. It was advisable, therefore, unless something was known of the caramel and its previous character, to take observations in an aqueous solution, and in solutions which had been rendered distinctly acid by the addition of acetic acid. Then there was the question of the behavior of the caramel in beer. Some which would dissolve in water and show a high colour intensity would, when added to beer, show a positive cloud. He had examined some samples of caramel which would not show any precipitation whatever with water containing 50 per cent. of alcohol, and yet when added to beer would, throw down a precipitate. All these different matters had to be taken into account in judging of a caramel. From the observations Mr. Lovibond had brought forward it seemed to him that he was just on the fringe of a new world, and that if his experiments were continued – and he was fully aware of the enormous amount of trouble, patience, and time required for such work as he had undertaken, and if they were extended to commercial samples, a good deal more light might be thrown on these questions. Some years ago (the speaker) read a short paper before the institute, in which he incidentally mentioned the examinations of caramel, and he found that what he then stated had since been fully confirmed. One of the principal points he insisted upon was the importance of the specific rotatory power, and what he stated then had since been confirmed; in fact, the lower the specific rotatory power shown by any caramel the better was that sample in every way. At the present time there were, as the members were aware, two classes of caramel on the market, differing in properties and qualities. One was a class of caramel made by what might be termed the old process, simply carbonising the sugar in an open vessel; the other was called the ammonia process, in which the sugar was heated with a certain percentage of ammonia in a closed vessel under considerable pressure, and an exceedingly high temperature arrived at. He was in hopes that they would have heard something about the relative merits of the two classes of caramel, and he hoped that at no distant date, Mr. Lovibond would give them another paper on a more extended examination of caramel from the commercial point of view.
Mr. LOVIBOND in reply, said he could only say with reference to Mr. Chapman’s first remark that this science is practically a new one, and in many cases, terms had to be invented to describe a state of things which was not describable previously. Still he knew the difficulty referred to was a real one, because he had met with it frequently before. With reference to the black factor it certainly was not due to suspended particles, in any case in the list before them, because all the caramels used there were soluble, and if some were insoluble, such as roasted malts, they were filtered before being measured. The question of the percentage of moisture had no doubt a great influence on the value of caramel during the manufacture, and he did make a very wide series of experiments on that point. He began by dipping malt and grain samples in water for a minute, then lengthening the time, then increasing the temperature until he got up to boiling for 20 minutes, and he might say that under no circumstances did he get as good results as he did with the raw material without soaking. There was a point when it approached the value of the dry, but the little moisture certainly damaged it, and the high temperature with moisture damaged it, and boiling damaged it. With regard to the scientific distinction between caramels of black malt and the others, he could only point to Chart 1, which showed the difference in the ray composition of the beam of light transmitted by each. With reference to the colour finding again he could only point to the experiments shown. They were carefully carried out, and so far as the limited number of examinations were concerned he believed they were reliable. He had already answered Dr. Thorn’s first question with reference to the temperature, and with regard to the different kinds of caramels these experiments were only initial. The subject was never touched upon in this light until his paper was accepted by this Society, and they would all understand there was a mass of work done of which this was only a portion; in fact, it had taken the whole time of himself and his staff. With reference to the solubility of different caramels, in all the sugar caramels, there was no residue, whether they were made by himself or whether they were market caramels. With reference to water and alcohol differences, that was a branch he had not yet touched. Mr. Bower asked with reference to the percentages of the solution, and said the colour of the stronger solution was better than the glasses and got within range again as the solution became weakened. In making colorimetric measurements in a solution it was important that whatever might be the strength or the colour-depth of the solution in hand you must bring it down to somewhere near the colours in everyday life, in order to be within the scope of judgment but the vision, because the vision was accustomed to judge by daylight, and by a moderate colour-depth. Therefore, it was desirable to bring your solution to such a state, and he found for caramels, a 0∙1 per cent. solution was that state. If a much deeper colour was used, you got out of the range of measurement by glasses themselves, and into a condition of things where the measure was no longer of any value. Therefore, in very deep colour and low lights you must reduce the solution within measurable scope. Experience would tell you in a very little while how near you could come, but if you got beyond the scope of colour-vision judgment you would do no work at all.
Mr. BOWER said his question was Whether a high percentage solution with a small thickness would give the same kind of colour as a low percentage in a larger cell?
Mr. Lovibond said it would so far as his experience went. The whole question was comparatively new, so that in all cases he got his solution within the scope of the judgment of the vision, and thought it was necessary to do so, at all events at present. He had done a lot of work in that direction, but had not arrived at any conclusion. Mr. Heron also dealt with the black factor and its value or otherwise, and illustrated it by quoting the black factor of pale malts as being an undesirable element. He was quite right with regard to pale malts, and it might be so in black malts, but it did not follow; the whole thing had to be proved by experience. It was quite possible that the black factor might be a desirable quality in roasted malts – at any rate, it existed in all the best samples. It was a question which had yet to be settled, but what was most important that it should be reached. Mr. Heron was mistaken in saying there were no market caramels included in the tables. Five caramels in the second division were all market caramels. It was quite true that this method of measurement would not answer all questions; it only answered questions where the colour had a correlation to the properties which produced the colour. He looked upon it simply as an outside aid to chemistry. He quite saw the great deficiency of the paper, that with those things he had not be able to give a chemical analysis, but he was quite sure if they had been given, the value of the paper would have been enormously enhanced. A remark was also made concerning the commercial value of the experiments. He could not settle that. He was in the position of the inventor of a new method which was a measure, and it was for those who used it to define what the measure would do for them. He knew what it had done for him, and should not like to carry on his business without it now – in fact, he should be lost without it. He quite felt that the thing was only in its infancy, and, as Mr. Heron said, it opened up a new field of investigation.
The President proposed a vote of thanks to Mr. Lovibond for his very valuable paper, which he hoped when published in the Journal would be accompanied by the figures and diagrams which it had been illustrated. As Mr. Heron had very rightly said, it opened up a new world, and he hoped it would not be very long before Mr. Lovibond would give another paper on the subject, diving perhaps more deeply into it than he had already gone.