JOURNAL OF THE INSTITUTE OF BREWING
MONTHLY REVIEW (1954)
ON HEAD RETENTION
By Drs. W. J. Klopper (Experimental Station of the Dutch Brewing and Mailing Industry, Rotterdam) Received 21st February, 1954
The Σ –test for foam stability may give misleading results, and it is inadmissible to determine this value after dilution; moreover, the foam residue towards the end of degradation has enhanced stability. In a described method for determining “foam time,” beer is foamed under standard conditions and the time is found for 95% of the beer to return from the foam. This measurement, however, does not sufficiently characterize the foam properties of a beer, and it is proposed also to measure “foam adhesion,” obtained from the position of the highest persistent foam ring on the wall of the experimental cylinder. Foam time depends on high molecular protein degradation products and on the hop rate, whilst foam adhesion depends almost entirely on the hop rate and, more specifically, on humulone transformation products, roaming properties are not significantly affected by esters, higher alcohols or barley fat, in the quantities likely to occur in beer.
Introduction
A good foam stability is of the greatest interest to the brewing industry on the Continent. Consumers appreciate beers retaining their heads as long as possible; besides, they want to see many foam rings on their glasses and, preferably, for each draught a ring. In the present study, analysis has been made of beers with a good foam stability but with a bad capacity to form foam rings. Therefore it seems urgent to distinguish between foam stability and a second property of foams which will be further referred to as “foam adhesion.” So far in studies of head retention this last feature of a foam has been completely overlooked. Nevertheless it can be claimed that it is just as important as foam stability.
As a result of the present investigation in our Institute, some papers have appeared or are to appear in the Internationaal Tijdschrift voor Brouwerij en Mouterji (see, eg., ref. 8, 9, 10), but the opportunity of being able to publish a survey of the results in this Journal, thus reaching a larger public, is much appreciated.
Methods of Analysis
The most common methods of foam estimation are those of Blom2, Ross & Clark13 and Brenner, McCully & Laufer.4 As is known, the first two methods are founded on the theory that foam degradation should be a reaction of the first order. This, however, is in contradiction of the results of Brenner,4 Lienert12 and Kolbach.11 Yet it is surprising that some of these investigators had recourse to a standardization of the many variable factors of the Ross & Clark method in order to get reproducible results. It seems inadmissable to base a method of analysis on a false theory and, as will be demonstrated, this may lead to bad results.
Brenner stated that foam degradation only approaches a first order reaction for a limited period of time. In the long run, sooner or later the velocity constant of unimolecular reaction decreases or, as Σ = 1/k, Σ increases. So the last remnants of a foam have an improved stability. This can be completely confirmed.
The characteristic of a first order reaction is that the velocity constant is independent of the concentration. Although it is questionable if we may extend chemical rules to a physical process such as foam degradation, the fact that this does, strictly speaking, not behave like a unimolecular reaction implies that the concentration (in this case the amount of fluid present as foam) has an influence on the result to be obtained.
Only when the measurements are done with constant amounts of fluid can comparable results be expected. It is desirable to state distinctly that the use of Blom’s method guards against false results, as with this method foam degradation is always measured handling a constant amount of liquid (100 g.). The Σ-test of Ross & Clark, however, is generally performed with a standardized scheme of time. When the foam has been formed, a constant time is waited (1-2 min.) before the foam is separated from the liquid and the estimation is begun. During this time there is not only a drainage of fluid from the foam, but also some foam degradation. So, when working with bad foams the measurement is run with smaller amounts of fluid than when handling stable foams. This may lead to results which are not representative of the condition of a foam in reality.
Working with a “foam tower” as in the method of Gray & Stone,6 a very improbable result was found on one occasion with the Σ-test. The de-foamed beer, from which the bulk of the foam-forming substances had been removed, yielded nearly the same Σ-value as the original beer. Nevertheless, there was an enormous difference in the practical foam properties. While the original beer had a good foam stability, the foams of the de-foamed beer very quickly degraded to poor residues.
The Σ-test was done with 150 ml. of decarbonated beer in a funnel of 7 cm. diam. and 600 ml. content. By means of a porous candle, this fluid was foamed up to a volume of 500 ml.; the beer was separated from the foam 60 sec. later. The foam was allowed to collapse during a period of about 180 sec. after which the fluid formed was drained off and measured (volume b). The foam remaining was destroyed with amyl alcohol and measured also (volume c). Σ was calculated from the formula:
______t____
Σ = 2∙303 log b + c
c
For the two beers mentioned Σ was calculated from the following data:
b c Σ
original beer 32∙9 ml. 9∙7 ml. 122
de-foamed beer 2∙6 ml. 0∙72 ml. 117
Although the Σ-value for the de-foamed beer is lower than for the original beer, the difference must be considered insignificant. For both determinations 150 ml. of beer had were still 40 ml. of fluid remaining in the state of foam when the measurement began. For the de-foamed beer, however, only 2-5 ml. remained, so that the bulk of the foam had already been degraded in the sixty seconds before the measurement began. In this way, the increased stability of the last residues was measured, and nearly the same Σ-value as for the original beer of good foam qualities obtained.
When working with a standardized scheme of time, one risks comparing unequal parts of the process of foam degradation. Gray & Stone6 state that a beer might be diluted twenty times without changing Σ significantly, but when diluting more than twenty times they could not perform estimations anymore, because of lack of fluid remaining in the state of foam. Blom,3 however, found a decrease of foam stability on dilution and the present work completely confirms his experience. Thus, it seems that the results of Gray & Stone must be explained by assuming that the more they diluted the more they estimated the (increased) stability of the last foam residues.
Perhaps the only good application of the Σ-test is that used at the British Brewing Industry Research Foundation at Lyttel Hall. Here foam degradation is measured by noting the time that passes before between 25 and 75% of the original foam is degraded. Hence the start and the time of the measurement vary with the nature of the foam, and comparable results can be expected.
The method of Brenner4 has the merit of being original in its method of producing foam by means of a drop of pressure. Nevertheless, this method is perhaps too far removed from the practical way of foam appreciation, that is the pouring test in a normal beer glass. With this method, foam degradation takes place in a narrow tube of some cm. diameter. It has already been demonstrated8 that the diameter of a tube has a very pronounced influence on the foam stability to be estimated; the smaller the tube the higher the foam stability. So it would, especially with very narrow tubes, be important to standardize the tube diameter very carefully in order to get reproducible results. Besides, with a narrow tube, one gets an abnormally high foam layer and a completely different ratio of foam stability and foam adhesion forces to the glass wall than would be the case in a normal beer glass of 7 cm. diameter.
Considering all these objections against the usual methods, it seemed necessary to develop a new method of analysis. The procedure is as follows.
A calibrated funnel of 7 cm. diam. and 500 ml. content is used. In this funnel, 100 ml. of slightly de-carbonated beer is foamed up to the calibration of 400 ml. by means of a porous candle. At the moment this calibration is reached, the candle is removed and a stop-watch started; every 60 and later 30 sec. the beer is separated from the foam remaining. By interpolation, the time is estimated after which 95 ml. have drained back. This time is called the “foam time.” Working in sextuplicate with two funnels one can make an estimation in half an hour. The range is from 180-600 sec. for beers.
The reproducibility of the method was checked by measuring the foam time of the same beer 12 times in sextuplicate. By means of a variance analysis it was demonstrated that all the results were part of the same population, so that it was decided to estimate the accuracy of each mean value of a sixfold measurement by a calculation of the mean error. This mean error was found to increase with the magnitude of the foam time. For a normal foam time (about 300 sec), the mean error is usually about 6 sec. So for a level of probability of 95%, differences between two foam times start to become significant when exceeding 20 sec.
This estimation of the foam time is, like all the others, a drainage method, i.e., one only measures the amount of fluid formed back by a foam without measuring the amounts of foam in the course of foam degradation. Experience shows that, with strongly-hopped beers, more foam remains in the vessel after the measurement than with normal beers. The foams of these strongly hopped beers lose their liquid in a normal way, but form very persistent films. So, with a drainage method, one always estimates for these kinds of beer proportionately a too low foam stability.
It is, however, possible to correct this effect by measuring the second foam property, the “foam adhesion.” This is estimated by measuring the highest continuous foam ring in the cylinder six minutes after the foam production, and the result is expressed as the volume which a fluid would contain if reaching this ring. This number is divided by the original foam height (400), and the quotient multiplied by 100 is considered to be a characteristic for the foam adhesion to the glass wall. Thus, when a continuous ring is remarked near the calibration of 200 ml., this number is divided by 400 and the foam adhesion is fifty.
The range is from 0-80, and the accuracy is determined from the mean error. Any method measuring an amount of foam is less accurate than a drainage method that only estimates the fluid formed back by a foam. Hence, the estimate of the foam adhesion is less reproducible than the measurement of the foam time. Nevertheless, the foam adhesion can give valuable information about the behaviour of a foam in practice.
The foam time and the foam adhesion are simultaneously estimated in one experiment. Eventually, also, the foam density (percentage of fluid present in the foam) can be estimated without any extra work. This measurement is performed one minute after foam production by observing the foam volume. If there has, e.g., separated off after one minute 60 ml. of beer from the foam and the foam volume is 320 ml., it follows that this foam contains 100 less 60 = 40 ml. liquid, and the foam density is 40/320 X 100 = 12∙6. The opinion may be expressed, however, that this foam density is only of minor importance.
Results of Foam Investigation
The most important factors influencing foam stability and foam adhesion of beer are the high-molecular degradation products of the proteins and certain transformation products of the hop bitter substances.
As to the connection between the foam time and the high-molecular degradation products of the proteins (expressed in the Lundin fraction A), reference may be made to a paper recently published9. It was found that the foam time is strongly connected with the magnitude of the Lundin fraction A. When analysing 33 beers, a very high correlation was demonstrated to exist between these two magnitudes. A beer from which the high-molecular degradation products had been removed by saturation with magnesium sulphate showed only very poor foam properties.
This statement gave occasion to an investigation, seeking to find means to influence the amount of high-molecular protein matter. The first conclusion in this respect is that only minor differences are to be achieved by a variation of the malting process; it is to be doubted if the small differences which can be caused in this way are such as to have any influence on foam stability.
The most important factors to influence the amounts of high-molecular degradation products of protein are the use of adjuncts like sugar, rice and maize and secondly the use of chill-proofing substances.
At this Institute, all-malt brewings and brewings in which part of the malt had been replaced by barley were made. In this way, beers were prepared with a high Lundin fraction A and a high foam time. It was, however, remarkable that the beers so prepared sometimes had a poor foam adhesion. These experiences led to the conclusion that estimation of foam stability by means of a drainage method (Σ, foam time, collapse rate, etc.) gives only an incomplete prediction of the foam properties in a beer glass, and that it is necessary to complete the measurement with a foam estimation, such as the foam adhesion.
Influence of the Hop Constituents
It is practical experience that strongly hopped beers have good foam qualities. This was confirmed by preparing a number of beers with different quantities of hops. The results are to be found in Table I. It is to be seen from this Table that the first quantities of hop have already a marked influence on the foam properties, especially on the foam adhesion.
The surface viscosity was measured with the ring-oscillation method using an apparatus built according to directions of Sandegren, who has already performed an investigation concerning the relation between surface viscosity and foam stability.14 The surface viscosity is expressed as the quotient of subsequent amplitudes (the damping factor). The standard for distilled water was 1∙08. It is to be expected that there should be close connection between the surface viscosity as measured and the toughness of the foam lamellae. The increase of surface viscosity is probably the first cause of the improvement of the foam properties by hopping.
In order to examine which constituents of the hop cause this foam-stabilizing effect, extracts were made of hard resins, soft resins, humulone and lupulone.
Model experiments with peptone solutions led to the conclusion that all these substances are foam destroyers when simply added.10 After boiling, however, the peptone solutions “hopped” with humulone showed improved foam properties. So, apparently, the foam destroying humulone is transformed into some other substance (perhaps isohumulone) which acts as a foam stabilizing agent. The other hop constituents maintained their foam-destroying actions even after boiling.
When experimental brewings were made, it was completely confirmed that the transformation product of humulone has a foam stabilizing action. The beers prepared with this hop bitter acid had the better foam properties the more humulone had been added on brewing. Table II shows some of the results found. It is to be seen from this Table that the humulone gives rise to an increase of the surface viscosity and foam properties; especially, the foam adhesion is considerably influenced. The practical behaviour of the beer was in agreement with these figures. The beers with the highest concentrations of transformation product of humulone did not lose their heads like a normal beer. Even after a day there was still foam present, in the state of an evaporated solid substance.
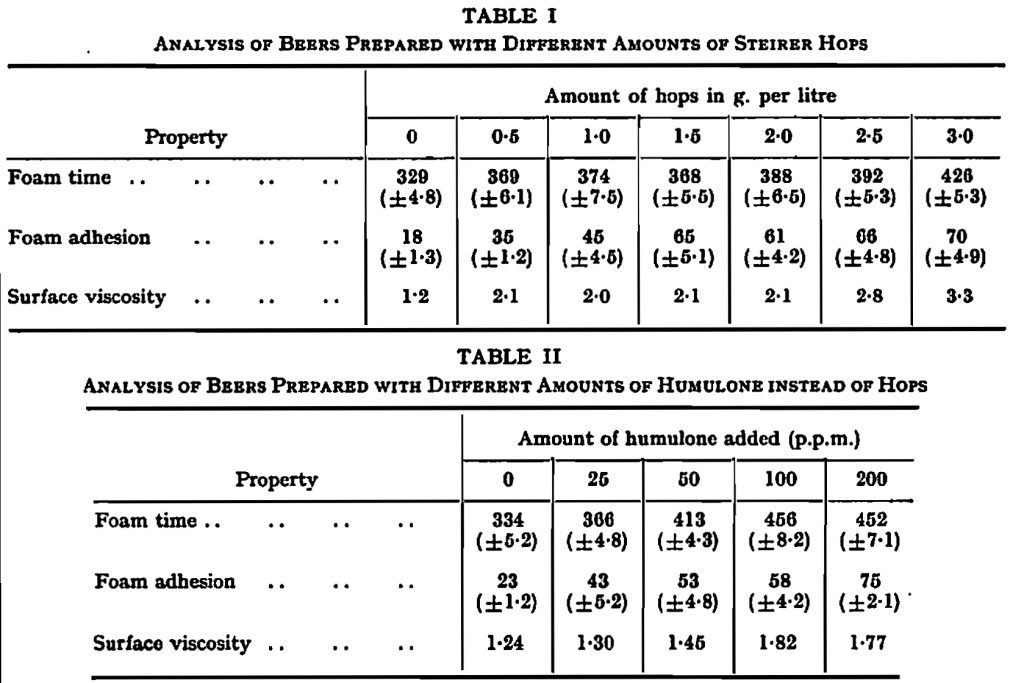
The beers prepared with hard resins, soft resins and lupulone had the same characteristics as the blank controls which obtained no hop constituent. Although these beers had a normal foam time, there was a nearly complete lack of foam adhesion. Nevertheless, a foam-destroying action of these hop constituents could not be made significant with beers, just as had been the case in model experiments with peptone solutions. Therefore, it is to be assumed that these substances are largely absorbed by the yeast during fermentation, so that the final concentrations are too low to exert a foam-destroying action.
The foam-stabilizing action of hopping seems to be attributable to the transformation of humulone into some other product. Although the only transformation product isolated so far is isohumulone, there is, however, no definite proof that there are no other products. Therefore it would be very interesting to investigate surface phenomena associated with isohumulone and with the other eventual transformation products of humulone.
Other Substances Influencing Foam Properties
In confirmation of the results of West, Evans & Becker,15 it was not possible to demonstrate higher alcohols and esters to be of any influence on the foam quality, at least in the concentrations in which these substances may be present in beer. This is in contradiction of the results of Sandegren & Säverborn.14 Ethyl alcohol, however, proved to have a negative effect just as had been found by Blom.3 The effect is small, and cannot be demonstrated by a variation of the ethyl alcohol content of 1%.
Barley fat often was thought to be a significant factor in head retention. Papers in this respect were published by Enders8 and by Hartong.7 At this Institute it was demonstrated indeed that barley fat is very active in its foam-destroying action. Even quantities of 0∙1 to 1∙0 p.p.m. had a marked influence on foam quality.
Every method of estimating foam stability which employs an artificial way to create foam, confirms the general fact that un-boiled wort has a markedly lower foam stability than hopped wort. By means of an ether extraction, this could be shown to be largely attributable to the presence of a fatty substance in the un-boiled wort. On boiling, the foam stability increased, apparently because of an adsorption of the fatty substance by the coagulating proteins. Nevertheless, significant amounts of barley fat were still estimated in the hopped wort, but not in the beer.
Because of the very small quantities of barley fat that may still have a foam-destroying action, it is hardly possible to say if this substance is a significant factor in the foam stability of beer. Accordingly, small-scale brewings were made with normal malt and with malt that had previously been extracted with ether in order to remove the barley fat. The beers prepared showed no differences. Hence, it is probable that barley fat, if present in significant amounts in the hopped wort, is largely absorbed during fermentation. It seems probable that this substance is not a significant factor influencing the stability of beer foam. The investigation is continuing.
Summary
1. The Σ-test of Ross & Clark may lead to false results.
2. A new method for the determination of the foam properties of beer is briefly described. A standard for the foam stability is the “foam time.” Additionally, for the exact characterization of the behaviour of a foam in practice, it was necessary to evaluate a criterion for the “foam adhesion” to the glass wall.
3. The foam time is largely influenced by the amount of high-molecular degradation products of proteins and the amount of hops used.
4. The foam adhesion is influenced largely by the amount of hops used.
5. This influence of the hops is to be attributed to the transformation undergone by humulone on brewing. Primarily, surface viscosity is influenced by this transformation.
6. Higher alcohols, esters and barley fat are not significant factors influencing the foam properties of beer.
References
1. Bikerman, J. J., Foams: Theory and Industrial Application. New York: Reinhold Publishing Corp., 1953.
2. Blom, J., Woch. Brau., 1036, 53, 11.
3. Blom. J. Woch Brau., 1036, 53, 25.
4. Brenner. M. W., McCully, R. E., & Laufer, S., Proc. A.M. Atner. Soc. Brew. Chem., 1950, 63.
5. Enders, C, Woch. Brau., 1941, 58, 107.
6. Gray, P. P., & Stone, I., Wallerstein Lab. Commun., 1940,3, 159.
7. Hartong, B. D., Int. Tijds. Brouw. Mout., 1941.
8. Kloppcr, W. J., Int. Tijds. Brouw. Mout., 1953-54, Nos. 5-6.
9. Klopper, W. J., Int. Tijds. Brouw. Mout., 1053-64, Nos. 7-8.
10. Klopper, W. J., Int. Tijds. Brouw. Mout., 1953-54, Nos. 9-10.
11. Kolbach, P., & Schilfarth, H., Brauerei, Wissensch. Beit., 1953, 6, 61.
12. Lienert, H., Schweis. Brau.-Runds., 1952, 63, 171.
13. Ross, S., & Clark, G. L., Wallerstein Lab. Commun., 1939, 2 (vi), 46.
14. Sandegren, E., & Säverborn, S., Int. Tijds. Brouw. Mout., 1948, Nos. 11-12.
15. West, D. B., Evans, R. F., & Becker, K., Brewers’ Digest, Mar., 1953.