STANDARD METHODS OF ANALYSIS – 1922
At the request of the Council of the Institute of Brewing, the Reports of the Malt Analysis Committee, which were adopted by the Council of the Institute on January 23rd, 1906, and September 30th, 1910, respectively, have been submitted to revision by the Standing Committee on Analysis, and the following is the revised Report:—
PALE MALTS
Purpose of the Methods
We have considered that the essential object of the uniform methods is the provision of a common analytical system to which, in cases of dispute, both buyers and sellers can resort, with the consequent avoidance of those differences in result which must arise out of the adoption of different methods. For this object it has been necessary to select methods which shall be simple, capable of rapid completion, and open to as few chances as possible for differences in result due to the “personal equation” of the worker. The methods would obviously also be available, between analysts and their principals and clients (whether brewers or maltsters) where both the principal or client on the one hand, and the analyst on the other, agree that these methods shall be used for commercial purposes; but they are in no way intended to supersede methods already in existence which analysts adopt when acting in an advisory capacity.
Limitations of the Methods
The methods described are not intended finally to stereotype malt analysis. It is obvious that, as fresh light is thrown upon the science of malting and brewing, these methods may require emendation, even for the special commercial purposes for which they have been formulated. The methods should be regarded, therefore, as only holding good for these purposes in the existing state of knowledge; and they are necessarily empirical, giving results which must be regarded as “values” and not as quantitative estimations of incontestable accuracy.
The Grist
A uniform grist is a first requisite to a uniform result, and the only mill known to us at the time of the first Report was the Seck Laboratory Mill. We have recently given careful consideration to a mill made by Messrs. Robert Boby, Ltd., Bury St. Edmunds, and we find that this mill is well suited for the purposes in question, and affords results in agreement with those given by the Seck Laboratory Mill. We recommend, therefore, that either of these mills should be used for all estimations, except that of moisture, where a coffee mill may be employed. The rolls must be set always at 0·5 mm., the setting being checked at frequent intervals by means of a 0·5 mm. feeler-gauge. In order to permit the passage of the gauge at all points along the rolls, it will be found necessary to revolve them slightly during the operation. In order to allow for loss in the mill, a quantity of malt, slightly in excess of that required for each determination, is to be weighed out separately and ground. Finally, the exact amounts of grist required for the various determinations are weighed out. It is not permissible to grind at the outset sufficient malt for all the determinations, and to weigh out the various quantities from this grist.
Extract
Fifty grams of ground malt are to be weighed out and mashed in a glass or suitable metal beaker of about 500 c.c. capacity with 360 c.c. of distilled water previously heated to 154—155° F. The beaker is covered with a clock glass, and placed in a water-bath, so that its contents are kept at 150° F. for one hour, the mash being stirred at intervals of about 10 minutes during this time. The whole mash is then washed into a flask graduated to 515 c.c., cooled to 60° F., and made up to the mark with distilled water at the same temperature. The flask is well shaken, and the mash filtered at once through a large ribbed paper into a dry vessel, the first 50 c.c. being returned.
When about 250 c.c. have been collected the specific gravity of the filtrate is determined at 60° F., compared with water at the same temperature. The excess specific gravity over water (= 1,000) multiplied by 3·36 gives the extract in brewers’ lbs. per standard quarter of malt.
If preferred, the mashing can be carried out directly in the 515 c.c. measuring flask, the distilled water having previously been heated to the temperature necessary to give a temperature for the mixture of 150° F. In this case the mash should be shaken at intervals of about 10 minutes.
Note.—If it is desired to determine the specific gravity at any temperature other than 60° the specific gravity bottle must be corrected for that temperature, and, in addition, the following correction must be made to allow for the difference in the coefficient of expansion of wort and water, viz.:—For each degree Fahrenheit that the solution is weighed above 60°, 0·02° is added to the specific gravity value, while for each degree Fahrenheit below 60°, 0·02° is deducted. This correction should only be considered (accurate for malt worts on which the specific gravity is determined between the temperatures of 60° and 70° F.
Example.—The specific gravity found for a hot mash malt extract when weighed at 66° F. = 1027·34°. 6 x 0·02° = 0·12o. As the temperature is above 60o F., this amount has to be added. The true specific gravity of the malt extract is therefore 1027·46.
Colour
For this determination the Lovibond tintometer is to be employed. The wort obtained in the extract determination, filtered as bright as possible without the use of any clarifying agent, is to be placed at once in a 1-inch cell, and its tint recorded in colour units of the series “52” glasses. The experiment should he carried out in bright diffused daylight (not direct sunlight), or with one of the recognized “daylight” lamps. In the latter case the operator should take steps to assure himself that the lamp used gives the same results as those obtained when using daylight. Whatever the source, the light must fall equally on both halves of the white plate, so that both fields—viz., the malt extract field and the standard field—are equally illuminated. To test this, the glasses and the cell should be reversed, and all results rejected when the figures do not agree, whichever side the cell is placed.
Moisture
About 10 grams of the sample (after having been thoroughly mixed) are ground as finely as possible in a small coffee-mill previously rinsed with the sample, and 5 grams of the ground malt are accurately weighed in a weighing bottle provided with a stopper, or in a beaker or crystallising dish provided with a watch-glass as cover. The weighing-bottle should be about 2 inches in diameter and about 1 inch in depth; the beaker or crystallising dish should also have a diameter of about 2 inches.
The vessel containing the ground malt is placed in a boiling water-oven for 3 hours. The stoppers or covers should meanwhile be kept on the top of the oven. At the end of this time the stoppers or covers are replaced and the closed vessels immediately transferred to a desiccator and weighed as soon as cold.
The percentage result should be returned to the nearest first decimal.
Precautions
1. Nothing but malts should be in the drying oven.
2. As the coolest part of the oven is nearest the door, none of the malts should be placed close to the door.
3. The ventilator in the door of the oven must be open and the orifice at the top of the oven kept free.
4. The products of combustion from the burner must be prevented from entering the oven. This may be accomplished by suspending a sheet of metal or asbestos (of the same width as the oven) by means of one edge bent at right angles inserted between the base of the oven and front top of the stand, any remaining crevices being packed with asbestos. The shield should extend at least 15 cm. below the base of the oven.
5. While the malt is being dried in the oven, the oven door is on no account to be opened.
6. The vessels containing the malts must be kept on the floor of the oven.
7. In order to prevent furring, it is advisable to use distilled water for heating the oven.
8. A control determination of moisture-content in a stock sample of malt should be made as above with each batch.
9. The sulphuric acid in the desiccator should be renewed at frequent intervals.
Diastatic Activity (Lintner Value)
First Method
Twenty-five grams of ground malt are to be extracted with 500 c.c. of distilled water for three hours at 70″ F., stirring well every half-hour. The extract is then filtered bright. 3 c.c. of the filtrate are allowed to act on 100 c.c. of a 2 per-cent. solution of soluble starch (see Appendix I, p. 782) at 70° F. for an hour in a 200-c.c. flask. 10 c.c. of N/10 caustic alkali are then added in order to stop further diastatic action, the liquid cooled to 60° F., made up to 200 c.c. with distilled water at the same temperature, well shaken, and titrated against 5 c.c. of Fehling’s solution, using ferrous thiocyanate as indicator (see Appendix I, p. 782).
The titration is carried out as follows:—
Five c.c. of Fehling’s solution (see Appendix I, p. 782) are accurately measured into a 150 c.c. boiling flask, and raised to boiling over a small Bunscn flame. The converted starch solution is added from a burette in small quantities, at first 5 c.c. at a time but gradually reduced as the end point is approached to ½ c.c, the mixture being kept rotated and boiled after each addition until reduction of the copper is complete. This is ascertained by rapidly withdrawing a drop of the liquid by a glass rod, and bringing it at once in contact with a drop of the indicator on a porcelain or opal glass slab.
The results are calculated by the following formula:—
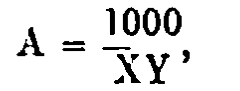
in which A equals the diastatic activity, X equals the number of c.c. of malt extract contained in 100 c.c. of the fully diluted starch conversion liquid, and Y equals the number of c.c. of the latter liquid required for the reduction of 5 c.c. of Fehling’s solution.
The above method (using 3 c.c. of malt extract to 100 c.c. of 2 per cent, soluble starch solution) is not accurate for malts having a diastatic activity exceeding 50 Lintner; in the case of such malts the relative volume of malt extract must be loss, say 2 c.c., or, for malts of the highest diastatic activity, such as are frequently used by distillers and vinegar makers (i.e., malts of over 80 Lintner), an even smaller volume of extract must be taken.
Second Method,
A cold water extract of malt is prepared as described under “First Method.”
Into each of ten test-tubes, placed in a suitable stand, are introduced 10 c.c. of a 2 per-cent. solution of soluble starch. A gradually increasing amount of the bright malt extract is added to each tube in the series, i.e. 0·1 c.c. to the first, 0·15 c.c. to the second, 0·2 c.c. to the third, and so on up to 0·4 c.c. After this the volume added might increase by 0·1 c.c. in each tube up to say 0·7 c.c. in the tenth tube, beyond which it will rarely be necessary to go. The tubes are then thoroughly shaken, and allowed to stand in a water bath for an hour at a constant temperature of 70° F. At the end of this period, 5 c.c. of Fehling’s solution are added to the contents of each tube, the tubes again shaken, and the whole series placed in a boiling water bath for 10 minutes.
The tubes are now observed, and the degree of reduction noted. Some of the contents will be found to be over-reduced, as shown by their yellow colour, and others under-reduced, as shown by their blue colour. The two consecutive tubes—one of which is slightly over-reduced and the other slightly under-reduced—are selected. The point of exact reduction will lie somewhere between these two, but sometimes, of course, one tube will be neither blue nor yellow, showing that the number of c.c. of malt extract added brought about exact reduction, in which case the number in question is noted.
In cases where the reduction lies between the first and second tubes it is advisable to repeat the determination, using a more dilute extract.
The diastatic activity is calculated by dividing the volume of extract required for complete reduction into 10. Thus, if the point of complete reduction is estimated to lie at 0·37 c.c, the diastatic activity will be:—

Cold Water Extract
Twenty-five grams of ground malt are to be digested with 250 c.c. of distilled water containing 20 c.c. of N/10 ammonia {i.e., 20 c.c. of N/10 ammonia made up to 250 c.c. with distilled water) for three hours at 70°, stirring about three or four times during this period. After filtering, the specific gravity of the bright filtrate is taken at 60° F., compared with water at the same temperature. The excess specific gravity over water (= 1,000) divided by 3·86 and multiplied by 10 gives the cold water extract per cent.
The percentage of ready-formed soluble carbohydrates may be approximately arrived at by subtracting 4 from the value obtained for the cold water extract per cent.
General Recommendations as to Checking Work
In order to check his results, we recommend that the analyst lay in a stock at the commencement of the season of a certain malt, and that each working day the extract, diastatic activity and moisture of this stock malt be estimated, no results for other samples being accepted when the results with the stock malt show any appreciable divergence from the normal values. The stock malt should be taken from one kilning, and not from a heap or bin containing several kilns; it should be pale and not high dried, and it should have, been made from a choice quality of barley. These conditions are requisite to secure the maximum of uniformity in the stock malt. It is obvious that this malt must be kept in an air-tight vessel.
Statements of Results
The results, expressed to the nearest first decimal place only, except in the case of Diastatic Activity, which should be recorded only to the nearest integer, are to be stated according to the following scheme :—
Extract per standard quarter, brewers’ lbs.
Moisture, per cent.
Diastatic activity (Lintner value).
Colour (10 per cent, wort, 1-inch cell, “52” series Lovibond).
Cold water extract, per cent.
Ready-formed soluble carbohydrates, per cent.
Appendix I
Preparation of Soluble Starch
Purified potato starch is to be digested for seven days with dilute hydrochloric acid of specific gravity 1037°, at a temperature which ought not appreciably to exceed 60° F, and the mixture should be thoroughly shaken daily. The operation is conveniently carried out in Winchester quart bottles, 1 lb. of starch and 1,000 c.c. of acid of the above strength being suitable quantities for each such bottle. It is washed very thoroughly by decantation, at first with tap water, and later on with distilled water, until the wash water is free from chloride. It is collected on a filter paper in a Buchner funnel, and pumped as dry as possible. During the washing the reaction should be tested at intervals by means of sensitive litmus paper, and the washing must be continued until the starch is neutral. It should be transferred to new unglazed porous plates, and dried at a moderate temperature (110° F.) as quickly as possible. When the moisture-content has been reduced to about 15 per cent, the starch is ground in a porcelain mortar and rubbed through a fine hair sieve.
Starch Solution.—In determining diastatic activity, the starch must be dissolved in boiling water at the rate of 2 grms. of the starch per 100 c.c. of water ; the solution is then cooled to 70° F. for use; 10 c.c. of this solution should not reduce more than 0·1 c.c. of the Fehling solution.
The water used in making up the starch solution must be pure. Commercial distilled water frequently contains ammonia, nitrous acid, and other impurities, which appreciably influence diastatic conversion, and, therefore, the result. The water should be redistilled with addition of a little potassium permanganate and caustic soda (the solution used in water analysis is suitable), in a glass apparatus provided with a trap, until the distillate is pure and neutral to litmus solution.
Preparation of Fehling’s Solution
(a) Copper Solution.—Recrystallised copper sulphate (69·2 grams) is to be dissolved in distilled water and the solution made up to 1 litre at 60° F. with distilled water.
(b) Alkaline Tartrate Solution.—Rochelle salt (346 grams) and caustic soda (130 grams) are to be dissolved in about 600 c.c. of distilled water, the solution cooled and made up to 1 litre at 60° F. with distilled water.
The two solutions are to be kept separate, and equal volumes mixed for each day’s work, from which mixture the volumes specified in the analytical methods are measured out at 60° F.
Preparation of the Indicator
One gram of ferrous ammonium sulphate and the same quantity of ammonium thiocyanate are dissolved in 10 c.c. of water at a moderate temperature, say at 120° F., and immediately cooled; 2·5 c.c. of concentrated hydrochloric acid are then added. The solution so obtained has invariably a brownish-red colour, due to the presence of ferric salt, which latter must be reduced. For this purpose zinc dust is the most satisfactory reagent to employ, and a mere trace is sufficient to decolorise the solution if pure reagents have been employed.
When kept for some hours, the indicator develops the red coloration owing to atmospheric oxidation. It may, however, be decolorised by the addition of a further quantity of zinc dust, but its delicacy is decreased after it has been decolorised several times. For practical purposes the indicator may be too delicate, and it is recommended to prepare it the day before it is required for use, as it gives the best results after the second decolonisation.
Recommendations as to Sampling
It is obvious that samples sent for analysis should be fairly representative of bulks, and this requires the more care when the bulks (whether from makings or deliveries) are large, and when the malt contains any appreciable number of hard corns, or when there is any marked irregularity in curing.
In the case of deliveries, samples should be drawn from at least 10 sacks if the consignment amounts to over 100 sacks, or if the delivery be smaller, then from 10 per cent, of the number of sacks. The sample should be drawn with a spear from a depth of at least 6 inches.
In sampling from heaps or bins, surface samples should be avoided as in the case of sacks, and 6 to 12 portions should be withdrawn from various depths, including the bottom.
These bulk samples should be put into a large tin kept specially for the purpose, and should be well mixed; a smaller tin (of at least a pint capacity) is then filled from the larger one, and sent to the analyst, the remainder being reserved in other similar small tins if the analysis is to be checked. The lids of all tins containing samples for analysis should fit well, and it is desirable as an additional safe-guard in those cases in which special airtight tins are not employed to affix gummed paper round the junction of the lid and tin. Malts are really better sent in clean, carefully dried, stoppered bottles. Stoppered beer bottles answer the purpose.