COMMUNICATION (July / August 1947)
by R. E. Essery
(of the Institute of Brewing Research Laboratory, The University of Birmingham) and
R. Gane and T. N. Morris
(of the Low Temperature Research Station, Cambridge)
A Description of Experiments on the Partial Freezing of Beer and the Separation of Ice From the Concentrated Liquid Phase
When dilute alcoholic liquors are cooled to temperatures below 0° C, ice crystallizes out and can be separated by means of a basket centrifuge from the liquid phase containing the alcohol and other solutes. As the temperature is lowered, ice-formation and the concentration of solutes in the liquid phase increase steadily, and the present work was undertaken to obtain data for the application of these principles to the preparation of concentrated beer.
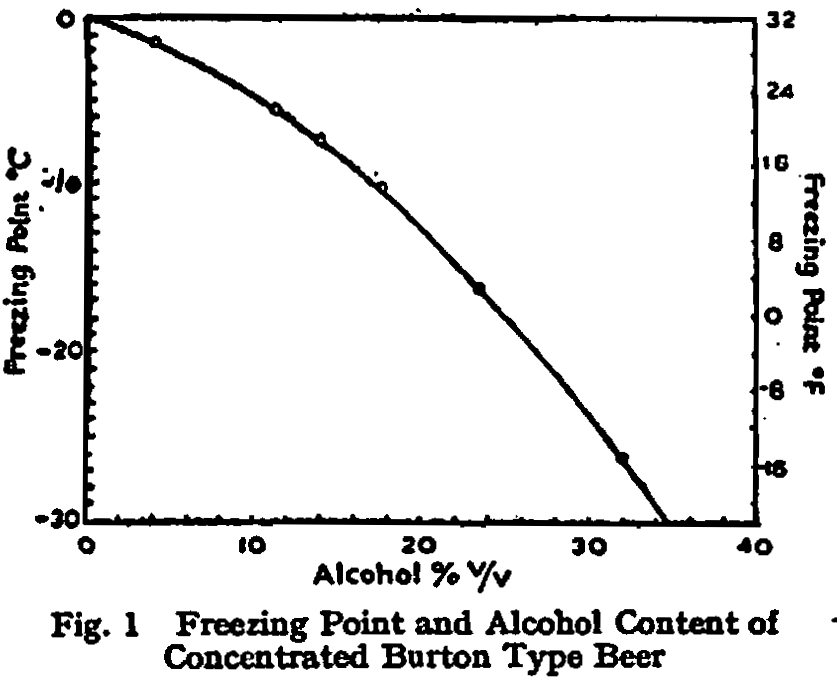
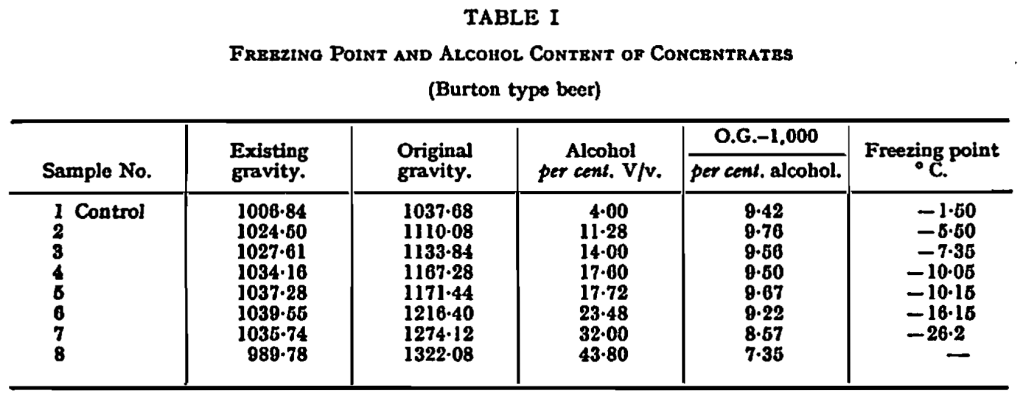
Freezing Point and Concentration.—Preliminary tests with concentrates obtained by freezing and centrifuging half-gallon lots of beer provided the data for Table I and Fig. 1, which show the percentage-concentration of alcohol by volume in the liquid phase after equilibrium has been established with ice, by the usual freezing-point methods, at temperatures ranging from -1·5° C. (29·3° F.) to -26·2° C. (-15·2° F.). The table also shows the gravity of the concentrates, the gravity of the wort which would, in the ordinary way of fermentation, yield such a beer and, in column 5, the figures for the ratio:
Original gravity — 1,000 / per cent alcohol
An examanation of column 5 shows that freezing was accompanied by a certain degree of deposition of solutes other than alcohol, which increased rather sharply between -10° C. (14° F.) and -16° C. (3·2° F.). More complete analysis of sample No. 8 of Table I showed that the loss of solute was greatest in the case of nitrogenous constituents (expressed as total nitrogen), and least in the case of mineral matter (expressed as ash); but the variation was not large and, for practical purposes, the solutes other than alcohol may be regarded as having been frozen out as a single entity.
As a result of these observations, -12° C. (10·4° F.) was chosen as a convenient temperature at which beer might be frozen in tests on a larger scale without a serious loss of any desirable constituents. Freezing tests were, however, also carried out at -20° C. (-4° F.) (see Table IV).
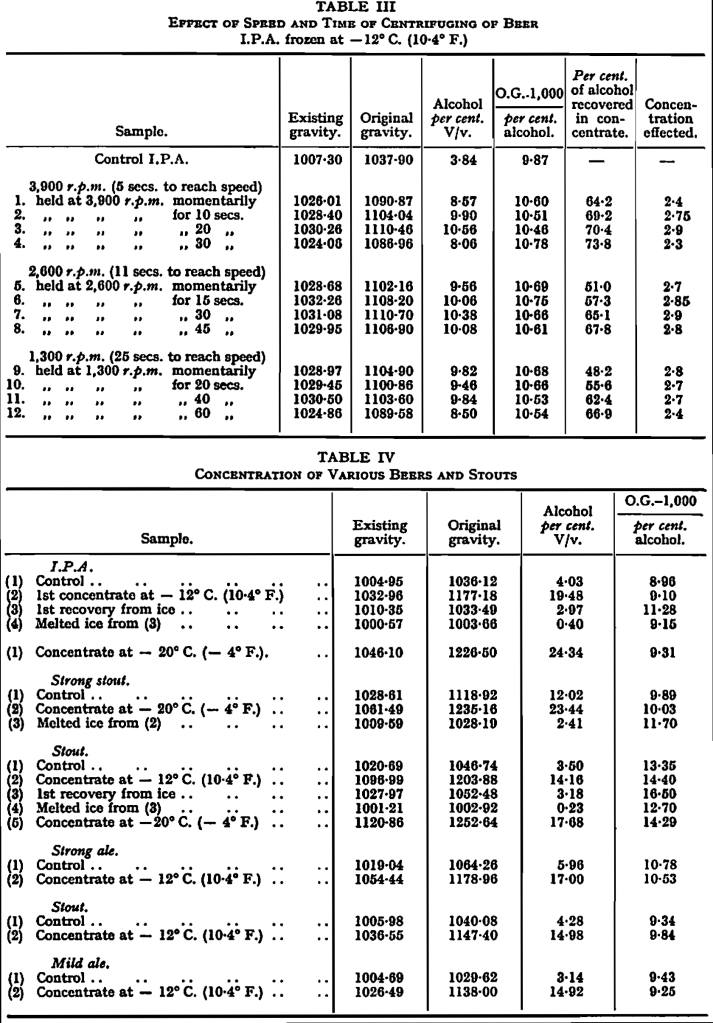
The optimum conditions for working the laboratory centrifuge were also determined in the preliminary tests, and the results of runs at different speeds and for different periods are seen in Table III. Account had to be taken of the percentage of alcohol in the concentrate from a given run and the percentage recovered, the best result being obtained by running at 3,900 rpm. for 20 sec. At other speeds and times there was either incomplete recovery or greater dilution of the concentrate through thawing.
Details of the Method of Collecting the Concentrated Beer.—Fined cask beer of the Burton type, brewed in Cambridge, was frozen with occasional stirring to break up the ice, in tins holding 1,700 gns, each in rooms at -12° C. (10·4° F.) and -20° C. (-4° F.). After 6 days the tins were brought singly into the laboratory at room temperature and the contents transferred to a basket centrifuge capable of holding a charge of about 500 gm. of ice at a time. The basket attained a speed of 3,900 rpm. (corresponding to g X 800) in 6 sec., and was allowed to run at that speed for 20 sec. The concentrates at each of the test temperatures were bulked separately and the ice formed at each temperature was also bulked separately in a large bowl, stirred to bring about some thawing and re-centrifuged to remove a dilute residue of entrapped beer which, in large scale working, would have been re-frozen with fresh beer. The ice was then clean enough to be discarded.
In one test 4 gallons of beer were frozen in an ordinary tapered ice-pail immersed in a brine tank at -10° C. (14° F.), and then crushed and centrifuged as above. The strengths of various concentrates and of liquors recovered from the ice are shown in Table IV. A comparison of this table with Table I shows that the concentrates obtained by freezing beer at -12° C. (10·4° F.) and -20° C. (-4° F.) and centrifuging in the laboratory at room temperature, as described, corresponded respectively to liquors in equilibrium with ice at about -10° C. (14° F.) and -16° C. (3·2° F.), the difference being due to the thawing which occurred in the process of centrifuging. In large-scale working or in working under different conditions these relations would be somewhat different.
The Quality of the Products.—The highly concentrated liquids obtained from Burton type ales, strong ales and stout were excellent drinks in themselves. India Pale Ale and bitter beers were, however, too bitter to drink in the concentrated form. When diluted with water to a concentration equal to that of the original liquid the taste was rather flat and watery; when carbonated, however, the diluted concentrate closely resembled the original liquid.
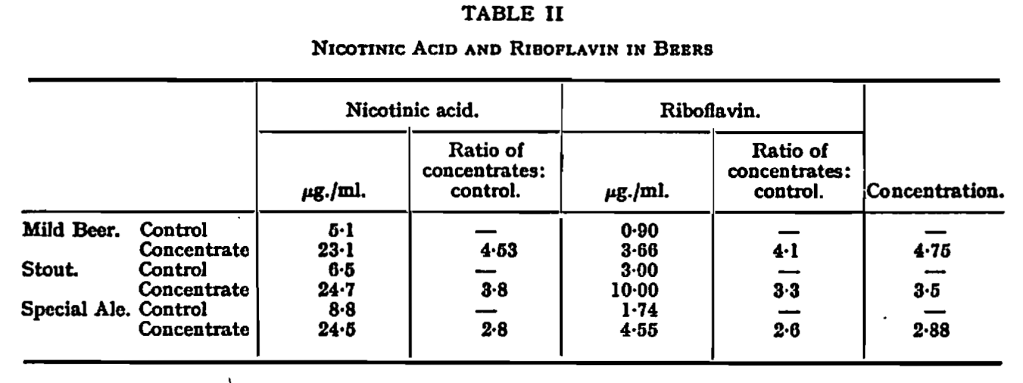
Vitamin Content of the Concentrated Beer.— Samples of various beers and the concentrates prepared from them were compared for vitamin contents by Dr. Kodicek of the Dunn Nutritional Laboratory, Cambridge. The results showed that concentration increased the content of nicotinic add and riboflavin in about the same proportions as it increased that of alcohol. There was thus practically no loss of these vitamins during concentration. The figures for vitamins obtained by Dr. Kodicek (see Table II) are somewhat higher than those usually cited for beers, but it is comparative, not absolute, values which are of interest in this paper.
Chemical Carbonation.—Attempts were made to carbonate the concentrate, after dilution approximately to the gravity of the original material, by the use of lactic acid and sodium bicarbonate, as a possible alternative in special cases, to carbonation by means of gaseous carbon dioxide.
It was found that quantities of these materials, sufficient to give adequate carbonation, imparted a distinct saline taste to the beer, which would readily be detected by a critical drinker, but that otherwise the beer was not undrinkable. Even when the quantities of lactic acid and bicarbonate were reduced to one-half of those required for satisfactory carbonation, the salinity was still perceptible to a taster drinking blind.
In view of these findings, experiments on chemical carbonation were discontinued.
Testing Experiments
Beers Brewed at a High Gravity.—To prepare beers of strengths comparable with the concentrates, worts of sp.gr. 1107 and 1108-7 were hopped for 2 hours at 2½ Ib. per barrel, and pitched at ¾ lb. per barrel, the former with a Burton-union yeast, and the latter with an attenuative skimming-yeast, temperatures of pitching being 14·7° C. (58·5° F.), and 14·4°C. (68° F.), respectively. The samples were fermented until the gravity had fallen to 1035° and 1040° respectively. Samples withdrawn at 1040° and 1045° respectively, and diluted half-and-half with carbonated water, tasted sweetish, but more like a normal beer than a “strong ale.” On racking, the beers were diluted with 1½ volumes of water, conditioned, fined and carbonated. These tasted like a sweetish, under-hopped ordinary beer, that fermented with skimming-yeast being preferred to that fermented with Burton-union yeast.
A second experiment, in which the fermentations were faster, gave similar results. Portions of these strong beers were stored at 5-6° C. (42° F.). and at room temperature. After 12 months, that preserved at 5-6° C. was smoother and more vinous in character than when first brewed, but still resembled an ordinary beer rather than a “strong ale.” The other sample was accidentally destroyed before it could be tasted.
Beers Concentrated by Freezing.—Samples of fined Burton-type cask beer, brewed in Cambridge, were concentrated by freezing, and similarly preserved (a) without further treatment, (b) after conditioning and carbonating for a week at 12·8° C. (66° F.), under a pressure of 10 lb. of CO2.
(a) Unconditioned Concentrate.—Shortly after concentration this, while vinous in the concentrated state, tasted, on dilution and carbonation, more like an ordinary beer than a “strong ale.” After 6 months at 5-6° C, the samples were smoother and more vinous than before but, on dilution and carbonation, the flavour remained more like that of an ordinary beer. A similar result was obtained after a further 4 months at the same temperature, but the flavour showed a distinct trend further towards the “strong ale” type. Further tastings could not be carried out.
(b) Conditioned Concentrate.—Four bottles of this product were prepared, and stored for 16 months at 0° C. (32° F.). They were then separated and stored for a further 2½ months, A at 0°C, B at 15° C. (59° F.), C at 20° C. (68° F.), and D at 26° C. (77° F.).
With the undiluted samples, of five tasters, one could detect no difference, while all the others favoured D as the best. Two agreed that A was more bitter than D, while one of these described D as being of the “strong ale” type, and A as being of the type of a normal beer.
The concentrates were diluted in the proportion of one of concentrate to three of soda-water, to correspond with the original unfrozen material. A and B were decidedly turbid, while C and D remained quite bright. C and D showed a decidedly stronger head than A and B. On tasting, the dilution was found to be too great, and further portions were diluted in the proportions of one of concentrate to two of soda-water. These dilutions were found to be sufficiently alcoholic, but deficient in bitterness and palate-fullness. The tasters decided that D gave the most satisfactory result, and all agreed that this would probably be accepted as a reasonable drink “in the absence of anything better.” Here again, one observer detected the “strong ale” type of flavour in D.
The O.G. of the concentrate was 1170°, so that a dilution of one to two would give an “O.G.” of 1067° for the diluted material. The taste of the latter indicates that, in view of its “OG,” freezing seems to cause a considerable loss of palate-fullness, and some loss of bitterness.
These results indicate that the esterifications, and other changes, which lead to the production of the characteristic “strong ale” (perhaps better described as “old ale”) flavour, are checked, but not completely inhibited, by storage at low temperature, both in beer brewed at a high gravity and in beer concentrated by freezing. Loss of samples prevented any very definite final conclusions, but the evidence seems to suggest that, to limit the production of the “strong ale” flavour, storage should be at as low a temperature, and for as short a time, as possible, with, possibly, a short storage at a temperature between 15° C. and 20° C, just prior to dilution and carbonation, to improve brilliance and head.
All the concentrated beers kept well, but all showed a tendency, greater in some cases than in others, to throw out a brown, finely divided flocculum, which appeared to be, in the main, of the nature of a protein-tannic acid complex. This is very similar to the deposit which is normally formed when a strong beer is matured, and it is evident that the freezing process does not eliminate this phenomenon, which is probably connected with the abovementioned loss of bitterness and palate-fullness. This question would need attention if the process is employed on a large scale.
The concentrates were tasted by a panel of experts and analyzed by Miss June Perret and Mr. J. Rowland at The Institute of Brewing Research Laboratories.
Mr. H. J. Shepherd, of the Low Temperature Research Station, determined the freezing points quoted in Table I and was responsible for the centrifugal separations.
The joint work described above was carried out by the Institute of Brewing Research laboratory of the University of Birmingham and the Low Temperature Research Station of the University of Cambridge and the Department of Scientific and Industrial Research at the request of the Ministry of Food, who were interested in the possible value of the method in connection with certain war-time problems.