MEETING HELD ON THURSDAY, 12th JANUARY, 1899, AT THE GROSVENOR HOTEL, MANCHESTER.
Mr. C. H. Hill in the Chair.
The following paper was read by Dr. A. K. Miller :—
The Manufacture and Use of Brewing-Sugars in America.
by G. W. Rolfe, A.M., and Geo. Defren, M.S.
Before proceeding with this subject it will be necessary to define what is meant by the term “glucose” as there is great variation in the use of this word and consequent confusion in the minds of many.
In England and France, apparently, the term is synonymous with dextrose and starch-sugar. In America the latter product is invariably called “grape-sugar,” “glucose” being applied exclusively to the thick, viscid syrup resulting from the partial hydrolysis of starch by acids, a familiar commercial product. In classifications of organic chemistry, the custom seems universal to use the word as a name for a sugar group, although quite recently the word “glycose” is being substituted in the interests of definiteness. In this paper we shall, in referring to the commercial products resulting from the acid “conversion” of starch, speak of solid sugar as “starch” or “grape-sugar,” confining the term “glucose” to the syrup previously mentioned.
The following outline of the chemistry of the acid conversion, or hydrolysis, of starch will make the details of manufacture more intelligible. When a thin starch paste is heated with a dilute acid, a change takes place much resembling in general appearances that caused by diastase, so familiar to you. Investigation would show this difference:— The acid-converted syrups are made up of three components—maltose, dextrose, and dextrin; whereas, as you well know, in the malt (diastase) converted worts only two of these are found—maltose and dextrin. (We are speaking of those bodies resulting from the starch alone.) The comparative conditions of formation of these carbohydrates in the two cases would be seen to be radically different. The conditions of production of maximum maltose in worts, never exceeding about 80 per cent, on dry substance, are defined by somewhat complicated procedure of heating at varying temperatures, considerable dextrin always remaining unchanged. In acid-conversion, on the contrary, within wide limits at any rate, the composition of the syrups follows fixed laws irrespective of the nature or amount of the hydrolysing acid or temperature conditions, these influencing the rate of change only. The amount of change at each stage of the conversion is shown by the following diagram (A), giving the percentage curves of each constituent, the stage of conversion being measured by the change in optical polarisation of the syrup. It will be noticed that the original starch paste is pure dextrin or, to be more exact, corresponds in its properties to the primary carbohydrate we call dextrin, which must not be confounded with the commercial products or the alcoholic precipitates so termed, these being in reality complicated combinations. As the hydrolysis proceeds, the dextrin continually diminishes, maltose being rapidly formed, dextrose also appearing almost immediately, but increasing at first very gradually. At what may be considered the middle stage of the hydrolysis, the percentage of maltose reaches a maximum and then begins to disappear like the dextrin, but with more rapidity. Finally, dextrose alone remains in the solution, except some products of decomposition, present in very small amounts unless the heat is excessive, or the acid too concentrated. The point of conversion for the manufacture of “grape-sugar” is then clearly defined by this diagram. In the case of “glucose” two conditions are imperative :— (1) The amount of dextrin must be sufficient to prevent the separation of crystallised sugar when the product is concentrated to 45° Bé, or about 84 per cent. (2) Under similar conditions dextrin should not separate out. These conditions limit the conversion stages to those represented by that part of our diagram lying approximately between the polarisation figures, 150° and 100°. Commercial glucoses in the market vary in polarisation from [a]D 386 = 145° to 120°. As the greatest consumption of glucose is in the manufacture of candies, jellies, and syrups, its composition has been determined by the demands of these trades. The use of glucose in beers, extensive as it is, takes a very small proportion of the total output. On this account there are practically two grades of glucose on the market, leaving out of consideration goods which differ only in concentration—(1) Mixing (syrup) glucose, with a conversion of [a]D 386 = 120° to 130° and confectioners’ goods of [a]D 386 = 130° to 140°. The best confectioners’ goods are commonly about [a]D 386 = 135°. There is by no means a rigid stand in the market, however, in fact many manufacturers grade according to the perfection of refining, the clearer, whiter glucose, independent of its conversion, being specially treated for confectioners’ use. Jelly goods differ from mixing glucose merely in concentration, although imperfectly refined candy glucose is often worked up into this grade, as a slight turbidity or tint is of no consequence in jelly work. Mixing glucose is the kind usually bought by the brewer. As we have stated, the composition may vary through wide limits, the proportion of dextrin, for instance, varying in extreme cases about 100 per cent. In short it should be well understood that commercial grading of a glucose is no criterion of the relative proportion of dextrose, maltose, or dextrin, matters of considerable moment to the brewer. The grading of starch-sugars being more plainly conformable to the brewer, their chief consumer, we need not consider them here.

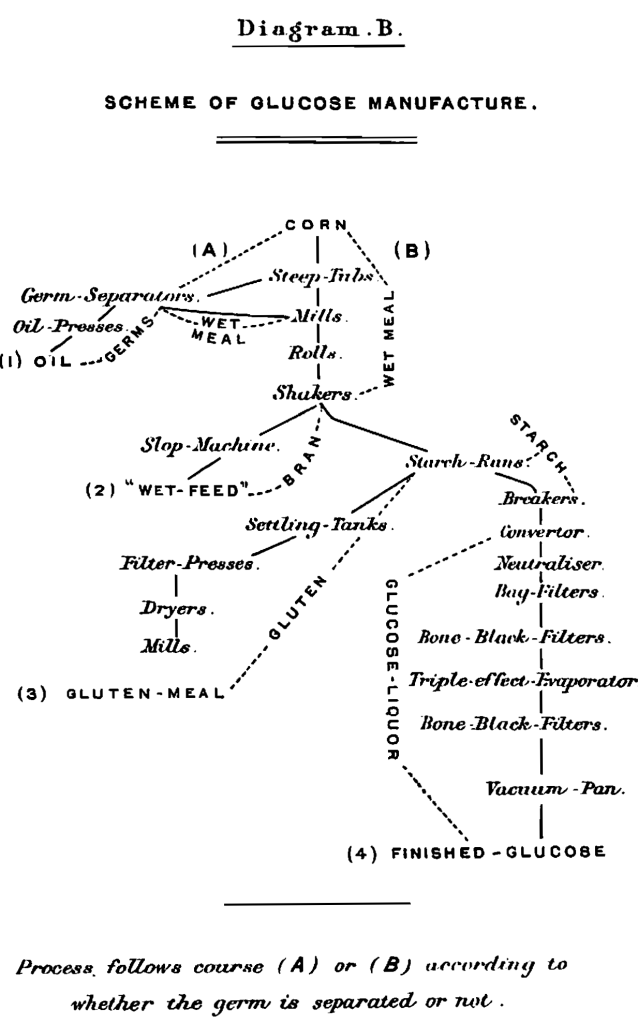
The time at our disposal will permit only the most superficial description of the manufacture of brewing-sugars. In general the process consists of three parts : (1) Separation of the starch; (2) conversion; (3) refining. (The diagrammatic scheme before you (B) will assist you in following this description.) All kinds of what is known as No. 3 or 4 corn (maize) are used,—the market being the main consideration. This corn is taken from the cars or the elevator of the works to the steep-tubs, which hold 2,000 bushels or more. In steeping, water at 150° F. is used at first, then the steep is allowed to cool till a temperature of about 90o F. is reached. Sulphurous acid is used to prevent putrefaction and assist softening. The steeping lasts 3—5 days. The separation of the starch consists of (1) grinding the wet grain, mixed with water, and softened by steeping; (2) separating the starch-grains from the woody fibre and germ by washing through sieves of bolting-cloth, rapidly shaken; (3) settling out the starch from the gluten by subsidence while passing over gently inclined runs (“tables”). The grinding is done so that the starch-grains are set free but not ruptured, an important point when yield is considered. The germ is removed separately, in many factories, by a patented process. This is accomplished by coarse grinding, and running the grain, mixed with much water, through a long trough, the mass being agitated slowly. The germ, which floats, is carried off by one channel, the rest of the grain by another. This separation of the germ is an important improvement, since the oil which is contained in it can be readily obtained as a by-product, and the quality of the glucose is much improved by its removal. The grain from the separators is ground and washed on the sieves (“shakers”) in the usual manner, and the separated liquor sent over the runs. The thin, highly diluted gluten is allowed to settle, pumped through filter-presses, and the dried cake, which contains over 30 per cent, of protein, sold for cattle-feed.
The starch collected on the runs, and containing about 50 per cent, of moisture, is now mixed with water to a thick cream of about 20° Bé preparatory to conversion. Conversion is carried on in large copper boilers at a steam pressure of 30 lbs., hydrochloric acid being the converting agent, the amount used being about 0.0006 of the weight of the starch.
In some factories sulphuric acid is still used as the hydrolyzing acid. In the manufacture of candy goods and certain hard sugars it seems to have some advantages. Oxalic acid has been used for the manufacture of fine candy goods, but the advantages gained are questionable. In the manufacture of grape-sugar a much larger proportional amount of acid is used, in some cases up to 1 per cent, or more on the weight of starch. The point of complete conversion is usually controlled by the disappearance of the dextrin precipitate when the liquid is poured into alcohol.
In glucose conversion, the acid is mixed with about 50 times its bulk of water, and run into the converter. Steam is then turned on till a pressure of 30 lbs. is obtained. This pressure is maintained while the starch-milk is pumped in, which takes about half an hour. Heating is continued after this for 40 minutes or more, “Dirty” starch, containing much gluten, increases the time of conversion 10 minutes or more. The degree of conversion is entirely controlled by iodine tests. By daily practice workmen become quite expert in making these tests, yet from week to week there is apt to be considerable variation in composition when tests are not checked by chemical control, which is rarely the case, as far as this point is concerned. As soon as the conversion is completed, the liquid is blown out into the “neutraliser,” where the alkali, usually sodium carbonate, is added. In many factories it is the practice to cool the liquids considerably before neutralising, but this seems unnecessary when the alkali is added properly. This neutralisation is a process of exceeding delicacy. Any excess of acid or alkali, even exceedingly small, will seriously affect the refining operations which follow. Properly neutralised liquid should show only the acidity caused by carbon dioxide or the weakest vegetable acids. The properly neutralised liquid is clear and of a bright amber colour, but contains large flocculent masses of coagulated gluten, which, in a test tube of ordinary size, form a layer about half an inch thick. “When the proper point of neutralisation is attained, this layer is greenish-drab, owing to the precipitated iron.
The refining process is in general similar to that of cane-sugar. The analogy is quite close in the case of the solid grape-sugars. In the case of glucose, however, there is a radical difference of principle which must not be overlooked. In the refining of glucose the purification must be carried to great lengths, at least as far as colour and appearance are concerned. All bodies affecting these characteristics must be absolutely removed from the liquid, or bleached in it, since there is no mother liquor in which they can be deposited, as in the case of a crystalline sugar. On this account the refining of glucose is a much more delicate process than that of sugar.
Neutralisation is an important put of the refining, as on the thoroughness with which this is done depends how successfully the albuminoids, calcium and iron salts are removed. As in sugar refining, the precipitated matter is removed by bag-filters, this often being supplemented by a press filtering. In the case of highly refined glucoses, precipitants are sometimes used, such as alum. The tendency seems to be for manufacturers not to push this part of the refining to greatest advantage, but rather to depend on a liberal use of bone-black to do much of what, it would seem, could be accomplished by less expensive means in preliminary clarification. The bone-black treatment is quite complicated, not only in the details of filtering but in the preparation of the black. Since the slightest trace of alkali in contact with the hot liquor will produce a brown stain of caramel, removable only to a limited extent by bone-black, the black itself must be freed from all traces of ammonia or caustic lime by a careful “tempering” with hydrochloric acid or some similar treatment, and as careful a washing to remove soluble salts of iron and calcium formed. The glucose liquors are, as a rule, put over the bone-black twice; first at their original concentration, about 18° Bé., and again, after concentration, to 28—30° Bé., the denser syrup going over the freshly tempered black. The revivifying of the black is carried out on lines similar to those of cane-sugar refining.
The “heavy liquor” goes directly from the filters to the vacuum-pan in most modern factories. Formerly a preliminary filtration was necessary to remove the calcium sulphate which separated out, but with the use of hydrochloric acid conversions and neutralisation with soda, this is avoided. In the final concentration sulphites are added in amounts varying from 0.008 to 0.050 per cent. SO2.
The function of these sulphites is as follows :— (1) To prevent oxidation and consequent coloration in the final concentration due to formation of caramel-like bodies, and sometimes ferric salts; (2) to bleach; (3) as a prevention of fermentation of the lesser concentrated finished products, as the thinner mixing syrups; (4) in candy goods, as a preventive of oxidation in the candy kettle. Confectioners’ goods are much more heavily “doped” than others. There is a tendency among manufacturers to excessive use of sulphites, in all classes of goods, a matter of some interest to the brewer.
The refining of grape-sugar liquors is practically identical with that of glucose. The concentrated syrups are drawn off into pans or barrels and allowed to solidify, a “seed” of crystallised sugar often being added to facilitate crystallisation. Anhydrous grape-sugar is made in a similar way from a syrup which is refined at lower concentrations throughout the process in order to obtain a purer product. In this case the “seed” is selected with the greatest care from absolutely pure anhydride, all hydrated crystals being scrupulously excluded. The crystallisation is complete in about three days, when the sugar is purged in centrifugals. The purged liquors are often worked up into the “climax” sugars, a dark product familiar to most of you. Of late years these sugars have been further refined as a white product, and even made directly by merely solidifying the anhydrous sugars as a concrete without purging. As you well know, these sugars also are often put on the market as “chips.” Glucose syrups are usually made at six concentrations—39°, 41°, 42°, 43°, 44°, 45° Beaumé. Mixing goods are usually finished up at 39° or 41° Bé; 41o mixing glucose is the grade usually sold to brewers. The higher concentrated products are confectioners’ or jelly goods, the former being characterised by greater perfection of refining and large amount of sulphites. These are often whitened with methyl-violet.
A few words as to the manner of taking concentration determinations may not be amiss here, as there seems to be considerable confusion of ideas on this point. This is not surprising when we remember that there are over 30 different Baumé scales in existence. The glucose manufacturers do not use the same scale as the sugar refiners, who employ Gorlach’s modification. The glucose scale is practically identical with that used by the alkali manufacturers, and has the following conversion formula d = 144/144o Bé. Owing to the great viscosity of glucose, the readings are taken not at the standard temperature of the instrument (60° F.) but at 100°. The determination is somewhat difficult and tedious. The slightly warmed glucose is poured into a cylinder, preferably of glass, which is placed in a water bath at 100° F. At the end of half an hour or so the glucose will have reached the temperature of the bath, and the air bubbles escaped. A Baumé spindle, reading to fifths, is then cautiously lowered into the glucose and allowed to come to equilibrium, which in the more viscous samples takes some minutes. With care the determination can be made on a Westphal balance. All samples of glucose should be tested for density, as the viscosity in goods of different conversions varies to a marked degree. A low converted sample of moderate density will apparently have much more “body” than a high converted glucose much more concentrated. Because a glucose runs thick when a barrel is opened is no indication that it is 43o Bé.
As, apart from concentration, the quality of commercial glucose is largely judged by its appearance, it will be interesting, possibly, to consider briefly some of the turbidities and colorations of the commercial products, their causes, and actual influence on the quality of the glucose. A well refined glucose is practically colourless and clear. If a white glass cylinder is filled with glucose, the colour of the sample can be seen, as well as any turbidity. If the colour is a pure white, the sample is dyed, us can be proved by exposing to the light for a few days, although this colouring is rarely done so well that a close inspection will not reveal the violet tint. As all glucoses darken slightly on exposure to the light, the colour balance soon becomes disturbed, and the presence of the dye is made more evident. If no dye is present, the glucose, unless quite turbid, will show some colour, usually green or yellow. These tints are almost invariably present, and seem to be caused by traces of iron salts and vegetable colouring matters. They are of little consequence, except as indicators of the thoroughness of the refining, and hence the removal of albuminoids and oil. These impurities affect the flavour of the product. Dyeing, therefore, likewise only concerns the brewer so far as it may be the means of compelling him to pay a high price for low-priced goods. A reddish-brown discoloration is the result of excess of alkali, either through imperfect neutralisation or defective treatment of bone-black, as a rule.
As to turbidity, cloudiness caused by faulty conversion, separation of dextrins in one case, or sugar in the other, is, in these days, of rare occurrence. A smoky appearance is often caused by bone-black dust, or in some cases from iron sulphide, when a large quantity of new black is used in refining; these, of course, are the results of improper preparation of the black. White cloudiness is caused either by calcium salts or by organic growth due to fermentation. The former may be sulphate or phosphate. Sulphates in goods converted by hydrochloric acid, arc in the main introduced through use of impure acid, or in sulphite liquors; phosphates from excess of “tempering acid,” or incomplete washing of the black. The clouds due to fermentation, which naturally are more common in goods made in hot weather, arc usually the result of storing thin liquor (in process) at too low a temperature. Of course the fermentation organisms can easily be identified by the microscope. A quick way of identification is to acidulate the sample with hydrochloric acid, when the ferment cloud remains un-dissolved. It is evident that the presence of foreign ferments possibly, even probably, in a state of activity, is a matter of no little concern to the brewer. Already attention has been called to this by Dr. Kriegcr, in an article in the American Brewer, of September, 1896, on “The Use of Turbid Glucose in the Brewery.” The author very properly advises a thorough boiling of such glucose in the wort. He, however, confines himself to finished goods which have fermented, presumably after they have left the factory, but often glucose to all appearances free from ferment, will show presence of organisms as described. It would seem decidedly doubtful whether these organisms are entirely killed by the moderate temperature of the vacuum pan and the light treatment with sulphite, which such goods often get.
While referring to this article of Dr. Krieger’s, we may also call attention to an important paper by the same author, on “The Manufacture of Starch Products,” in the Zeitschrift fur Spiritus-Inditstrie, 1894.
It is evident that the calcium salts have no influence on brewing in the quantities present in glucose.
The valuation of the solid starch sugars is practically based on their dextrose content. Whiteness of late years seems to be more of a desideratum than formerly; hence the practice of dyeing is becoming common. The principal mineral impurity objected to is iron. This is rarely present in more than traces. A delicate test for iron in sugars or glucoses is cochineal. Sulphites must be first removed, and the solution made neutral or faintly alkaline. If iron be present, the pure crimson of the cochineal gradually passes into violet. We think in the past brewers have been more alarmed than injured over the iron in glucose. In the first place iron is only present in glucose in traces, never enough to cause damage in brewing: and secondly, if by any chance iron be present, it is precipitated in the form of tannate, which is removed in the “hop-back,” or through settling in the settling-tuns.
Moreover, most brewers will find that their brewing waters generally contain more iron salts than does any glucose, and also if they use iron mash-tuns, or iron rakes in these tuns, some iron will always pass into solution by the action of the small amount of lactic acid in the malt, which would also be considerably more than that which occurs in glucose.
Our attention has been called to an iridescence on the surface of wort containing much glucose in the copper. This was undoubtedly due to traces of corn-oil, which occasionally is present in finished goods.
We have omitted mention of any method of determining degree of conversion of glucoses, as we have already published this in detail (J. Amer. Chem. Soc., October, 1896).
Solutions of grape-sugar show the same characteristic colorations and turbidities, in a greater or less degree. Usually they show caramel tints, owing to the decomposition products, especially inferior goods.
No exact statistics are at hand, by which we can state with positiveness just what is the amount of brewing sugars made and consumed in the United States. The glucose industry uses about 35,000,000 bushels of corn (maize) annually in making about 1,000,000,000 lbs. of glucose.
As far as we can estimate, from 15 to 20 per cent, of this is consumed in brewing in the United States; about 160,000,000 lbs. goes to England, a considerable portion of which, we understand, is used in candy-making.
Apart from the foregoing brief account of the manufacture of commercial glucose and grape-sugar, it is the object of the present article to show how these products can be, and in many cases are, used by lager and ale brewers. The authors are wholly cognisant of the fact that there is yet a considerable amount of ignorance prevailing among many brewers concerning the uses of commercial glucose and grape-sugar, and are on that account prepared for adverse criticisms from many quarters.
It is the object of the present article to try to clear away a small quantity of the many cobwebs which arc today hampering the brewing trade. Ignorance of the uses and advantages to be obtained from an advanced scientific work may be excusable at times, but in these days of commercial activity the law of the survival of the fittest holds with peculiar tenacity, and only those concerns which try to keep in the van of scientific development are generally successful, although we are frank to admit that we are acquainted with some breweries which have done a profitable business in the hit and miss style. It was only a matter of good fortune that they succeeded as they did. On the other hand, it can be truly said that a superficial knowledge of scientific requirements is as harmful as, and we warrant in many cases even more so, than a total lack of knowledge would be in the uses of exact methods; and this is especially true of those brewers who, having a slight smattering of chemistry and biology, assume that they know the whole subject, and, instead of their being successful, make their affairs so complicated that they finally flounder in the mire, and then have the so-called beer doctor help them out of their sad plight. It is only where a judicious combination of scientific knowledge and practical experience are combined that the best results are obtained.
There is a tremendous popular prejudice against the use of any malt substitute in brewing, especially among those unacquainted with brewery requirements, and this is one of those cases in which “where ignorance is bliss ’tis folly to be wise.” One truly humane pamphlet, The Book of General Membership of the Ralston Health Club, in speaking of glucose says : “Glucose, a perverted form of corn, is prevalent in beer and other drinks, and especially in soft caramels and cream, and in syrups, jellies, and similar things. Although derived from a nutritious food, it is in a perverted shape, and to glucose may be attributed the rapid spread of Bright’s disease.”
There is even a marked prejudice among the brewing fraternity to the use of glucose as a substitute, yet these same brewers use large quantities of corn grits and corn flakes, forgetting for the moment that most glucose is really nothing more than common corn starch hydrolyscd by acids under steam pressure. This has been already explained in the first part of this paper, and shows that dextrose, in addition to dextrin and maltose, occurs in this glucose, whereas diastase converts only these two latter from starch, the hydrolysis ending before the maltose is decomposed into dextrose.
One brewer known to us, in speaking of 43° M. glucose, said that 43° M. glucose meant 43 per cent, of maltose, and he is the head of a large establishment and doing n good business at that. This lack of knowledge may not hurt him much, but when we had shown him that considerable of the glucose he was then using, instead of being 43° M. as he supposed, which for this degree of gravity contains 17 per cent, of water, was really 41o M. glucose and held 22 per cent., he saw where he was losing a matter of 10 cents on every 100 pounds, which was quite an item if continued for any length of time. It will be a great satisfaction to us if, when this paper has been read, the brewer gets no more benefit from it than to exercise a greater degree of caution in buying one thing and getting another considerably inferior. We know that the practice exists very extensively where 41° M. glucose is sold as 43° M., and would herewith caution the brewers to keep a sharp look out for this method of deception.
Very little can be said on the use of sugar substitutes. What follows is probably known to almost every brewer, and we will therefore sum up such facts pertinent to the matter in hand in as concise a manner as possible, believing that those who have had more experience in the use of glucose will be lenient with us for any unintentional omission of important matters, and hoping that the subject matter may prove instructive to those brewers who have not had very much experience with the chemistry of this compound and the varying conditions which may be presented by its use.
Malt substitutes may be divided into two classes: those in which the extract is obtained by a mashing process with malt; and, second, those in which the extract is already formed: First, maize, rice, wheat, prepared corn, and corn flakes, &c. When using any of these substances for brewing purposes, certain proportions of them are either added to the malt in the mash-tun directly, or else the starch in these substitutes is first gelatinised in a separate vessel and rendered soluble by a little ground malt, and afterwards added to or sparged over the goods in the mash-tun. Second, refined cane-sugar, invert-sugar, glucose and grape-sugar, and refinery syrups.
These substitutes are used for some one or more of the following reasons:—
- Ease of manipulation.
- Economy.
- An improvement in, or changing the character of the finished beer.
- Quick clarification.
- Flavour.
- Priming.
- Pale beers.
1. “When an ordinary pure malt wort has been prepared according to the established rides, it is simplicity itself to add glucose to this, either in the underback, copper, or hop back. The whole is mixed by boiling or stirring, and then fermented as usual. The amount of glucose or grape-sugar to be added to the malt wort should be based upon the same considerations as when using corn grits, corn flakes, and other similar substitutes. Glucose comes in barrels highly concentrated, and at once gives a maximum return for material used. No extract is lost through non-saccharification of any starch, as is the case with corn grits, &c. The only care to be exorcised is that the barrel be steamed out, thus insuring complete removal of all glucose which might be attached to the staves. This is very much simpler than making a corn mash. It takes but a few minutes, requires no careful manipulation or expensive apparatus, and consumes but little steam.
Another great advantage of glucose substitution is that any lack of gravity of the wort may easily and quickly be brought to any desired stage, an advantage the brewer keenly appreciates, when through some mishap or change of malt he finds his wort several tenths of a per cent below the accustomed gravity.
2. From an economical standpoint glucose and grape-sugar are sometimes used, although at times the prices of these substitutes have been higher than malt or prepared grain, which would contain equivalent amounts of extract. A little more than a year ago American glucose was selling pound for pound of extract at less than half the price of malt. Today the market conditions have placed the prices of glucose and malt on a more uniform basis, although it is still a decided economy for the brewer to use glucose at present quotations in combination with malt.
If brewers made a few calculations upon the price of the extract in glucose in competition with raw corn grits, prepared corn flakes, &c., they would easily save money at various times. There is no economy in purchasing 43° glucose at $l.60 (6s 6d) when corn grits sell at $0.96 (4s.) per 100 lbs., and yet we have brewers here who tell us they use glucose because it is the cheapest malt substitute obtainable, where if a little calculation were made, surprising results would appear.
3. Yet there are many brewers who do not use glucose solely for reason of economy. Glucose and grape-sugar arc used very extensively in America for the purpose of improving the finished beer. If used judiciously, there is no question about these substitutes improving this article. Glucose contains only a very small percentage of nitrogenous matter, and it has always been believed that the smaller the percentage of nitrogen in the wort the sounder would it be. This is a delightfully easy explanation, and we regret to say we do not believe the statement entirely. Although this may apply to small amounts of sugars used, and may have an important hearing on the subject, the amount of nitrogenous matter may be reduced to such small proportions that the beers are really thin in taste, and are derisively styled “corn beer,” “glucose beer” &c, with considerable justice. Some nitrogenous (proteid) compounds in beer precipitate after standing or when cooled. In itself this may do no harm in affecting the taste, yet very few people to-day wish beer placed before them so clouded with albuminoid turbidity that it is almost impossible to look through it. A consumer’s esthetic feeling should be taken into account as well as his taste.
It is here, then, that the addition of glucose is an advantage. A well refined glucose contains almost no nitrogenous matter, and, when added judiciously, commensurate with the quality of malt used and the kind of beer desired, the results obtained well warrant the employment of this substitute. The wort obtained from poorly manufactured malt will not permit the use of much substitute, whereas a fine malt wort may have as high as 30 to 40 per cent., or even more glucose added to it without endangering the quality of the beer. Many lager beer brewers use as much substitute as this in their wort and derive good results. On the other hand, the beer must have a certain full bodied taste and retain its foam-keeping qualities, and one of the easiest ways of failing in these requirements is to add too much grape-sugar or glucose.
We have had some very marked examples of this placed before us at various times, and are not at all surprised at the adverse criticisms made on the beers. We know of one case where the amount of glucose was 65 per cent, of the entire raw material used, and although what was called “beer” resulted, people are usually fastidious enough not to desire any more of it. When one calls for a glass of beer one wants something other than a fermented glucose solution.
Many brewers say they wish their wort brewed in at 12, 13, or 14 per cent Balling. If it possess this percentage of extract their wishes are satisfied. The composition of the wort and the dependent degree of attenuation of fermentation troubles them but slightly. Their conscience is satisfied if, as they declare, their beer has so much extract, &c, &c. Yet beer may have a large percentage of extract and still be thin in taste; whilst we also know of beers with 1 or 2 per cent, less extract, which possess a better head of foam, taste more full bodied than heavier articles, and are considered better in quality in every respect. Each brewer must use such amounts of glucose or grape-sugar as he finds his trade will warrant.
There is another matter which should be touched on here, and that is, the choice of kind of glucose, as the following examples will indicate. A malt wort mashed at a low initial heat and brought up slowly to a tap heat of 163° F., with a gravity of 10 per cent Balling, if mixed with a glucose of a specific rotatory power of [a]D 386 = 145° until the gravity was 14 per cent Balling, and then fermented, it would have about 1½ per cent, more extract in the finished beer, and conversely less alcohol, than if the malt wort were mixed with glucose of [a]D 386 = 120° specific rotation, and brought to the same gravity. The difference would be even more marked if the finished wort were made up of 4 per cent, grape-sugar of a rotation of 70°, in which case the amount of extract in the finished beer would be small, and the percentage of alcohol correspondingly high.
This is accounted for as follows:—A nominally converted glucose of .a rotation of [a]D 386 = 145° contains, in dry substance, about 40 per cent, dextrin, 17 per cent, dextrose, and 43 per cent, of maltose. The dextrose quickly ferments, whilst only a part of the malto-dextrins would be decomposed, these complex dextrins not fermenting until broken down by the action of the invertase of the yeast. A glucose of 120° specific rotation would, on the contrary, only contain 21 per cent, of dextrin, 34 per cent, of dextrose, and 45 per cent, of maltose on the dry substance; whilst a highly converted glucose—in other words; commercial grape-sugar—of 70° specific rotation, would have its dextrin reduced to about 2 per cent., whilst the amount of maltose, dextrose, and decomposition bodies, gallisin, &c, would constitute the remaining 98 per cent. You will thus recognise the importance of an intelligent examination of the glucose you intend to use, for on this selection the composition of your finished beer will largely depend, especially when the amount of substitute is added with a liberal hand. A glucose should therefore be selected and its delivery insisted upon, which will give a definite result. We are optimistic enough to believe that the day is coming when the brewer will state whether he wishes the glucose of an absolute specific rotation of 145° or of 120°. One could be made about as easily as the other, and though they may look alike, still the results obtained by using them side by side would be decidedly astonishing in the finished product
The mixing and confectioners’ trades have been the means of placing the glucose of today on the market, and the brewers simply take what is set before them without having any voice as to what type of goods they desire.
We will now touch upon another possibility. By employing higher mashing heats, and adding a higher converted product, i.e., grape-sugar, we obtain a finished beer which possesses nearly the same percentage of alcohol and extract as a beer made by low mashing heats, to which low converted glucose of 130—135° specific rotation has been added. This is explained by the fact that high mashing heats cause the diastase of malt to act on starch in such a way that the amounts of maltose formed are small in comparison with dextrin, as has been so clearly demonstrated through the brilliant researches of Messrs. Brown, Morris, Heron, and O’Sullivan, &c. If grape-sugar be added to this, the amount of fermentable material necessarily increases, the percentage of alcohol would also increase, whilst the extract would diminish. An analogous explanation would apply to the use of low mashing heats and low converted glucose of, say, 130° rotation.
To use sugars without reducing the stability of the wort they should possess nearly the same compositions in malto-dextrin and fermentable sugars as the original wort made entirely from malt. Stock ales can be satisfactorily made in this way, although we know of many ale brewers who, in using anything but grape-sugar as a substitute, would probably be greatly appalled if they even considered the use of any glucose syrup at all, according to the above recommendation. There is no question about grape-sugar, 70° rotation, being most advantageously employed in the manufacture of stock ales, as the dextrose percentage in this substitute is very high and easily fermentable. Stock ale, to keep well, should be highly attenuated, i.e., the beer should contain a large percentage of alcohol and a small percentage of extract, and that of a composition not easily broken down by the ordinary brewers’ yeast. We know of brewers here who, in using a low initial mashing heat, employ a small amount of glucose of 130° rotation, and report no harmful effects; but the majority of ale brewers prefer to use a higher mashing temperature and add more grape-sugar instead, a process which gives eminently satisfactory results.
The ale and lager beer brewing industries have been aided within the past few years by the use of so-called “carbonators.” These artificially charge the “still” ale or beer with carbonic acid gas, which gives it “life.” Beer that has been thoroughly fermented, cooled, filtered, carbonated, and then bottled, though a comparatively rare article today, will surely become common before many years go by. A carbonated stock ale made by ordinary methods, and with the addition of grape-sugar or high converted glucose, should make a very palatable article, and we hope to see more advancement in this line.
4. What has been said in the preceding paragraphs has a bearing on the fourth condition, that of clarification. Clarification in sugar-brewed beers i.e., beers in which high converted grape-sugar is added, is quicker than in all malt beers, probably on account of the reduction of the viscosity of the wort, owing to the substitution of the dextrin and malto-dextrins, by fermenting the sugars, which causes a more ready elimination of the yeast, as well as a more advantageous action of artificial finings when used in the “chip” casks in lager beer manufacture, and even in ales which are today treated after the main fermentation, very much as ordinary lager beer.
Bright, sparkling, draft ale is very much desired at the present day, and it is essential to have the fermenting wort clarify quickly. This is easily accomplished by simply employing grape-sugar or high converted glucose of 100—110° rotation. The dextrose ferments rapidly; the solution becomes less viscid; the yeast is eliminated more easily, and filtration becomes simpler. This is very advantageous in bottling beers, which should be free from turbidity, in that the addition of glucose or grape-sugar has reduced the amount of nitrogenous matter to a minimum, thus requiring less to be removed by cooling.
It was formerly claimed that the yeast would degenerate if glucose were put into malt wort, on account of diminishing the nitrogenous matter. This has been proved entirely fallacious by the practical experience of everyone in the business. It has been shown that yeast can ferment considerably more sugar in wort than it is called upon to do on the average, although we frankly admit that a limit must eventually be reached beyond which the yeast will not act. Yeast does not decompose sugar into alcohol and carbonic acid gas indefinitely without some additional nourishment; and this necessarily implies a certain quantity of nitrogenous matter in the fermenting wort. Quick clarification by the addition of grape-sugar should not be too diligently sought, to the detriment of the life of the yeast form.
5. A pure malt beer is prized by many beer drinkers. It generally possesses a good head of foam, is full-bodied, and is characterised by a distinctive flavour. It was at one time believed that foam-holding capacity and full-bodiness were due to dextrinous compounds, but this idea has died out. It is today considered that peptones and albumoses are the real causes of foam-keeping and full-bodied taste, and if mashing be carried out so that peptones are formed in maximum quantity, the amount of sugar substitute can be considerably increased without hurting the beer. The distinctive character of stock ale is obtained by fermenting wort to a low gravity, thus increasing the amount of alcohol to a maximum, and reducing the extract. The percentage of alcohol in beer depends on the amount of fermentable sugars present. If this be high, the alcohol percentage is high, and vice versa. Grape-sugar consists almost wholly of dextrose, which is entirely fermentable, and when this is added to malt wort and fermented, the alcohol percentage rises, and gives to the stock ale its peculiar tart flavour. This is the real reason for its employment in stock ale. Mild running ales, pale ales, and pale lager beers, on the contrary, have generally a lower alcohol percentage, and it is in their preparation that glucose syrup is added as a substitute.
6. The subject of “priming” is pretty well known to the average brewer. So-called “kräusening” in lager beer consists in injecting about 10 per cent, of a vigorously fermenting wort into the storage lager beer in the chip cask; whereas “priming” is simply an addition of a grape-sugar solution to the storage ale, which is then bunged down. The sugar present ferments, thus forming alcohol and carbonic acid gas, the latter impregnating the ale, giving it the requisite “life”.
Grape-sugar is preferable to glucose syrup in priming, on account of the quick fermentation, and no long, drawn-out decomposition as is the, case with glucose.
7. Glucose and grape-sugar are very extensively used in the United States for pale beers. The American people have become so accustomed to drinking light beers, that a very great demand has been created for them. Dark beers are not much desired. In manufacturing pale beers the brewer demands malt kilned at a low temperature, and adds to this a considerable quantity of substitute, be it corn grits, corn flakes, or other corn product, rice, &c. Glucose is added directly to the wort in the copper, and then fermented as usual. The ordinary malt wort possesses a very considerable amount of colour, even if the malt employed has been dried at a very low temperature. A glucose or refined grape-sugar solution, on the other hand, is practically colorless, and when added to the malt wort necessarily gives the article a much lighter appearance. An attractive beer is as essential as a well tasting one, and where a good appearance and a fine flavour are combined, the brewer’s labours are accomplished.
In conclusion, we would say that we hope these somewhat fragmentary notes on the manufacture of glucose and grape-sugar, and their application to brewing purposes, will accomplish their sole object; that is, a more extended discussion and study of these useful materials, carried on with the conscientiousness and thoroughness which their importance deserves.
There was no discussion following the paper.
Mr. Hill moved a vote of thanks to the authors of the paper, and also to Dr. Miller for reading it, and said they would all agree that it had been very interesting. The resolution was carried unanimously.
Dr. Miller acknowledged the compliment. He said that somewhat over a year ago he had some correspondence with Mr. Rolfe, concerning some of his (Mr. Rolfe’s) researches, and he took the opportunity to ask him to write a paper on some subject of interest to brewers, to be read before that Institute. Mr. Rolfe at once expressed the pleasure it would give him to contribute a paper in conjunction with his colleague, Mr. Defren; but owing to great pressure of work, the paper had been delayed, and had only recently been received. It was, however, he was sure, none the less welcome on that account, and in the name of the authors, he thanked the members present for the way in which they had received the paper.