MEETING HELD AT THE GROSVENOR HOTEL, MANCHESTER,
ON THURSDAY, 27th APRIL, 1899.
Mr. J. Arthur R. Greaves (Vice-President) in the Chair.
The following paper was read and discussed:—
The Nutrition of Yeast. Part II
On the Odour of the Gases evolved during Fermentation.
by Arthur L. Stern, D.Sc, F.I.C.
In the first portion of this investigation it was observed that the gases evolved during fermentation always have an unpleasant odour, due to the presence of sulphuretted hydrogen or some of its derivatives. Many observers have recorded the production of similar unpleasant odours from alcoholic fermentations; in fact, it is common knowledge to workers in the fermentation industries. No doubt this phenomenon may be produced by a variety of causes. The experiments here recorded illustrate one particular cause. They were carried out so as to be strictly comparable with those described in Part I.
The yeast used in any one experiment was produced by growth in a malt wort (sp. gr. 1056), and the gases evolved during fermentation were never evil-smelling; on the contrary, they always had a pleasant aroma.
The substances in the experimental solutions before the addition of the yeast were dextrose, asparagine, potassium phosphate, magnesium and calcium sulphates. As sulphates are the only sulphur compounds present, it is evident that the sulphuretted hydrogen, or its derivative, must he derived from them. If, however, the sulphates be removed before fermentation, the experiments recorded below (Table I) show that the fermentation is very incomplete, and the yeast crop and nitrogen assimilation small.

The letters A and B indicate two different inorganic nutriments; both contain approximately the same amounts of potassium, magnesium, calcium, and phosphoric acid, but B also contains sulphate (J. Chem. Soc. 1899, 203). Comparing experiments in which either nutriment A or nutriment B with its sulphate removed was used (Experiments 1 and 2) with the other experiments where nutriment A with the addition of sulphuric acid or nutriment B was used (Experiments 3, 4, and 5), it will be evident that the sulphate performs a very important part as a yeast food, and that in its absence the functions of the yeast are materially restricted.
When it is considered that this yeast, when fermenting malt worts, produces no unpleasant smelling gases, whereas it does when fermenting the artificial solution used in these experiments. It appeared possible that malt worts might contain some sulphur compound which served as a yeast food without the production of any sulphuretted hydrogen or allied compound. If this be so, it might be possible to find a sulphur compound of known composition which could replace the sulphate in the above experiments, and be equally satisfactory as a yeast food, yet without the production of any sulphuretted hydrogen.
To this end, comparative experiments were made in which different sulphur compounds were substituted for the sulphate. The following substances were tried:—Thiocarbamide CS(NH2)2, potassium sulphocyanide KCNS, sulphonal (CH3)2C(SO2·vC2H5)2, sulphur S, sodium thiosulphate Na2S2O3, and the results arc recorded in the following table:—

It is evident the thiocarbamide, potassium sulphocyanide, or sulphonal were unable to take the place of sulphates as a yeast food. (Experiments 6, 7, 8, 9, and 10.) In these experiments the fermentation of the sugar is incomplete, the crop of yeast, the nitrogen assimilation, and the percentage of nitrogen in the yeast are low. In Experiment 12, where thiosulphate was substituted for sulphate, the fermentation was fairly complete, and the crop of yeast, the assimilation of nitrogen, and the percentage of nitrogen in the yeast are practically the same as when sulphates were present. In Experiment 11, in which free sulphur was substituted for sulphates, the figures are not quite as satisfactory, but still almost as good, a result which is probably due to its insolubility. In this experiment, the gases evolved during fermentation contained sulphuretted hydrogen, whereas those evolved in the one where thiosulphatc was added (12), smelt strongly of mercaptan. In Experiments 6, 7, 8, 9, and 10 no unpleasant odour was perceptible during fermentation.
These results lend no support to the hypothesis advanced, and some other explanation of the production of the unpleasant odour must be sought.
The conversion of sulphate into sulphuretted hydrogen involves the removal of a large amount of oxygen. Is it this that the yeast is seeking?
Experiments were made to test this. In these the sulphates were replaced by other acid radicles containing a large proportion of oxygen. Chlorates and nitrates were tried, and the results are shown in the following table:—

It is evident these results lend no support to this hypothesis. Another possible explanation then presents itself. There might be some peculiarity in the constitution of asparagine which caused the production of the disagreeable odour when the substance was being used as a yeast food.
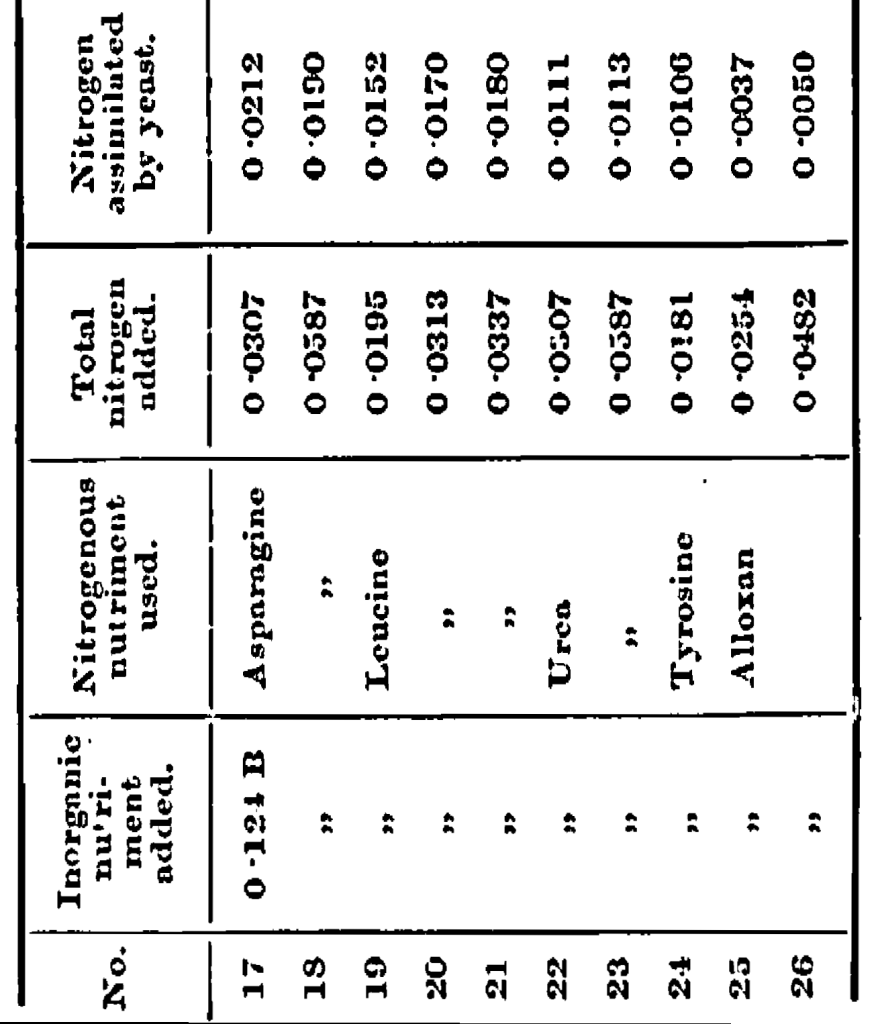
In order to test this, experiments were performed in which the asparagine was replaced by amounts of leucine, urea, tyrosine, or alloxan containing approximately the same amounts of nitrogen, all other conditions being kept as before. (I am indebted to the kindness of Mr. G. O’Sullivan, F.R.S., for the specimens of leucine and tyrosine.)
The results are shown in Table IV
The gases evolved during fermentation were free from any odour of sulphuretted hydrogen, and the fermentations in which the different nitrogenous nutriments were used, each had a distinctive odour, so that it was possible by this to recognise the particular nitrogenous food which had been added.
Nos. 17 and 18 give the mean of the experiments with asparagine described in Part I. Comparing these experiments with 19, 20 and 21, in which leucine was substituted for asparagine, it will be seen that the fermentations are as complete, the yeast, crop, the amount of nitrogen assimilated, and the percentage of nitrogen contained in the yeast are a little less, but not materially so. Next considering Experiments 22 and 23, in which urea was the nitrogenous food, the fermentations of the sugar are also as complete, but the yeast crop, the amount of nitrogen assimilated, and the percentage of nitrogen in the yeast are all materially less. Much the same may be said of Experiment 24, where tyrosine was the yeast food, although, owing to the small amount of this compound that could be used on account of its insolubility, the figures are not so comparable. With alloxan as the nitrogenous yeast food the amount of sugar unfermented is very high, and the values relating to the yeast crop, assimilation of nitrogen, and percentage of nitrogen in the yeast are very low, showing that this is not a suitable yeast food.
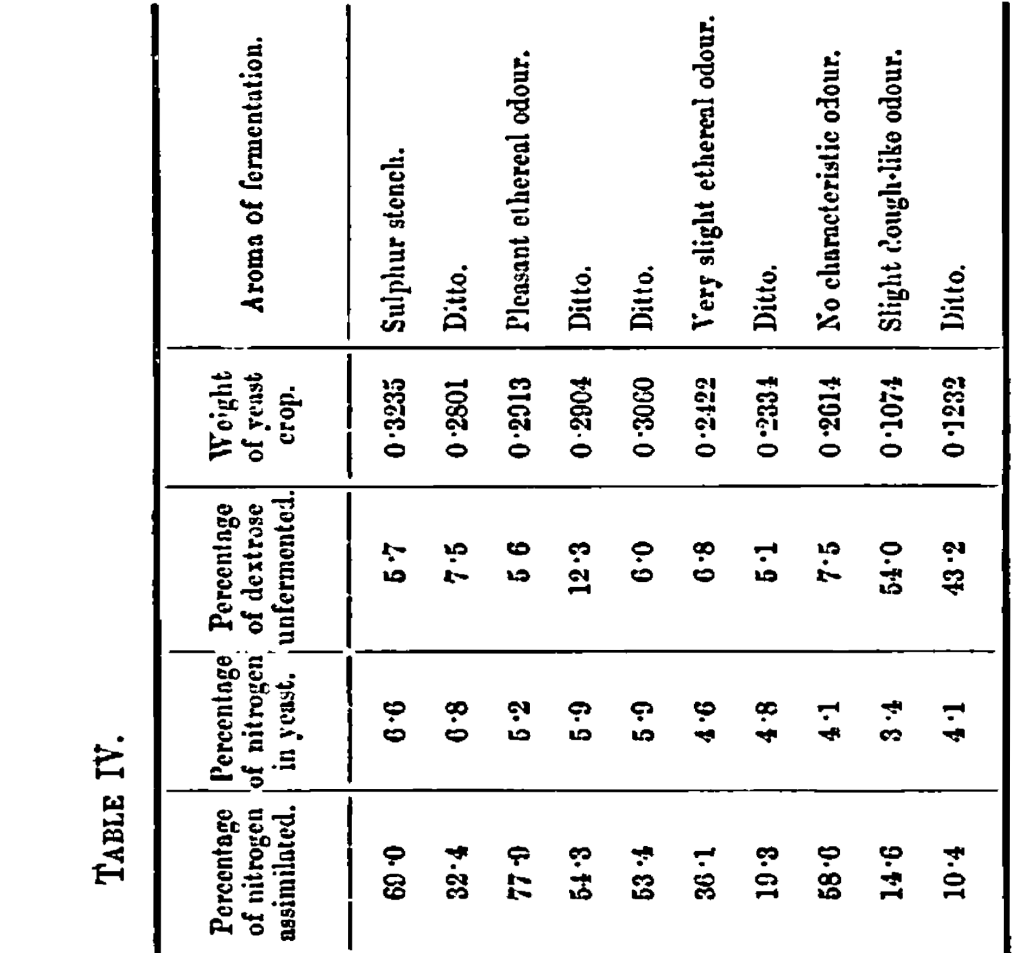
Besides showing that, other conditions being the same, the odour of fermentation is determined by the nitrogenous food used, these experiments also indicate that whilst many nitrogenous compounds can supply yeast with food that will enable it to ferment dextrose almost completely; yet with the same amount of nitrogen supply, they all differ as to the amount of yeast crop which is produced, the amount of nitrogen which is assimilated, and the percentage of nitrogen which is contained by the yeast.
There are also indications that the same general deductions, which were made as the result of the experiments described in Part I, will also be found to be true when nitrogenous food other than asparngine is employed. Further experiments on these points arc, however, desirable.
Discussion
The Chairman said it was rather difficult to follow the many tables that had been placed before them on the screen, and digest the results so as to be in a position to discuss the paper. To do that effectually, copies should be placed in their hands previously. Nevertheless, the paper which had been read was a very interesting one, and he knew, from personal experience, the importance of the subject. Where and how to obtain yeast foods were very important points indeed.
Mr. Weld-Blundell said he should like to ask a question. The author had told them that he had made his experiments from one cultivation of yeast which he obtained from Burton-on-Trent. Had Dr. Stern made any experiments on any other classes of yeast derived from other sources? It would be interesting to know whether yeast from Manchester, London, or other places yielded different quantities and percentages of nitrogenous matters.
Mr. T. A. Glendinning said he thought the question which Mr. Weld-Blundell had raised was a most important one. Dr. Stern had apparently dealt purely with Burton yeast. “Burton stench” was an expression known to all of them. That the presence of asparagine or other amide body was essential, or, at any rate, very favourable, to the production of “stench” but by no means proved that every yeast could produce “stench,” provided asparagine was present. It would be necessary to examine various yeasts to discover whether the property of producing “stench” was a common one under favourable circumstances, or whether the presence of a particular organism was essential. For years it had been known that a yeast which produced “sickness” or “stench” existed in Burton yeast, and only quite recently Dr. Frew and also Dr. Morris had directed attention to its isolation. With regard to the figures relating to the amount of nitrogen assimilated by yeast, it might he of practical value to determine in a malt wort to what extent the percentage of nitrogen could be decreased by the employment of sugar before the yeast began to deteriorate in vigour and power of reproduction.
Mr. Weld-Blundell said with reference to the smell—from a practical point of view, did it matter what the smell was like during fermentation so long as the smell was not present in the beer? Where the unpleasant smell existed, did the sulphuretted hydrogen mean imperfect fermentation or evil to the ultimate products? The smell might be perfectly harmless. When the fermentation was completely at an end, and the beer was clear and racked, would there be sufficient sulphuretted hydrogen given off to injure the beer? The most important point, he took it, was the amount of sugar which was left undecomposed and unaltered, and the extent of the crop produced. A yeast that was thoroughly healthy and sound, and acting vigorously, ought to reproduce to the maximum amount. That was more important than the amount of smell.
Mr. Hyde said he had tested fermentations when there was a smell from them; ho had also noticed the smell when using the same yeast with different malts, but failed to find any evidence of sulphuretted hydrogen. He remembered using at one time a lot of slack malt, two years old. He knew it was very unsatisfactory. If they got a good malt, the fermentation did not smell. Some time ago he made experiments, and afterwards read a paper on fermentation before the Institute. His experiments dealt with the amount of nitrogenous matter assimilated during fermentation. The amounts of yeast added to the vat were varying quantities, from 1 lb. upwards. He went up to 4 lbs. The result of his experiments was that where the fermentations were most vigorous, ho got the greatest amount of yeast reproduction, and the largest amount of nitrogeneous matter removed. The amount of yeast produced during normal fermentation was in direct proportion to the nitrogen removed. Ho would ask the author whether ho found some difficulty in drying yeast. His experience was that it was a most difficult thing to accomplish, and in some experiments he had made the amounts of nitrogenous matter in yeast varied, and he attributed the discrepancy to the moisture.
Dr. Stern, in briefly replying, said he hoped to publish Part III of his researches on “The Nutrition of Yeast” shortly, and other parts might follow. Ho wished to work out one experimental condition at a time. So far he had worked with Burton yeast. Hereafter, he might use other yeasts. The same statement applied to the use of other nitrogenous matter, wort, &c. When he spoke of sulphuretted hydrogen as the cause of the stench, ho meant to include also its volatile compounds with alcohol and other constituents of the fermenting solution. The point he most particularly wished to emphasize was that the odour of the gases evolved during fermentation varied with the nature of the nitrogenous yeast food. What the aromas actually were he could not tell. Their weight was very small, and he had not attempted to estimate them. Unpleasant odours were probably produced by many different causes, and in this paper he only dealt with one particular one. In reply to Mr. Hyde, ho said it took time to dry the yeast, but he had never found any difficulty with the small weights he had to deal with.
Dr. A. K. Miller proposed a vote of thanks to Dr. Stern for his paper. The subject was of great importance, and its treatment by the author had been very interesting. The number of experiments he had described to them must have occupied a great deal of time and attention. It was best to work under one set of conditions at a time. Further results would, he hoped, in due time be placed before thorn.
Mr. Hyde seconded the motion, which was carried unanimously.
The author having suitably tendered his acknowledgment of the vote of thanks, the proceedings terminated.